Neutralization Reaction NEUTRALIZATION REACTION DOUBLE REPLACEMENT RX an
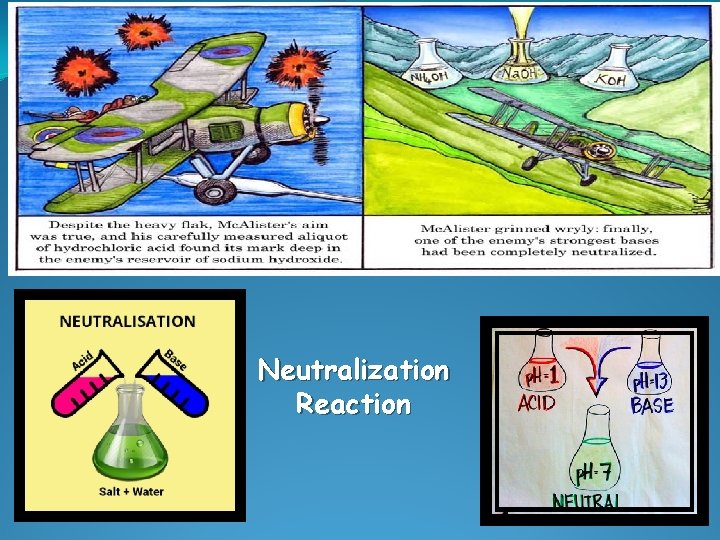
Neutralization Reaction
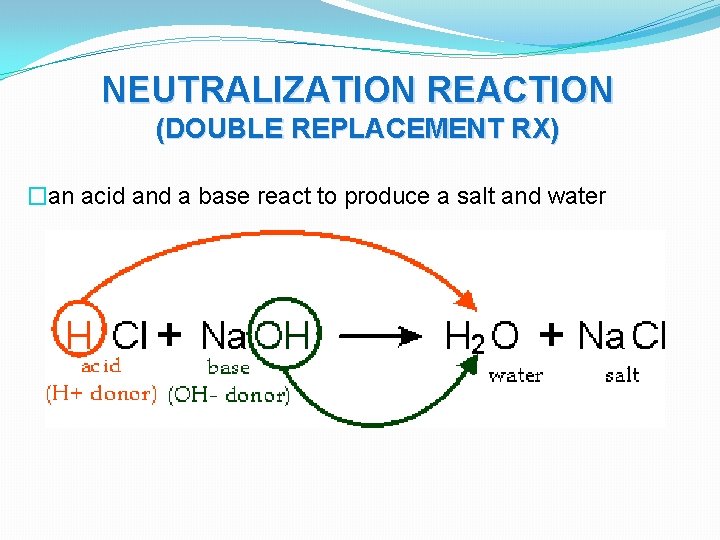
NEUTRALIZATION REACTION (DOUBLE REPLACEMENT RX) �an acid and a base react to produce a salt and water

NEUTRALIZATION REACTION (DOUBLE REPLACEMENT RX) � HBr + KOH KBr + HOH � H 2 SO 4 + 2 NH 4 OH (NH 4)2 SO 4 + 2 H 2 O � 2 Na. OH + H 2 CO 3 Na 2 CO 3 + 2 H 2 O
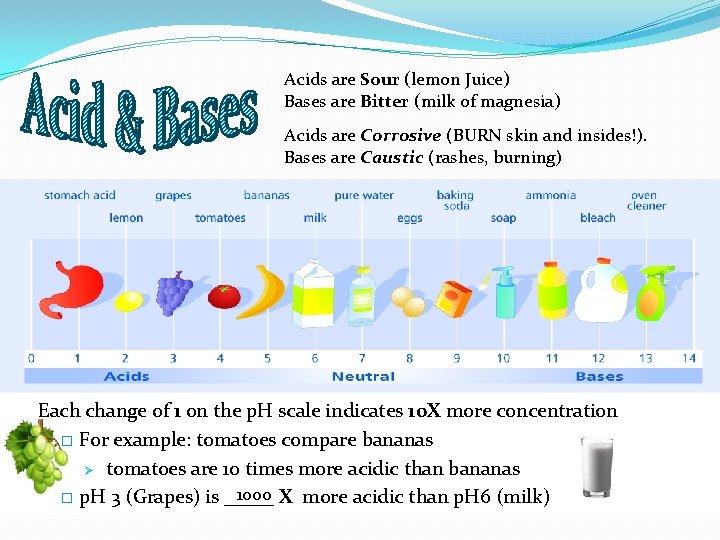
Acids are Sour (lemon Juice) Bases are Bitter (milk of magnesia) Acids are Corrosive (BURN skin and insides!). Bases are Caustic (rashes, burning) Each change of 1 on the p. H scale indicates 10 X more concentration � For example: tomatoes compare bananas Ø tomatoes are 10 times more acidic than bananas 1000 X more acidic than p. H 6 (milk) � p. H 3 (Grapes) is _____
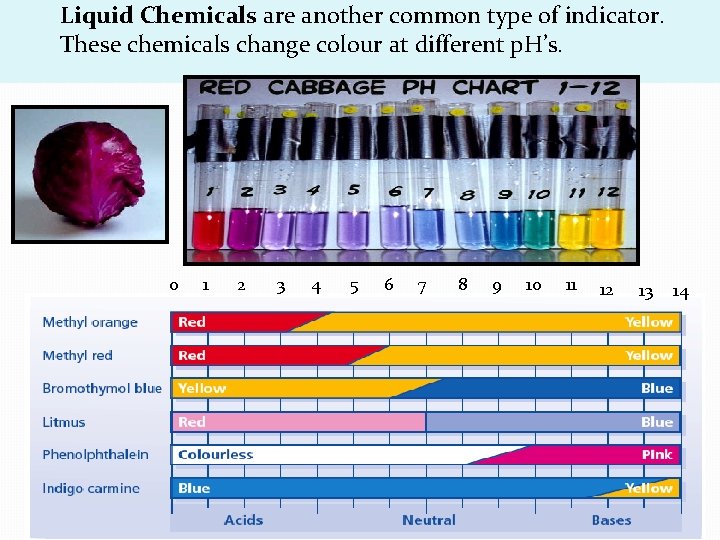
Liquid Chemicals are another common type of indicator. These chemicals change colour at different p. H’s. 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14

