Neuroplasticity Neurobiologie Les 3 1 st Master Biomedische

Neuroplasticity Neurobiologie Les 3 1 st Master Biomedische Wetenschappen Robrecht Raedt

Overview • Introduction • Synaptic plasticity – Short term plasticity – Learning and memory mechanisms • Short-term sensitization/long-term sensitization • Long-term potentiation • Long-term depression • • • Intrinsic neural plasticity Homeostatic plasticity Memory systems in the mammalian brain Cortical Neuroplasticity and neuro-prostheses Deep brain stimulation

Introduction on neuroplasticity • Neuroplasticity = changes in activity and organization of the brain due to ‘experience’ • Changes: – Physiological – Anatomical • Previous dogma’s: – The brain is rigid – Plasticity is limited to the hippocampus – Plasticity is limited to development/childhood • All brain regions show some form of plasticity, even in adulthood

Synaptic plasticity • Changes in input-output relationship in neuronal networks due to changes in synaptic efficacy – Excitatory/inhibitory – Activity-dependent – Different time scales: milliseconds, hours, days • Short-term plasticity (msec-min) • Long-term plasticity (min-lifetime)
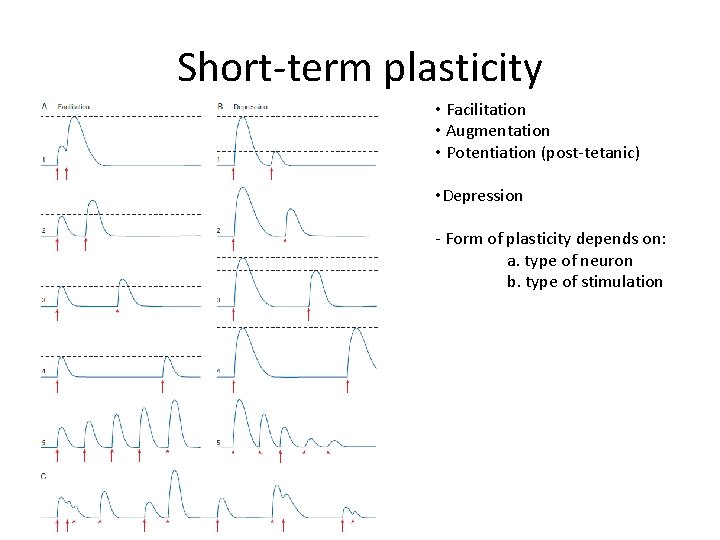
Short-term plasticity • Facilitation • Augmentation • Potentiation (post-tetanic) • Depression - Form of plasticity depends on: a. type of neuron b. type of stimulation

Short-term plasticity • Mechanism: Changes in calcium-concentration Changes in neurotransmitter release (quanta) PRESYNAPTIC Repeated neuronal activity - more: facilitation/augmentation/potentiation -less: depression
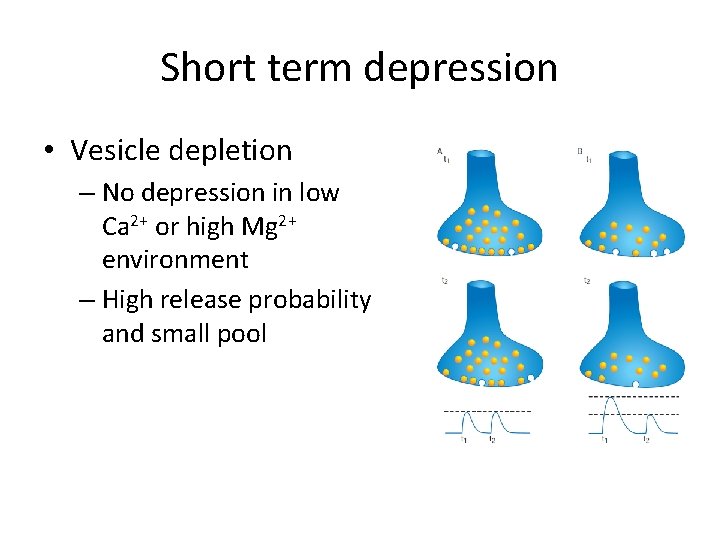
Short term depression • Vesicle depletion – No depression in low Ca 2+ or high Mg 2+ environment – High release probability and small pool
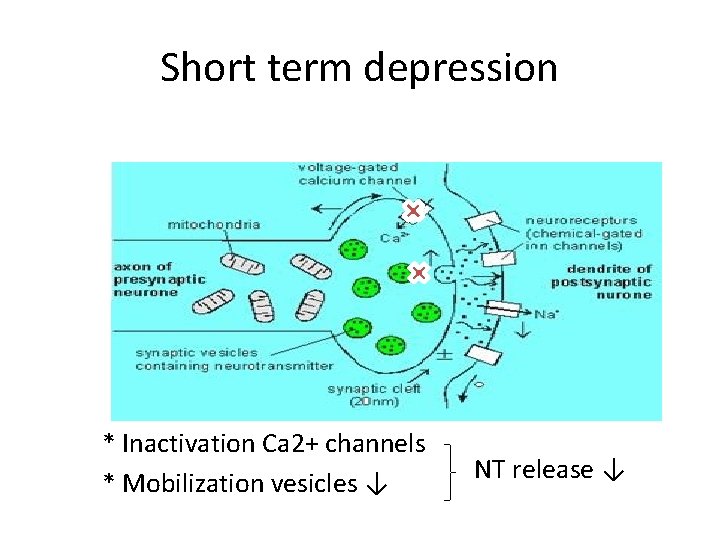
Short term depression * Inactivation Ca 2+ channels * Mobilization vesicles ↓ NT release ↓
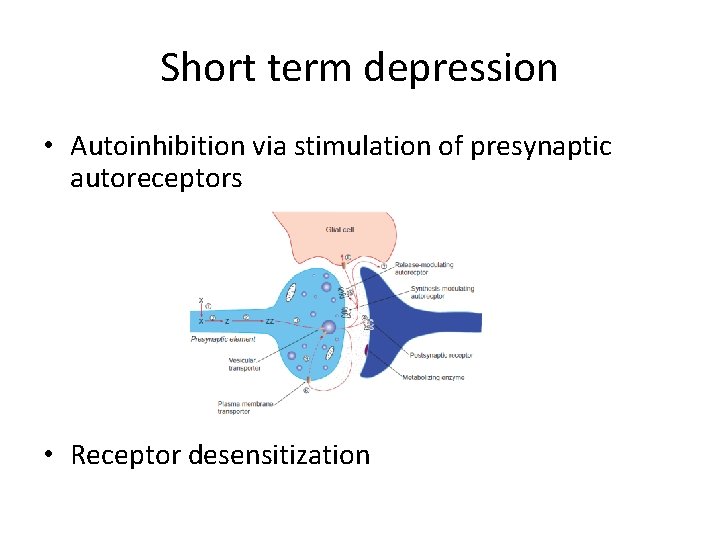
Short term depression • Autoinhibition via stimulation of presynaptic autoreceptors • Receptor desensitization
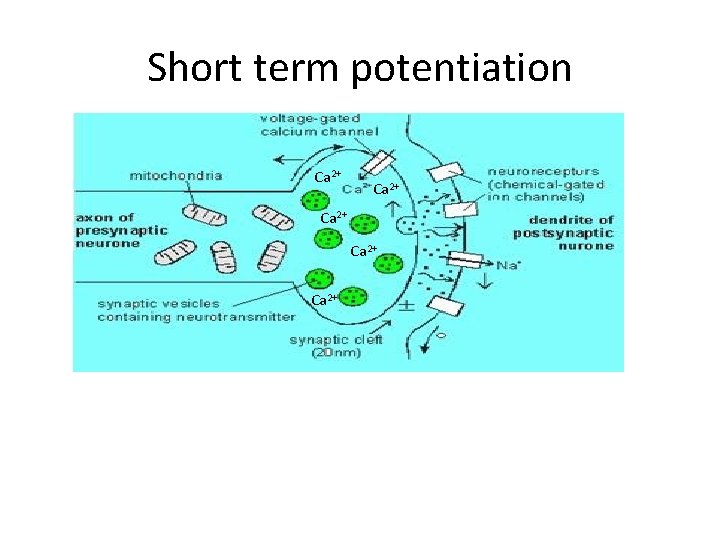
Short term potentiation Ca 2+ Ca 2+

Short term potentiation • Residual Ca 2+ remaining in active zones after presynaptic activity • Summating with Ca 2+ peak during subsequent action potentials at site triggering exocytosis • More distant facilitation sites (second messengers systems/kinases) • Potentiation: longer period after strong tetanus – Overloading of processes responsible for removing excess Ca 2+ • Ca 2+ extrusion pumps • Plasma membrane ATPase and Na+- Ca 2+ exhange • Ca 2+ uptake in organelles

Learning and memory • Long-term plasticity • Repeated synaptic activity → changes last for hours/days – Sensitization – Long-term potentiation – Long-term depression – Intrinsic synaptic plasticity – Homeostatic plasticity
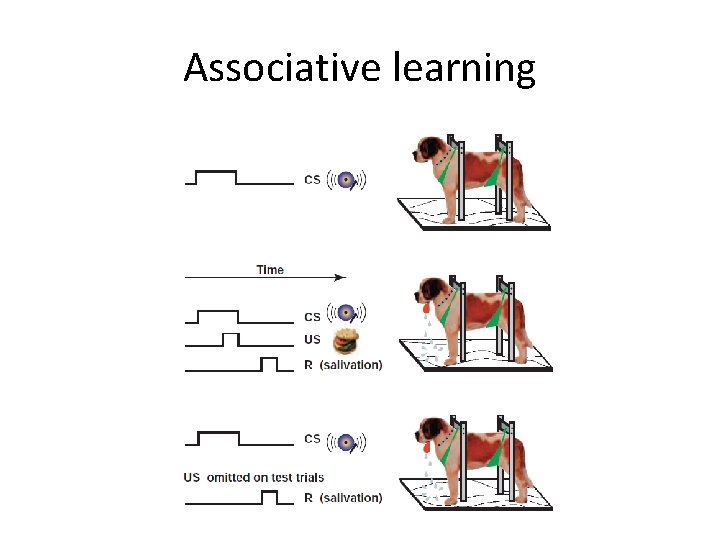
Associative learning

Non-associative learning • Habituation : reduction in response to a stimulus • Dishabituation: restoration/recovery of a response due to presentation of another strong stimulus • Sensitization: enhancement of response due to presentation of a strong stimulus

