NEUTRALISATION Learning Objectives Describe Demonstrate neutralisation in neutralisation
NEUTRALISATION Learning Objectives Describe Demonstrate neutralisation in neutralisation with the real world word equations KEYWORD: neutralisation Your keyword for today describes a type of reaction. What do you think it is about? Look at the start of the word to help you neutralisation

Mixing an acid and a base GROUP THOUGHT: What happens when an acid an alkali are mixed? + acid alkalki What will be the outcome of mixing dilute hydrochloric acid with the base sodium hydroxide? 1. Pour 2. 0 m. L of 1 M sodium hydroxide solution into a clean evaporating dish. 2. Add only one drop of phenolphthalein indicator solution to the sodium hydroxide. 3. Add, drop by drop, 1 M hydrochloric acid to the evaporating dish, using a dropper. Swirl constantly while adding the acid. Continue adding acid until the colour of the solution just disappears within one drop. Then add one more drop.

Mixing an acid and a base GROUP THOUGHT: What happens when an acid an alkali are mixed? + acid alkalki What will be the outcome of mixing dilute hydrochloric acid with the base sodium hydroxide? HCl (aq) + Na. OH (aq) Na. Cl (aq) + H 2 O (l) How do you know this chemical reaction has taken place? The p. H of the mixture will become closer to p. H 7. The reaction of an acid and a base is, therefore, an example of neutralisation.

The products of neutralisation What are the products of the reaction between hydrochloric acid and sodium hydroxide? HCl (aq) + Na. OH (aq) Na. Cl (aq) + H 2 O (l) hydrochloric acid + sodium hydroxide sodium chloride + water Sodium chloride and water are produced. Sodium chloride is a salt. A salt is a substance in which the hydrogen ion of an acid has been replaced by a metal or other positive ion. In this reaction the hydrogen ion of hydrochloric acid has been replaced by a sodium ion.
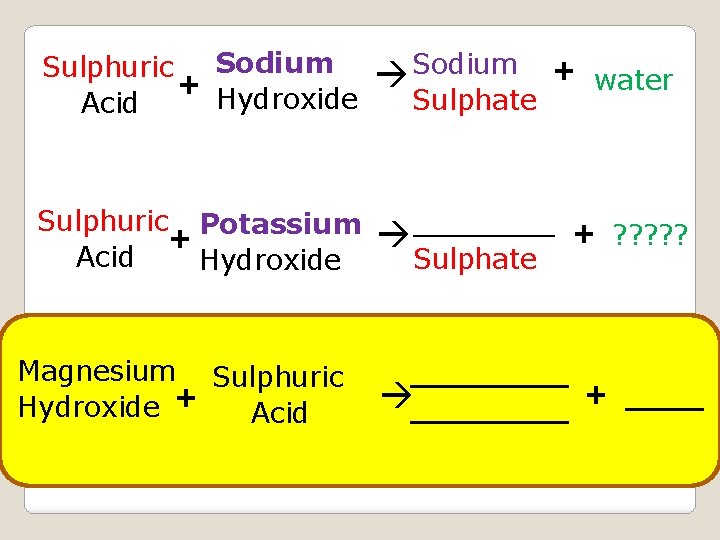
Sodium Sulphuric + Hydroxide Acid Sodium + Sulphuric Potassium + Acid Hydroxide ____ + Magnesium Sulphuric Hydroxide + Acid Sulphate water Sulphate ________ + ? ? ? ____

SUMMARY • Neutralisation : a reaction that happens between an ______ and a _____. It produces a _______ solution • Neutralisation produces _____ and ____. • A ____ is a substance in which the hydrogen ion of an acid has been replaced by a metal or other positive ion. – Salts are IONIC compounds
- Slides: 6