Neutralization Reactions Neutralization Reaction between an acid and

Neutralization Reactions

Neutralization Reaction between an acid and a base Products: salt (ionic compound) and water Double replacement reaction Example: Na. OH + HCl Na. Cl + H 2 O

The products do not necessarily have a p. H of 7 Depends entirely on the strength of acid and base reactants Real life examples Antacids neutralize your stomach acid Fertilizers (lime) can be used on soils that are too acidic

Practice problem 1 What are the products of the neutralization reaction between Mg(OH)2 and HBr?
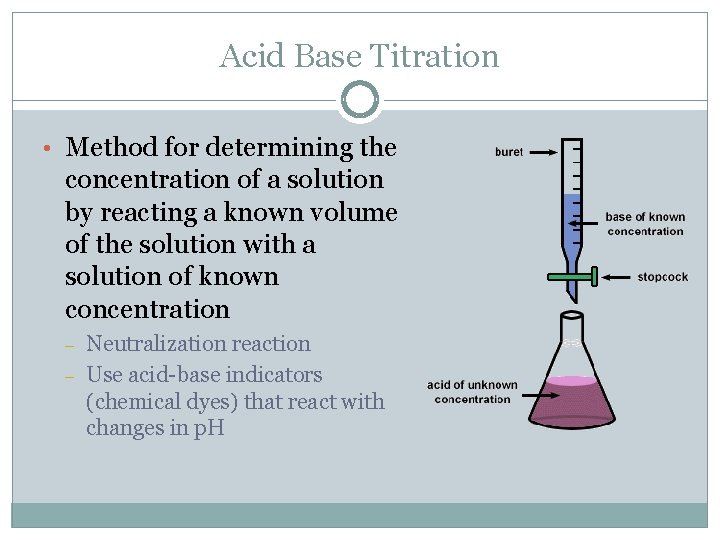
Acid Base Titration • Method for determining the concentration of a solution by reacting a known volume of the solution with a solution of known concentration – – Neutralization reaction Use acid-base indicators (chemical dyes) that react with changes in p. H

Procedure • Measure volume of solution with unknown concentration • Have buret filled with titrating solution of known concentration (standard solution) • Measured volumes of standard are added slowly until acid-base indicator (chemical dye) changes color to indicate the end point (equivalence point) where the acid and base have neutralized each other
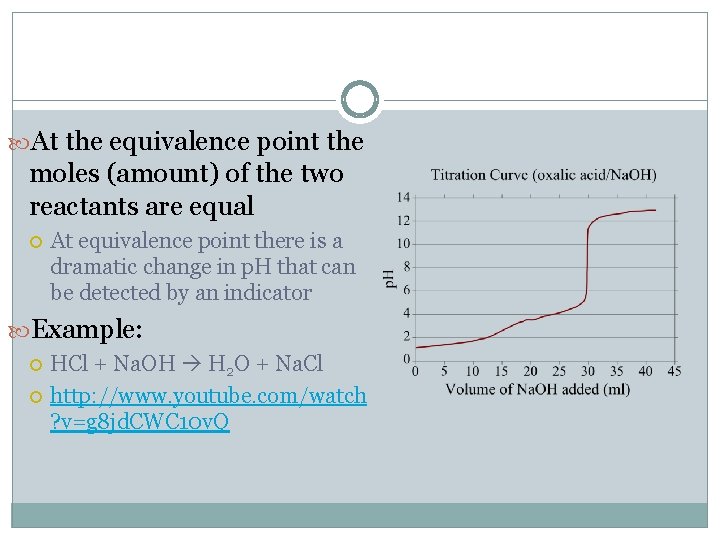
At the equivalence point the moles (amount) of the two reactants are equal At equivalence point there is a dramatic change in p. H that can be detected by an indicator Example: HCl + Na. OH H 2 O + Na. Cl http: //www. youtube. com/watch ? v=g 8 jd. CWC 10 v. Q

http: //www. youtube. com/watch? v=Iiu 5 mc. AA 8 p. U
- Slides: 8