Aplysia studies • Kandel: Nobel Prize in Physiology or Medicine in 2000 • Simple nervous system (few cells) • Accessible for detailed anatomical, biophysical, biochemical and molecular studies • Neurons and neural circuits that mediate behavior have been identified • Changes during learning have been identified • Memory mechanisms – Induction – Expression – Maintenance (consolidation)
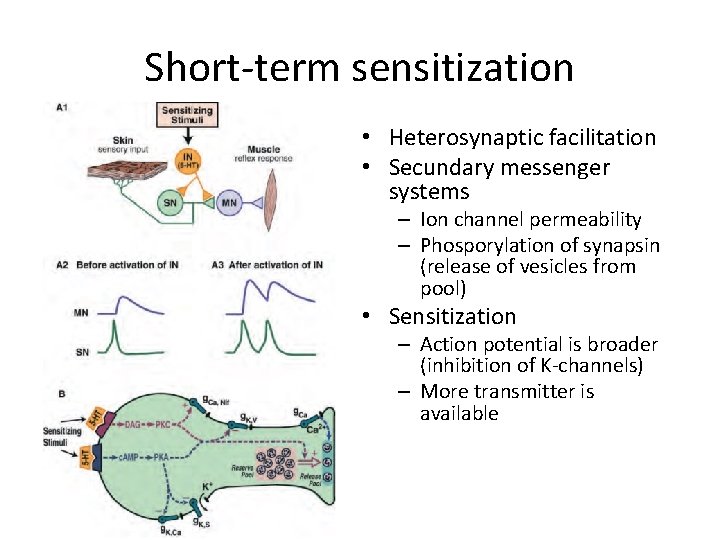
Short-term sensitization • Heterosynaptic facilitation • Secundary messenger systems – Ion channel permeability – Phosporylation of synapsin (release of vesicles from pool) • Sensitization – Action potential is broader (inhibition of K-channels) – More transmitter is available
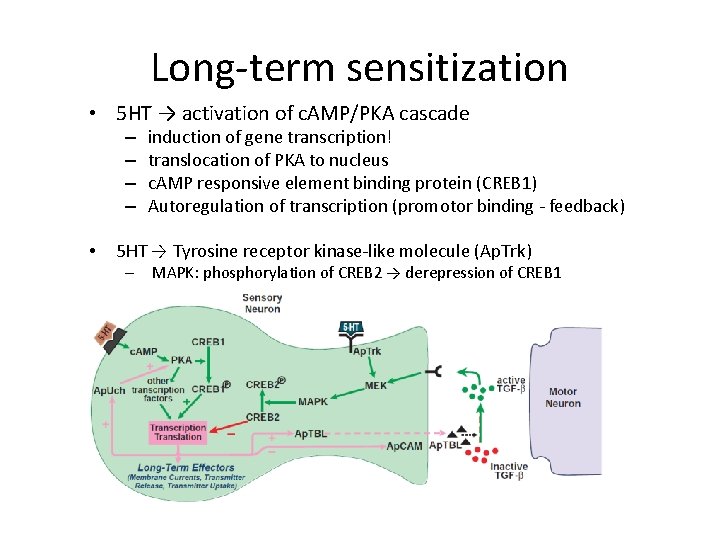
Long-term sensitization • 5 HT → activation of c. AMP/PKA cascade – – • induction of gene transcription! translocation of PKA to nucleus c. AMP responsive element binding protein (CREB 1) Autoregulation of transcription (promotor binding - feedback) 5 HT → Tyrosine receptor kinase-like molecule (Ap. Trk) – MAPK: phosphorylation of CREB 2 → derepression of CREB 1
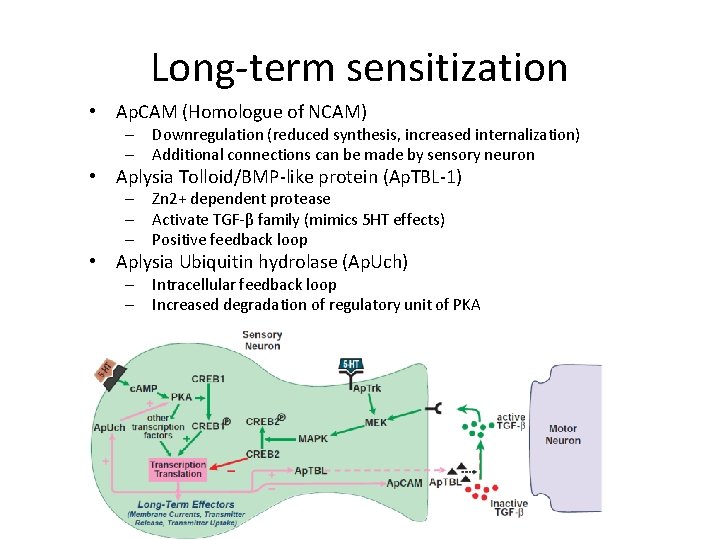
Long-term sensitization • Ap. CAM (Homologue of NCAM) – Downregulation (reduced synthesis, increased internalization) – Additional connections can be made by sensory neuron • Aplysia Tolloid/BMP-like protein (Ap. TBL-1) – Zn 2+ dependent protease – Activate TGF-β family (mimics 5 HT effects) – Positive feedback loop • Aplysia Ubiquitin hydrolase (Ap. Uch) – Intracellular feedback loop – Increased degradation of regulatory unit of PKA

Long-term vs. short-term sensitization • Decreased duration of AP • Structural changes: neurite outgrowth • Increased high-affinity glutamate uptake – Nt. available for release – Nt. clearance (duration of EPSP/receptor desensitization) • Changes in postsynaptic cell
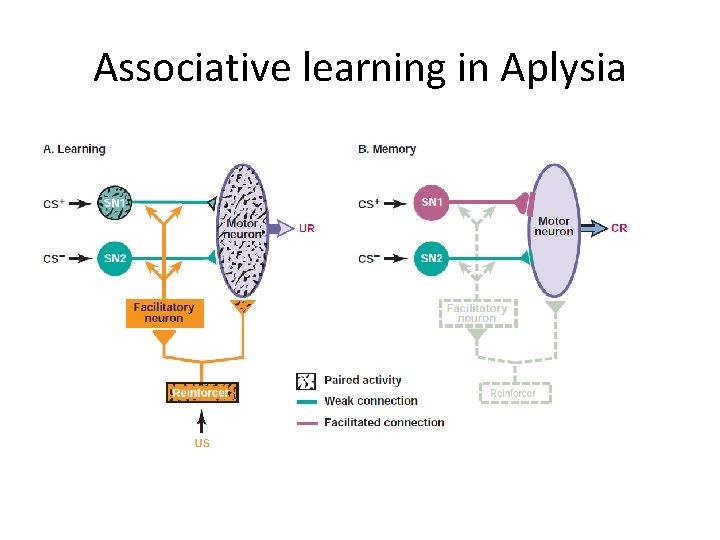
Associative learning in Aplysia
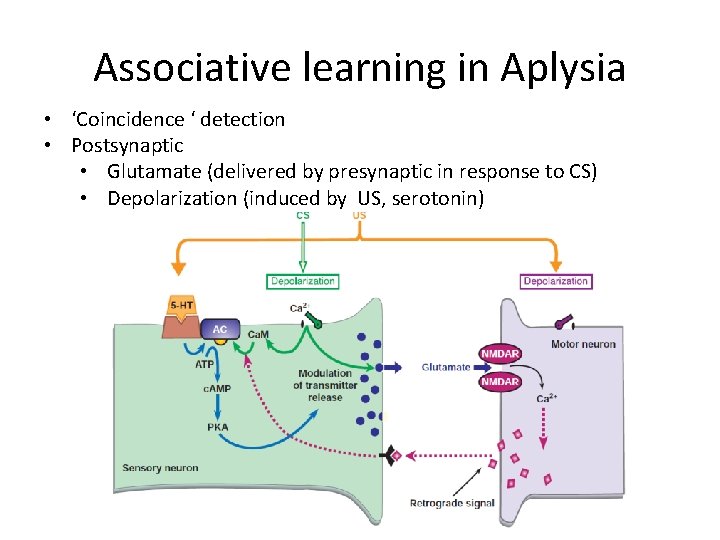
Associative learning in Aplysia • ‘Coincidence ‘ detection • Postsynaptic • Glutamate (delivered by presynaptic in response to CS) • Depolarization (induced by US, serotonin)

Vertebrate studies: LTP More difficult to link synaptic plasticity with learning Increase in synaptic strength Induced by brief burst of spike activity in presynaptic afferents Responsible for information storage in several brain regions, different animal models • No uniform mechanism for inducing LTP • • – Depending on experimental conditions
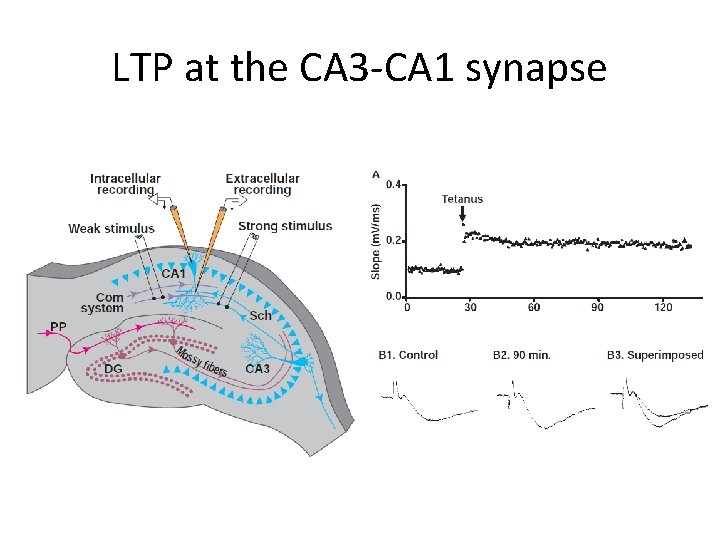
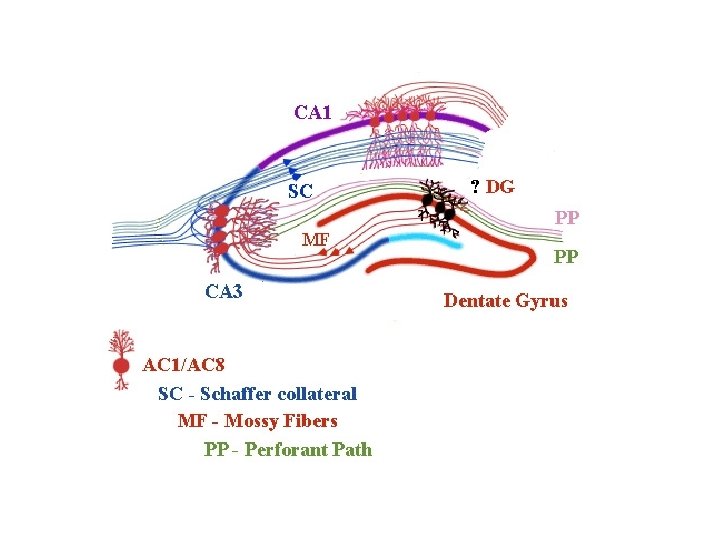
LTP at the CA 3 -CA 1 synapse
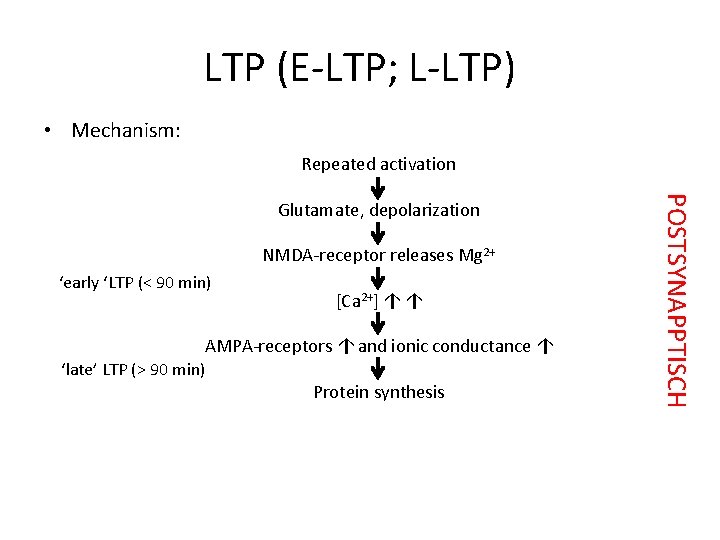
LTP (E-LTP; L-LTP) • Mechanism: Repeated activation NMDA-receptor releases Mg 2+ ‘early ‘LTP (< 90 min) [Ca 2+] ↑ ↑ AMPA-receptors ↑ and ionic conductance ↑ ‘late’ LTP (> 90 min) Protein synthesis POSTSYNAPPTISCH Glutamate, depolarization