There are easy way to tell if something is an Acid or a Base… �Acid usually start with Hydrogen (H__) � HCl = Hydrochloric Acid � H 2 SO 4 = Sulphuric acid � H 2 SO 3 = Sulphurous acid �Base usually ends with hydroxide (__OH) � Na. OH = Sodium Hydroxide � NH 4 OH(aq) = Ammonium Hydroxide
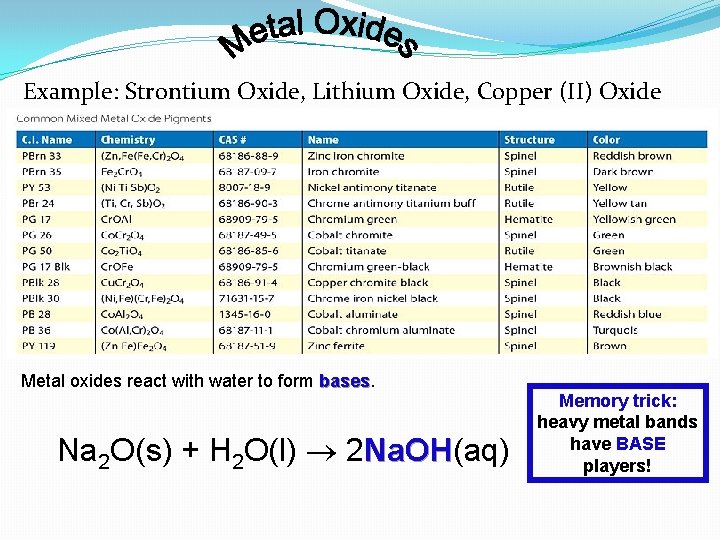
Example: Strontium Oxide, Lithium Oxide, Copper (II) Oxide Metal oxides react with water to form bases Na 2 O(s) + H 2 O(l) 2 Na. OH(aq) Na. OH Memory trick: heavy metal bands have BASE players!

Examples: carbon monoxide, carbon dioxide, sulphur dioxide Non-metal oxides react with water to form acids SO 2(g) + H 2 O(l) H 2 SO 3(aq) Memory trick: pollution makes ACID RAIN!
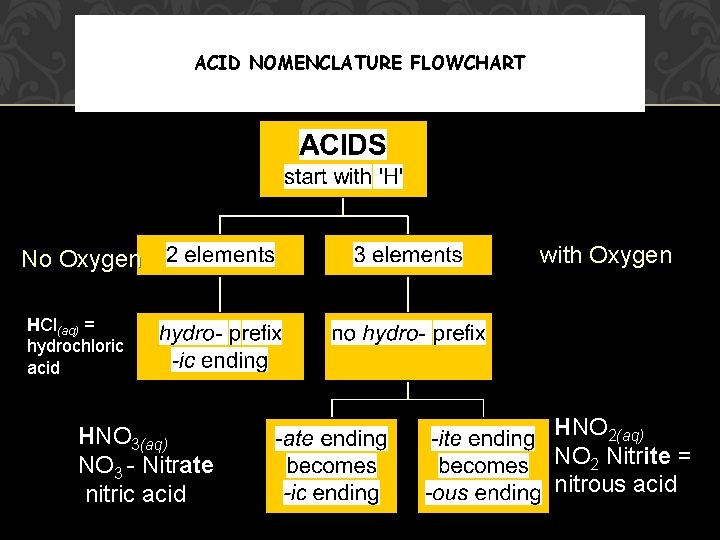
ACID NOMENCLATURE FLOWCHART No Oxygen with Oxygen HCl(aq) = hydrochloric acid HNO 3(aq) NO 3 - Nitrate nitric acid HNO 2(aq) NO 2 Nitrite = nitrous acid

ACID NOMENCLATURE • HBr • H 2 CO 3 • H 2 SO 3 hydrobromic acid carbonic acid sulfurous acid

- Slides: 11