LTP • Classical properties: – Cooperativity: probability of LTP, magnitude of change increases with number of stimulated afferents – Associativity: LTP only induced at weak input when associated with activity in strong input – Input specificity: Unstimulated weak pathway not facilitated after tetanus of strong pathway

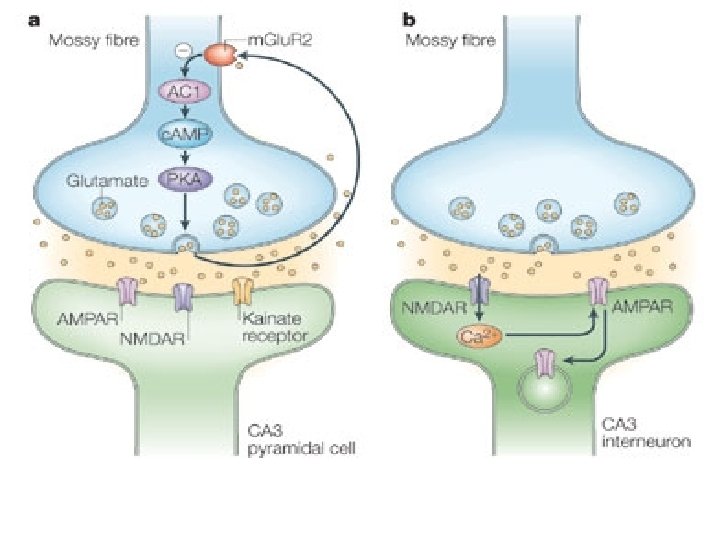
Hebbian Mechanism • Donald Hebb (1949): ‘When an axon of cell A is near enough to excite a cell B and repeatedly or persistently takes part in firing it, some growth process or metabolic change takes place in one or both cells such that A’s efficiency, as one of the cells firing B, is increased. ’ • ‘Cells that fire together, wire together’ • Coincident activity in two synaptically coupled neurons increases the synaptic strength between them • Not all forms of LTP obey Hebb’s law: e. g. Mossy fiber-CA 3 synapse
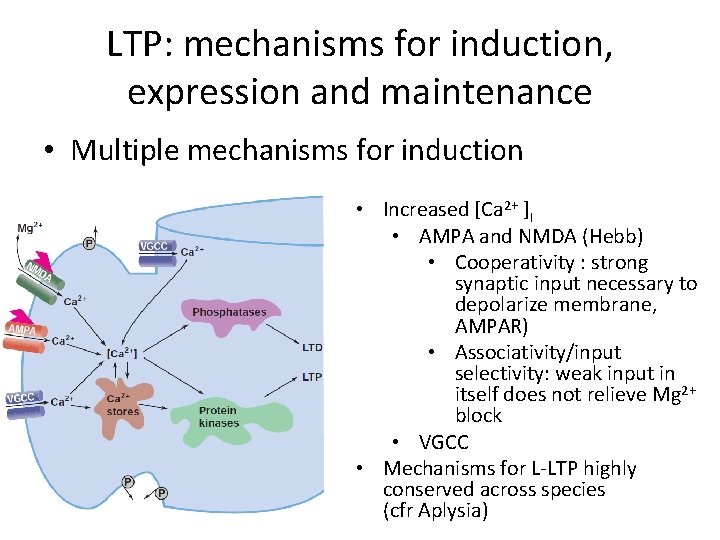
LTP: mechanisms for induction, expression and maintenance • Multiple mechanisms for induction • Increased [Ca 2+ ]I • AMPA and NMDA (Hebb) • Cooperativity : strong synaptic input necessary to depolarize membrane, AMPAR) • Associativity/input selectivity: weak input in itself does not relieve Mg 2+ block • VGCC • Mechanisms for L-LTP highly conserved across species (cfr Aplysia)
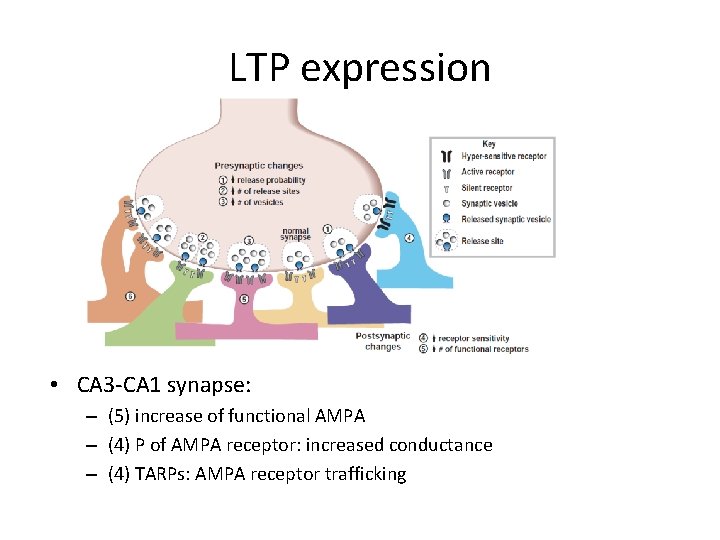
LTP expression • CA 3 -CA 1 synapse: – (5) increase of functional AMPA – (4) P of AMPA receptor: increased conductance – (4) TARPs: AMPA receptor trafficking

LTP maintenance • E-LTP: phosphorylation of substate protein • L-LTP: alteration in gene expression – Transcription factors (fos, zif 268) – Cytoskeletal proeins (arc) – Signal transduction molecules (Ca. M kinase II) – Critical time window (<2 h) – Synapse specificity: tagging by kinase(s) – Positive feedback/re-activation of L-LTP mechanisms
![Long term depression Depolarization NMDA-receptor releases Mg 2+ [Ca 2+] ↑ AMPA-receptor defosforylatie internalisation Long term depression Depolarization NMDA-receptor releases Mg 2+ [Ca 2+] ↑ AMPA-receptor defosforylatie internalisation](http://slidetodoc.com/presentation_image_h/9c40c8acc3dee19ac042010f49576f95/image-30.jpg)
Long term depression Depolarization NMDA-receptor releases Mg 2+ [Ca 2+] ↑ AMPA-receptor defosforylatie internalisation AMPA-receptors • Learning mechanism in cerebellum (eye-blink reflex: decrease in synaptic strength in a postsynaptic inhibitory neuron) • Reversal of LTP • NMDA-dependent and – independent mechanisms POSTSYNAPPTISCH Repeated activity (Hippocampus: 10 min, 1 Hz)
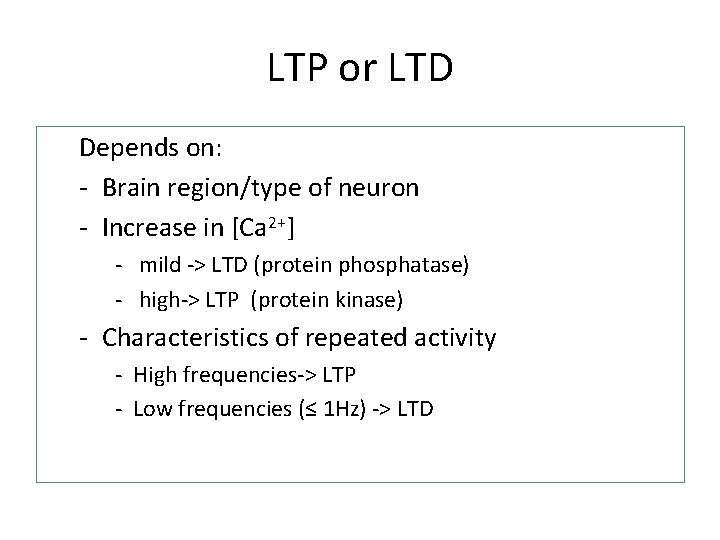
LTP or LTD Depends on: - Brain region/type of neuron - Increase in [Ca 2+] - mild -> LTD (protein phosphatase) - high-> LTP (protein kinase) - Characteristics of repeated activity - High frequencies-> LTP - Low frequencies (≤ 1 Hz) -> LTD

Intrinsic neural plasticity • Changes in input-output relationship in neuronal networks due to changes in density or functional properties of voltagegated ion channels • Probability that a cell fires in response to depolarization by EPSP • EPSP to spike coupling • Different between neural dendrites, soma and axons
Intrinsic neural plasticity

Intrinsic neural plasticity • Dendritic ion channels – Voltage attenuation of EPSPs, EPSP to AP – Voltage attenuation and filtering of backpropagating AP • STDP (spike-timing dependent plasticity) – Voltage gated Na+ and Ca 2+ channels allow dendrites to generate own spikes (dendritic spikes)
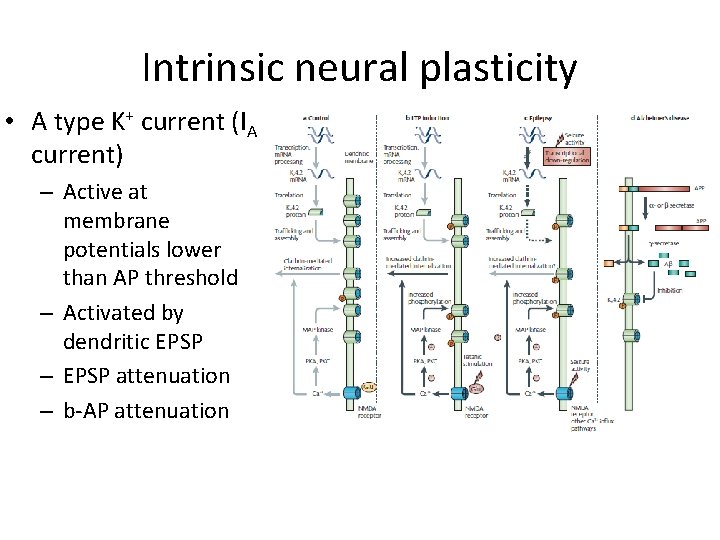
Intrinsic neural plasticity • A type K+ current (IA current) – Active at membrane potentials lower than AP threshold – Activated by dendritic EPSP – EPSP attenuation – b-AP attenuation

Homeostatic plasticity • Allow neurons to sense how active they are and to adjust their properties to maintain stable function • Stabilizes the activity of a neuron or neuronal circuit in the face of perturbations that alter excitability (e. g. changes in number of synapses) 1. 2. 3. 4. Synaptic scaling Regulation of intrinsic neuronal excitability Regulation of synapse number ‘Metaplasticity’
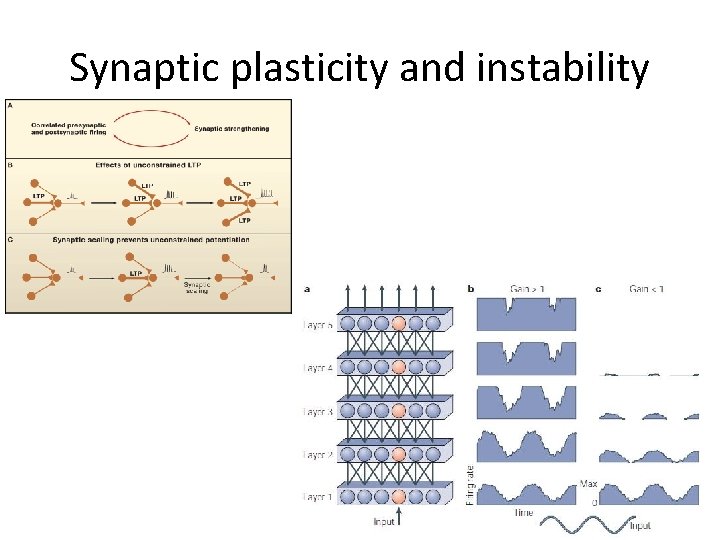
Synaptic plasticity and instability
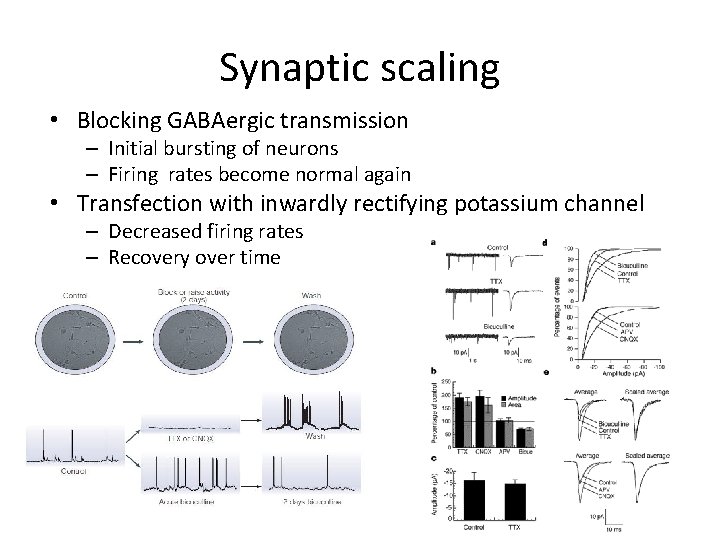
Synaptic scaling • Blocking GABAergic transmission – Initial bursting of neurons – Firing rates become normal again • Transfection with inwardly rectifying potassium channel – Decreased firing rates – Recovery over time
Synaptic scaling
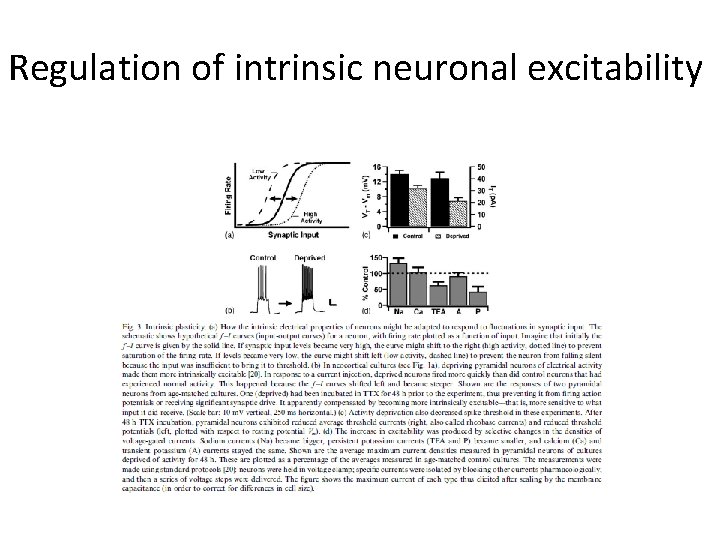
Regulation of intrinsic neuronal excitability
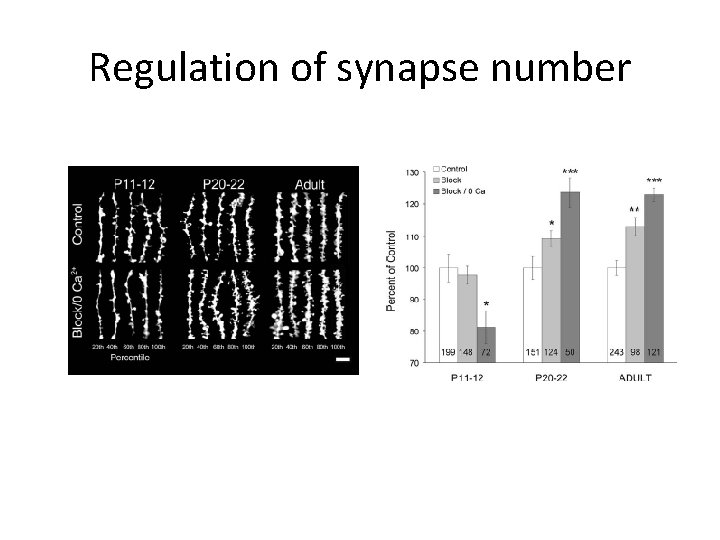
Regulation of synapse number
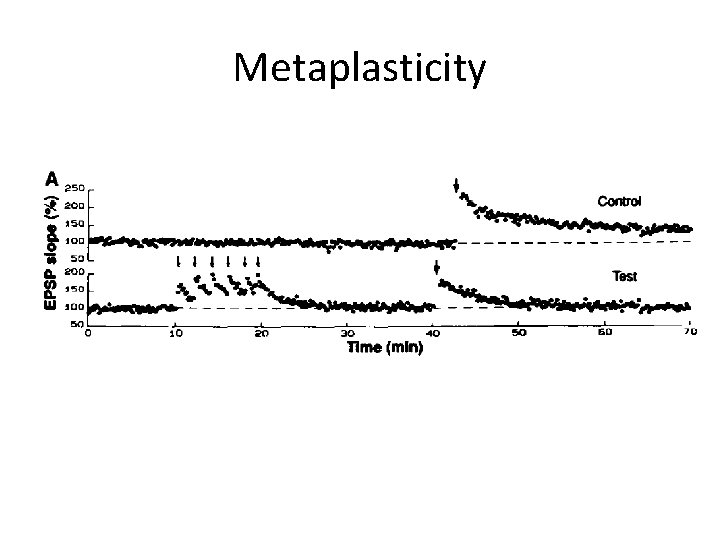
Metaplasticity
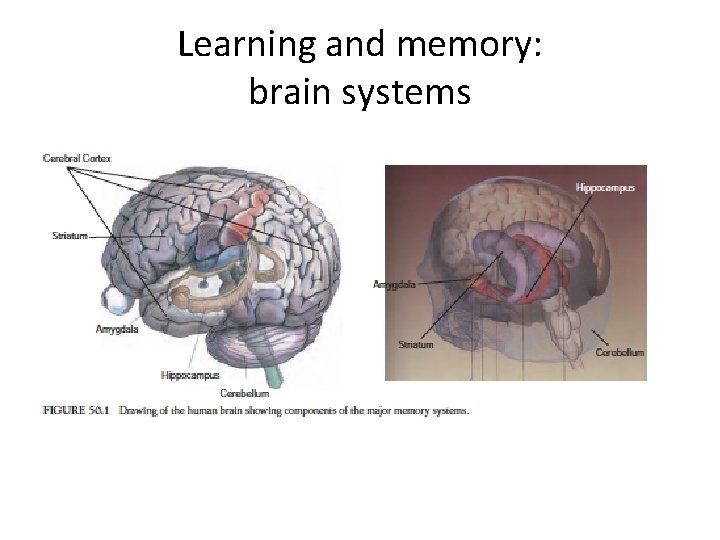
Learning and memory: brain systems
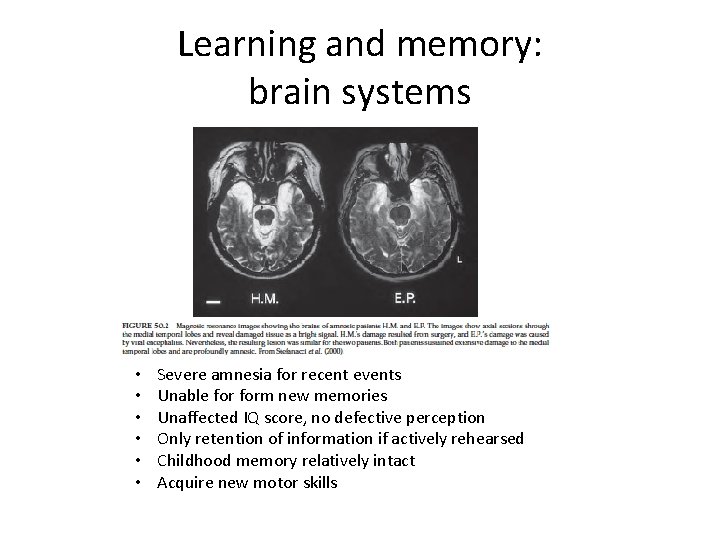
Learning and memory: brain systems • • • Severe amnesia for recent events Unable form new memories Unaffected IQ score, no defective perception Only retention of information if actively rehearsed Childhood memory relatively intact Acquire new motor skills

• Declarative (explicit) memory – episodic memory • personal events – semantic memory • learning new facts • Procedural (implicit) memory
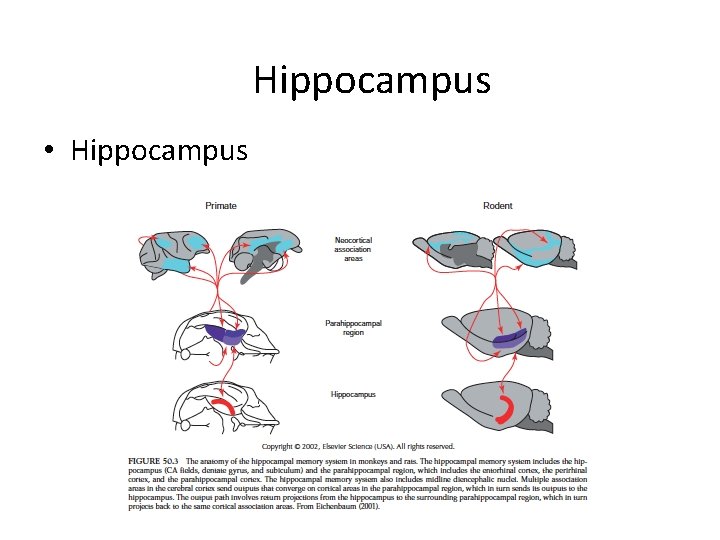
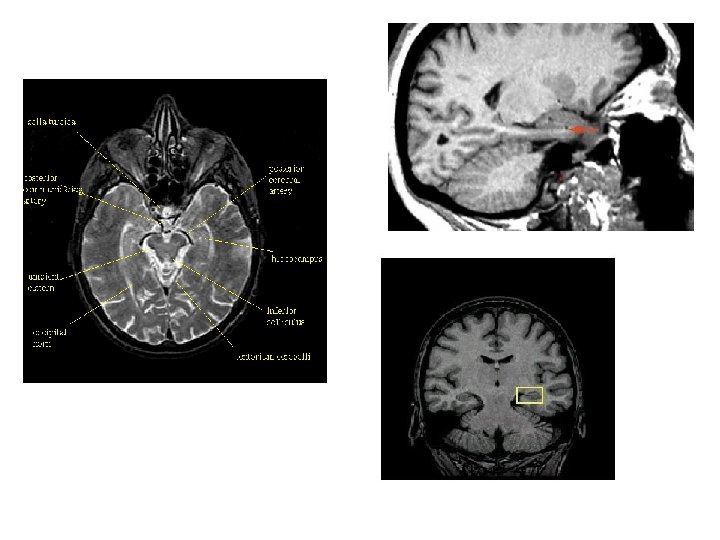
Hippocampus • Hippocampus

Hippocampus • the subiculum • hippocampus = hippocampus proper = Ammon’s horn • dentate gyrus – a thin band of cortex that lies on the upper surface of the parahippocampal gyrus. – an input centre and receives signals that are relayed to it via the enthorhinal cortex and its cells project to cells in the hippocampal formation.

dentate gyrus (1) cornu ammonis (2) Their three layered cortex is continuous below with the subiculum (3) which has four, five then six layers as it merges with the parahippocampal gyrus (4).

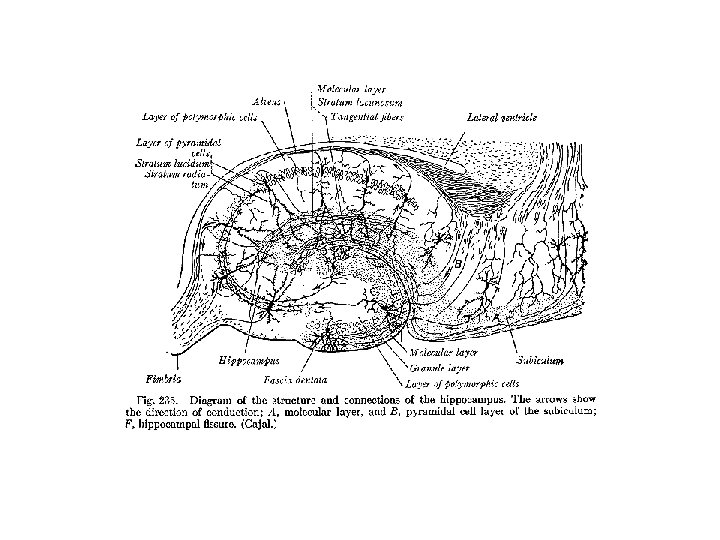
Hippocampal formation • subiculum – transitional area between 3 -layered hippocampus and 5 layered parahippocampal gyrus – area essential for flow of information into hippocampal formation

Hippocampal formation • dentate gyrus and hippocampus – 3 -layered – external layer: molecular layer with afferent axons and dendrites – middle layer: granule cell layer in dentate gyrus and pyramidal layer in hippocampus with efferent neurons – inner layer: polymorphic layer: axons of granule and pyramidal cells, intrinsic neurons and many glial cells

Hippocampal formation • 4 regions: CA 1 -CA 4 (CA: cornu Ammonis) – CA 1: located at subiculum-hippocampal interface – CA 2 and CA 3: located in hippocampus – CA 4: located at junction of hippocampus and dentate gyrus
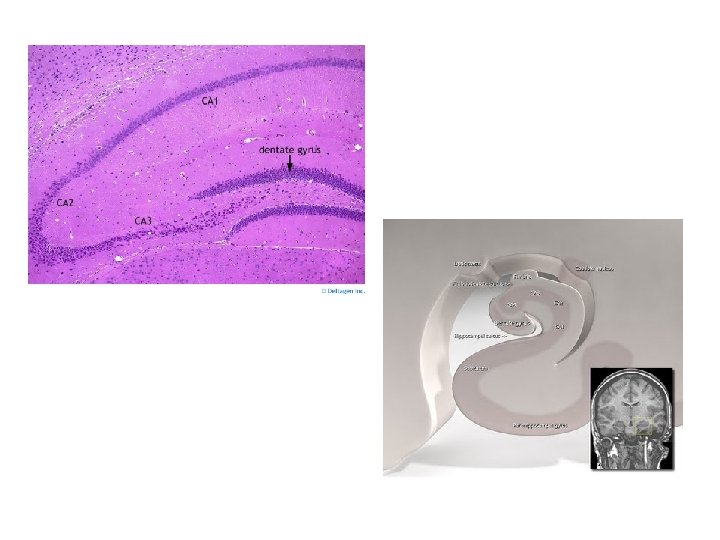

Hippocampal formation • afferent fibres – major input in hippocampus from parahippocampal gyrus via ‘perforant path’: terminates in molecular layer of dentate gyrus – granule cells in dentate gyrus→ molecular layer of CA 3 of hippocampus → CA 1 of hippocampus → input to subiculum – subiculum receives input from amygdala

Hippocampal formation • efferent fibres – outflow from subiculum and hippocampus towards fornix – from subiculum → postcommissural → mammillary bodies – from hippocampus → precommissural → septal nuclei, frontal cortex, hypothalamus, nucleus accumbens

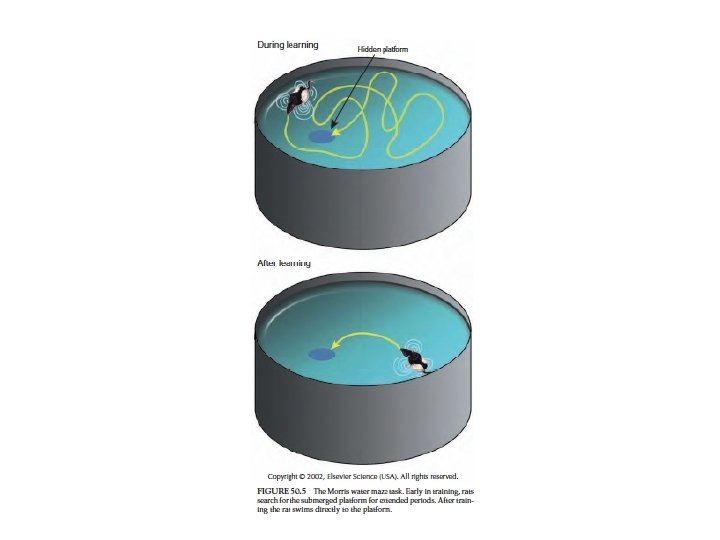
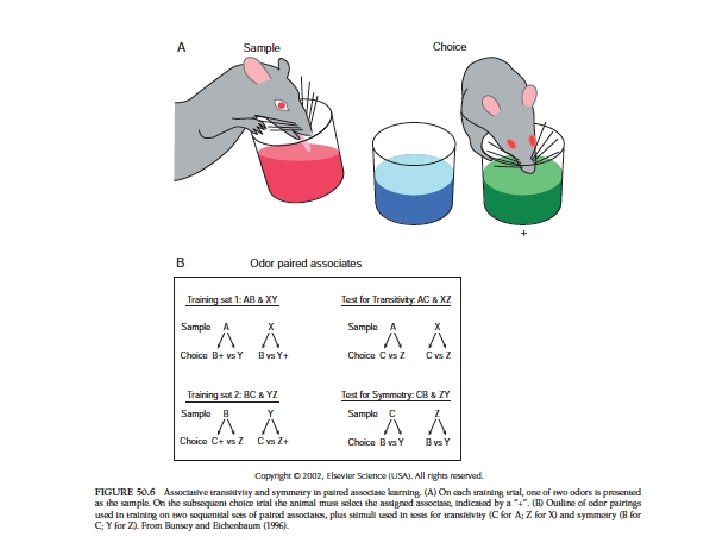
Hippocampal function • Emotion • patients with hippocampal lesions: – anterograde amnesia – able to perform tasks for sec or min – when distracted they don’t remember what they were doing • learning and memory • consolidation of long-term memories from immediate and short-term memories • spatial memory

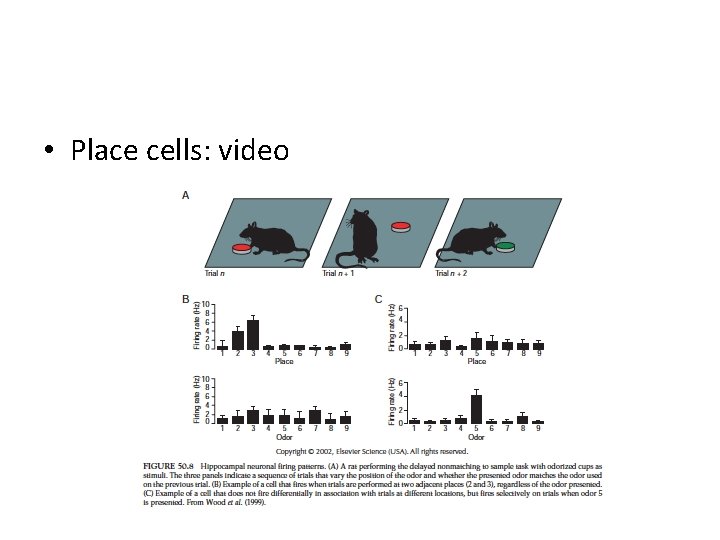
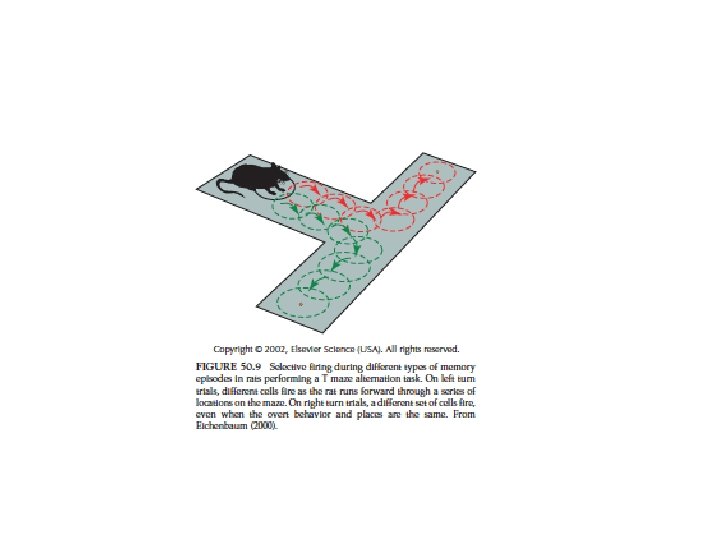
• Place cells: video
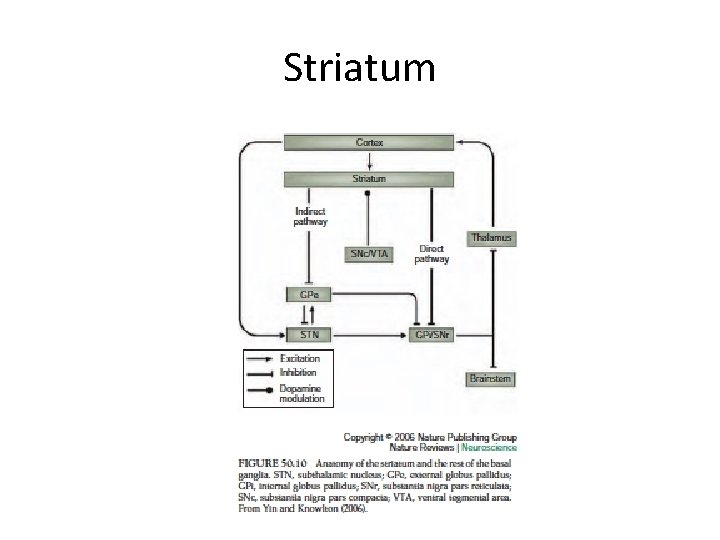
Striatum
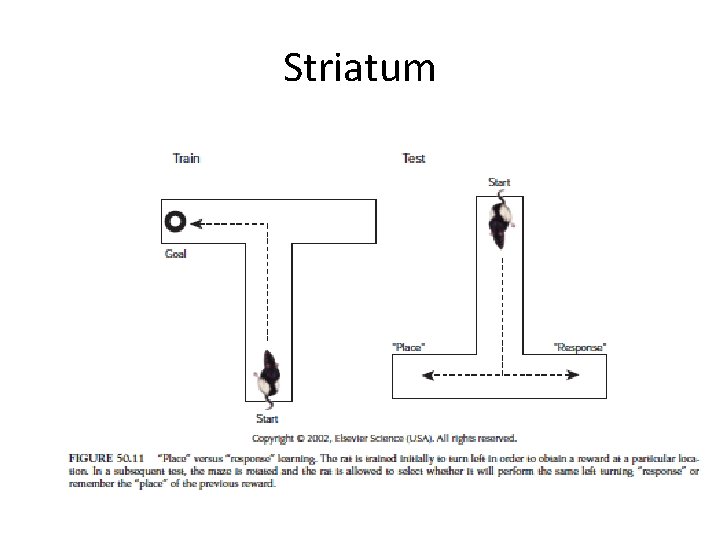
Striatum

Striatum

Cerebellar cortex
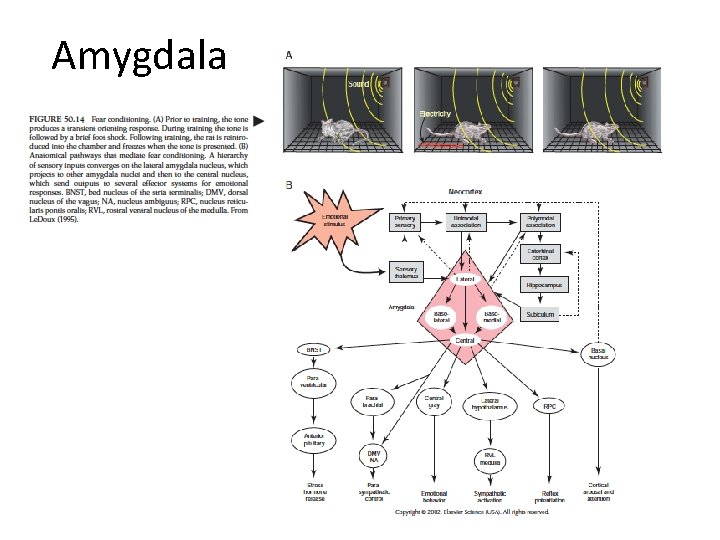
Amygdala
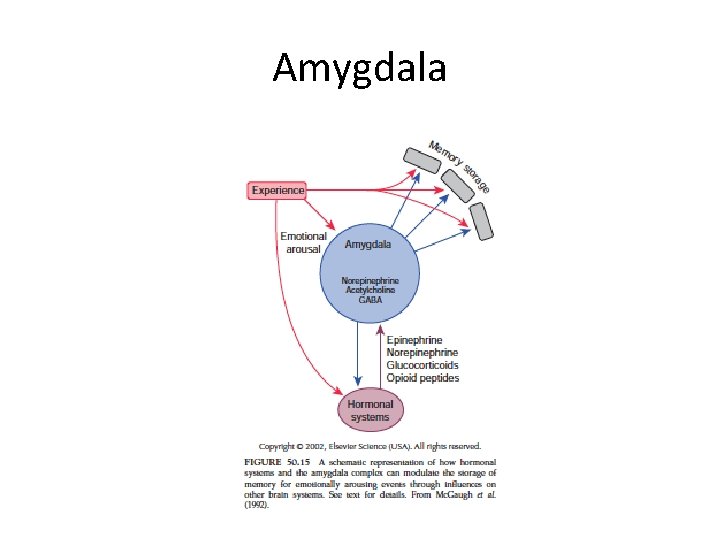
Amygdala

Amygdala function • Fear and negative emotional reactions • Appetitive, emotional reactions – Association of tone with food • Taste (rewarding) association affected by lesion of basolateral nuclei • Visual appearance (non-rewarding) association not affected • Context conditioning (place-preference) – Place cues – Hippocampus (binding a variety of sensory information about place)

Amygdala function • Unconscious emotional state – Connection with hypothalamus and ANS • Conscious feeling – Connection with cingulate gyrus and prefrontal cortex • Arousal – Direct projection to various nuclei – Indirect projections to nucleus basalis • β-adrenergic blokker (propanolol) impaired memomry for emotional but not neutral story
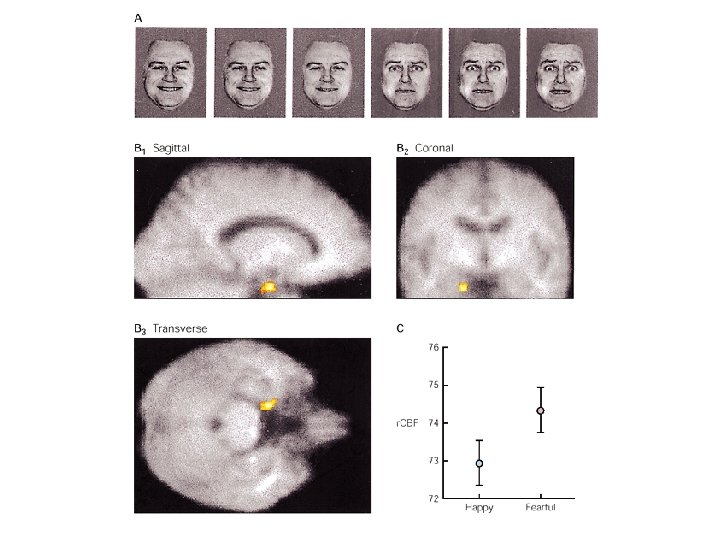

Amygdala dysfunction • Kluver-Bucy syndrome • behavioural changes due to bilateral temporal lobe lesions (abolishment of amygdala and the hippocampal formation, as well as the nonlimbic temporal cortex) • first: visual agnosia, sometimes tactile and auditory agnosia • second: hyperorality: tendency to examine objects by mouth

Amygdala dysfunction • third: hypermetamorphosis: compulsion to intensively explore the immediate environment and overreact to visual stimuli • fourth: placidity: no more fear or anger • fifth: hyperphagia: eat in excessive amounts even without hunger and objects that are not food • sixth: hypersexuality: augmentation in sexual behaviour: suggestive behaviour, talk, attempts at sexual contact • amnesia, dementia, aphasia

Amygdala dysfunction • Urbach-Wiethe disease – Calcium deposition in amygdala – Lesion early in life : fail to learn the cues that normal persons use to discern fear in facial expression and to discriminate fine differences in other facial expressions.

Cerebral cortex • Perceptual learning – tone discrimination – repetition priming • Both not-affected in HM

Cortical plasticity during development • Brain development Sensory information = crucial ‘Unused‘ synaptic connections -> disappear(= ‘pruning’) ‘Used’ synaptic connections -> strenghtened
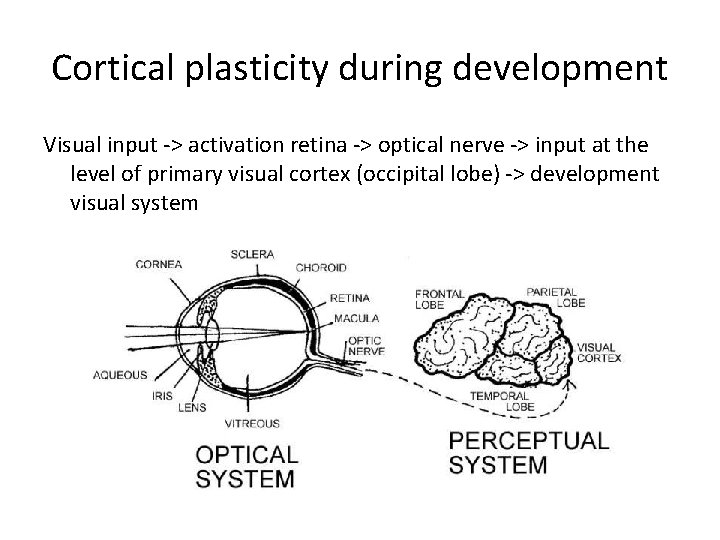
Cortical plasticity during development Visual input -> activation retina -> optical nerve -> input at the level of primary visual cortex (occipital lobe) -> development visual system

Cortical plasticity during development # Blind at birth (or <2 yr) visual cortex unsufficiently developed, optical system intact -> not reparable # Blind at later age (>2 jr) 1. eye defect (cataract, diabetic retinopathy…): visual cortex sufficiently developed-> visual prothesis (‘bionic eye’ – see further) 2. occipital lobe damage: damage of visual cortex, eye intact-> repair? # Developmental disorder
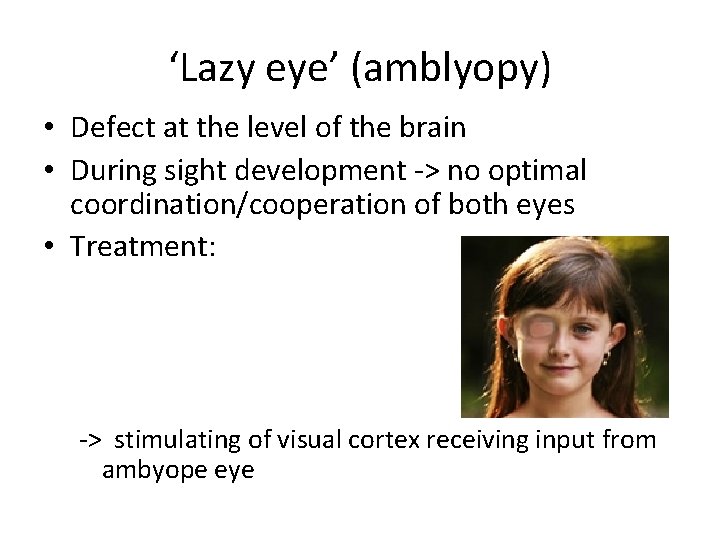
‘Lazy eye’ (amblyopy) • Defect at the level of the brain • During sight development -> no optimal coordination/cooperation of both eyes • Treatment: -> stimulating of visual cortex receiving input from ambyope eye
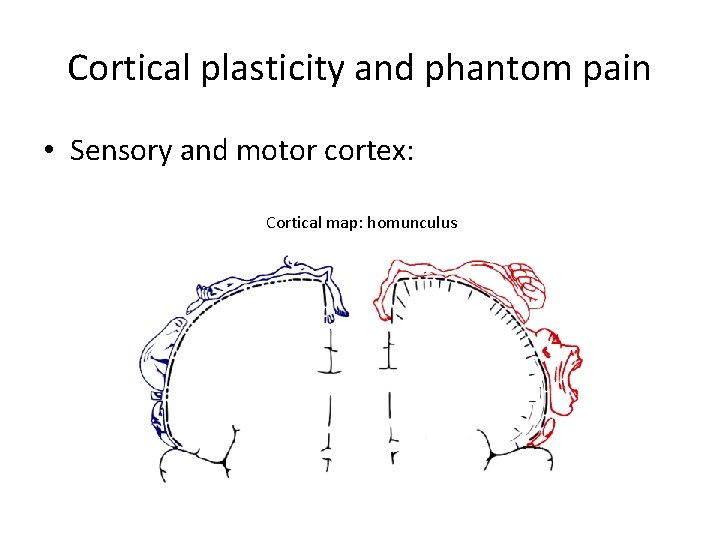
Cortical plasticity and phantom pain • Sensory and motor cortex: Cortical map: homunculus

Phantom pain = after amputation; sensation (of movement) in amputated extremity; sometimes pain Causes: – in the stump • Defective blood supply • Stimulation of pain (Aδ) nerve fibers (neuroma) – in the brain • Reorganization of somatosensory cortex

Phantom pain • Ramachandran – mirror box • Mirror Box and Phantom Limb Pain #1. mp 4

Neuroplasticity and synesthesia • Synesthesia: = by stimulating 1 sensory/cognitive pathway, a second sensory/cognitive pathway is activated automatically beyond our will E. g. Color-grapheme synesthesia: ‘seeing’ color with numbers
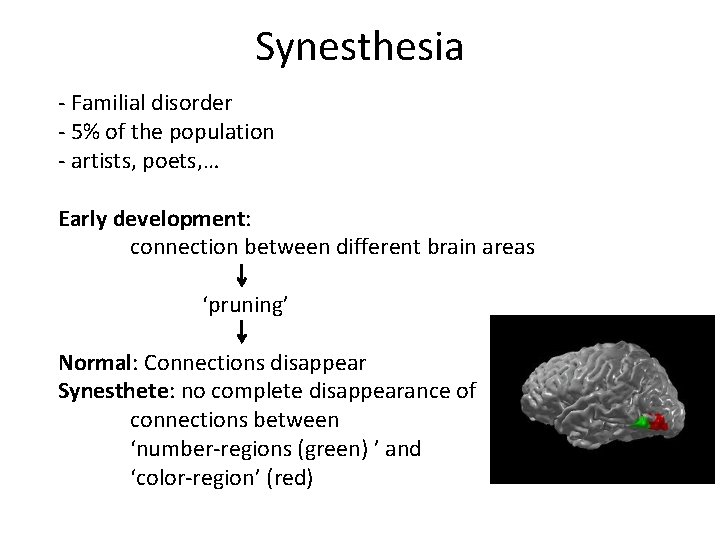
Synesthesia - Familial disorder - 5% of the population - artists, poets, … Early development: connection between different brain areas ‘pruning’ Normal: Connections disappear Synesthete: no complete disappearance of connections between ‘number-regions (green) ’ and ‘color-region’ (red)

Neuro-prostheses • Device that replaces sensory, motor or cognitive function that is damaged by disease or injury • Links machine with nerve system (via interface) • Prosthesis types: a) b) c) d) e) Visual Auditory Motor Sensory Cognitive
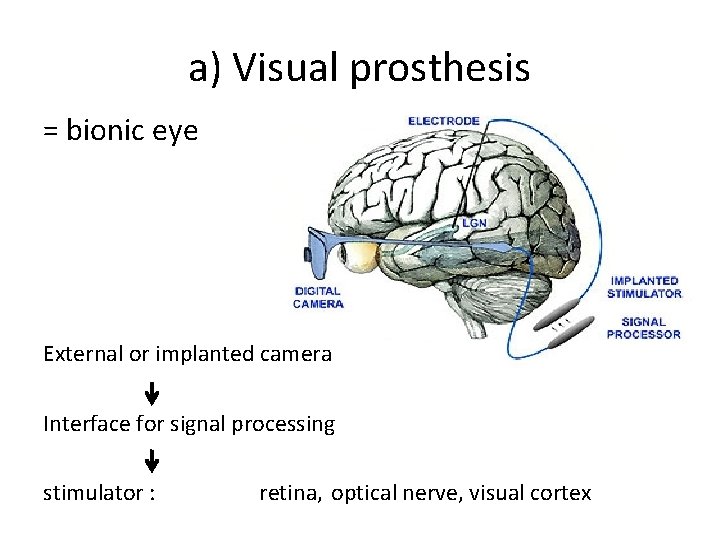
a) Visual prosthesis = bionic eye External or implanted camera Interface for signal processing stimulator : retina, optical nerve, visual cortex

a) Visual prosthesis
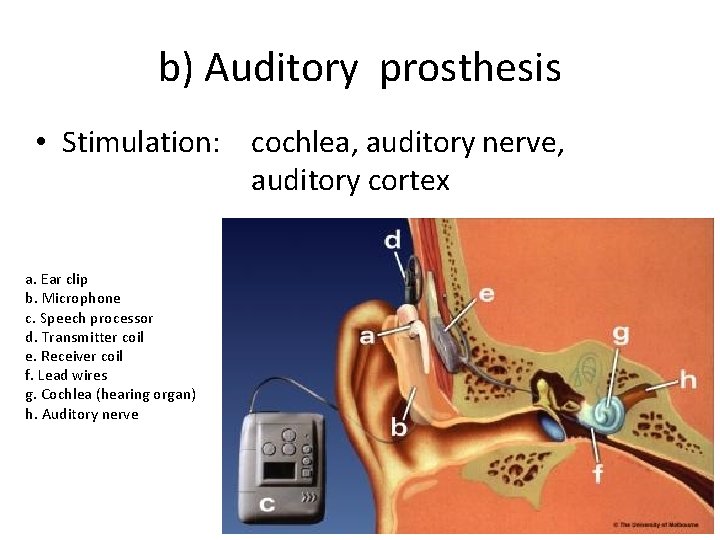
b) Auditory prosthesis • Stimulation: cochlea, auditory nerve, auditory cortex a. Ear clip b. Microphone c. Speech processor d. Transmitter coil e. Receiver coil f. Lead wires g. Cochlea (hearing organ) h. Auditory nerve

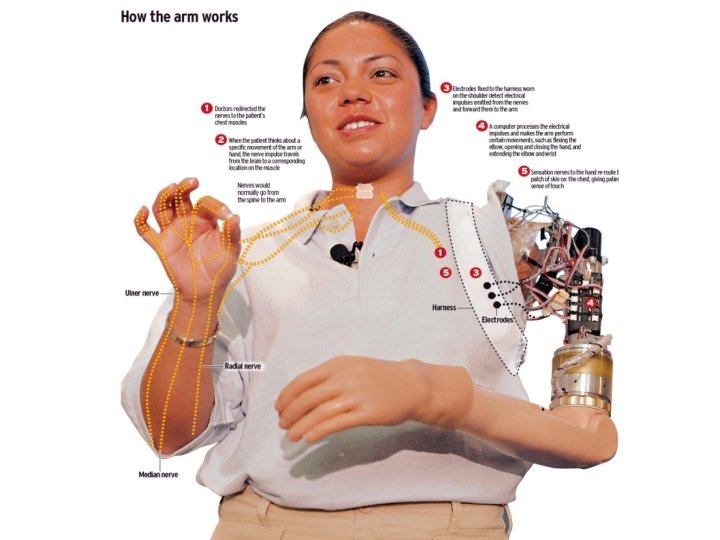
c) Motor neuroprosthesis = depends on which part of the motor system is defective • Bvb. – Paralyzed limb: stimulation of intraspinal nerves stimulation of muscles – Amputated limb : bionic limb
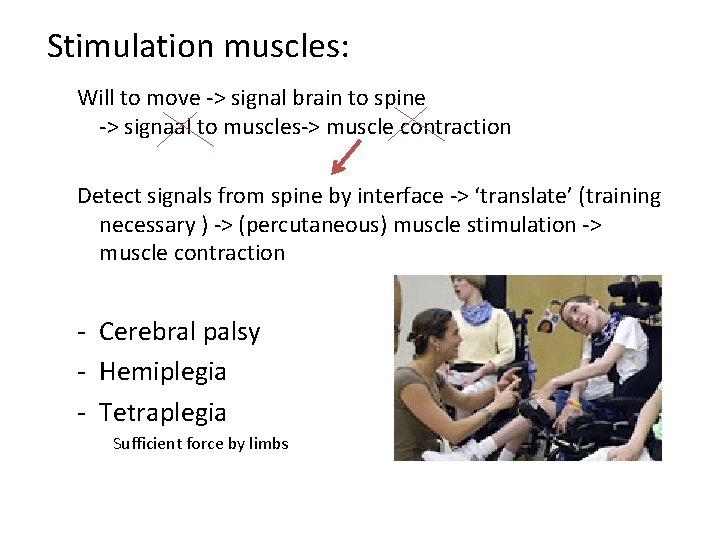
Stimulation muscles: Will to move -> signal brain to spine -> signaal to muscles-> muscle contraction Detect signals from spine by interface -> ‘translate’ (training necessary ) -> (percutaneous) muscle stimulation -> muscle contraction - Cerebral palsy - Hemiplegia - Tetraplegia Sufficient force by limbs
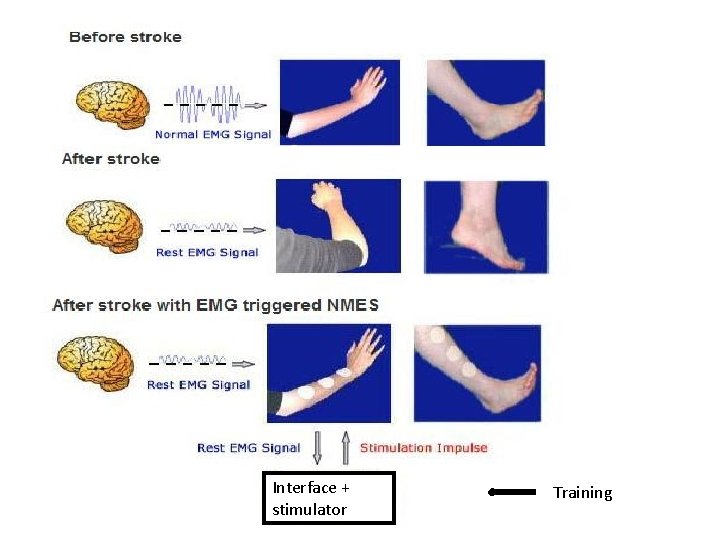
Interface + stimulator Training
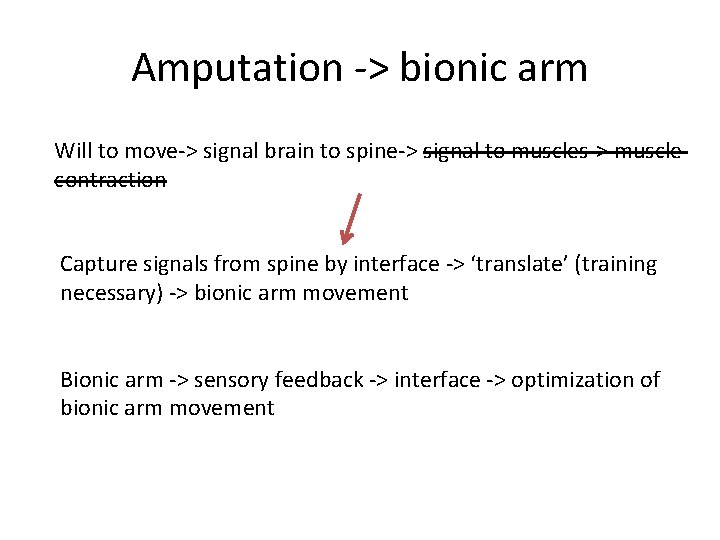
Amputation -> bionic arm Will to move-> signal brain to spine-> signal to muscles-> muscle contraction Capture signals from spine by interface -> ‘translate’ (training necessary) -> bionic arm movement Bionic arm -> sensory feedback -> interface -> optimization of bionic arm movement
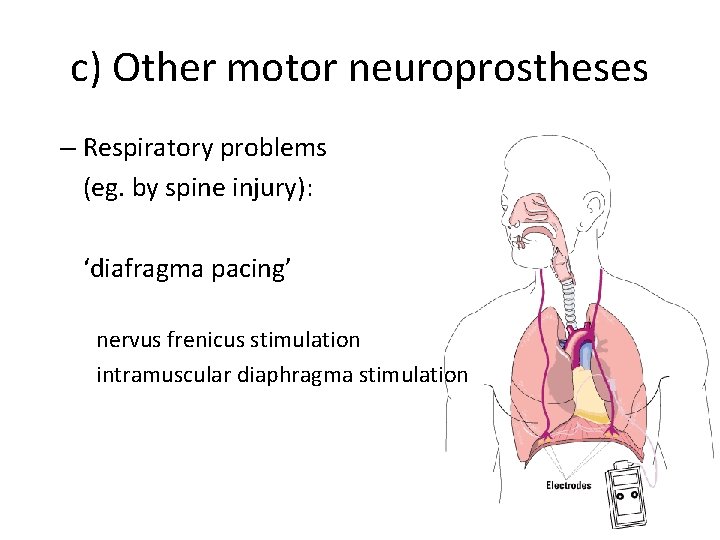
c) Other motor neuroprostheses – Respiratory problems (eg. by spine injury): ‘diafragma pacing’ nervus frenicus stimulation intramuscular diaphragma stimulation

c) Other motor neuroprostheses - Incontinence & micturition problems: stimulation of bladder muscles
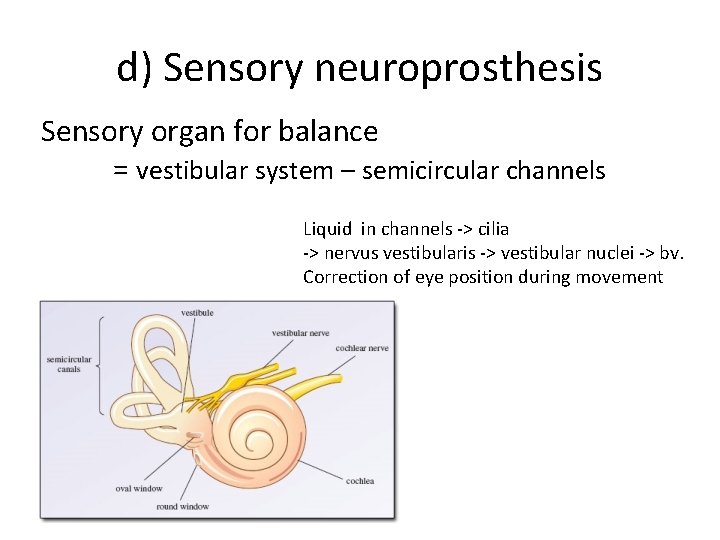
d) Sensory neuroprosthesis Sensory organ for balance = vestibular system – semicircular channels Liquid in channels -> cilia -> nervus vestibularis -> vestibular nuclei -> bv. Correction of eye position during movement
d) Sensory neuroprosthesis • Injury (unreparable) in vestibular system -> sensation of ‘continuous falling’ ‘Brain. Port®’ (=sensoric substitution): accelerometry (head) connected to ‘grid’ of 144 stimulation electrodes on tongue e. g. Bend to front: stimulation in front of tongue Bend to back: stimulation at the back of the tongue etc…
Signal from stimulation-electrodes -> Sensory neurons of tongue-region in brain -> Interpretation of movement-> correction of body movement by vestibular system -> finally (after training): vestibular system is directly sensitive for sensory information from tongue (active interpretation no longer necessary)
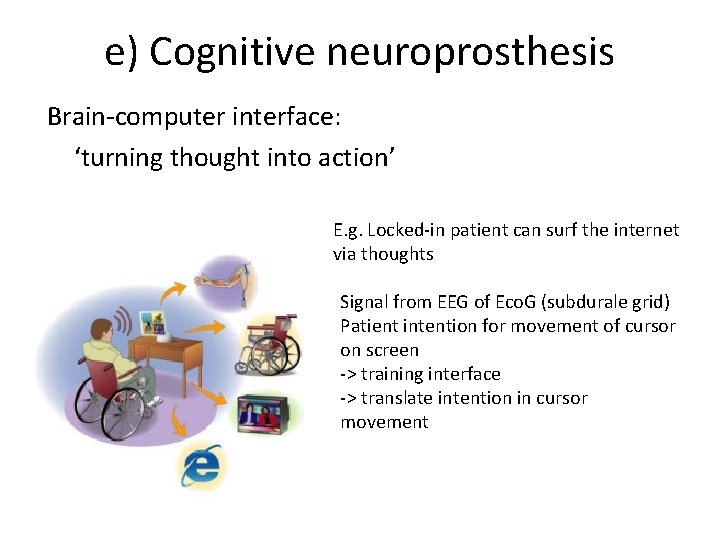
e) Cognitive neuroprosthesis Brain-computer interface: ‘turning thought into action’ E. g. Locked-in patient can surf the internet via thoughts Signal from EEG of Eco. G (subdurale grid) Patient intention for movement of cursor on screen -> training interface -> translate intention in cursor movement

e) Brain-computer interface
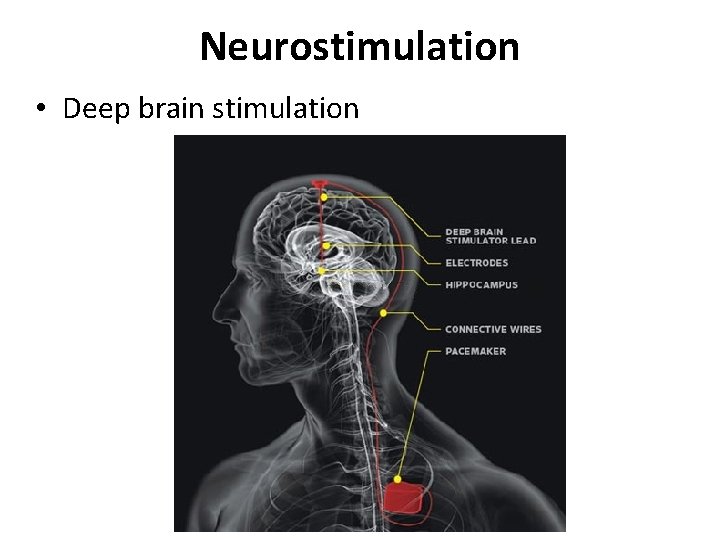
Neurostimulation • Deep brain stimulation

Deep brain stimulation 1973: chronic pain 1987: movement disorders (eg. Parkinson’s disease) 1992: epilepsy 1999: Gilles de la Tourette 1999: obsessive-compulsive disorder 2003: cluster headache 2005: addiction 2005: depression 2007: obesitas 2007: hypertension 2008: memory-improvement-> ethical? ?

DBS for Parkinson’s disease
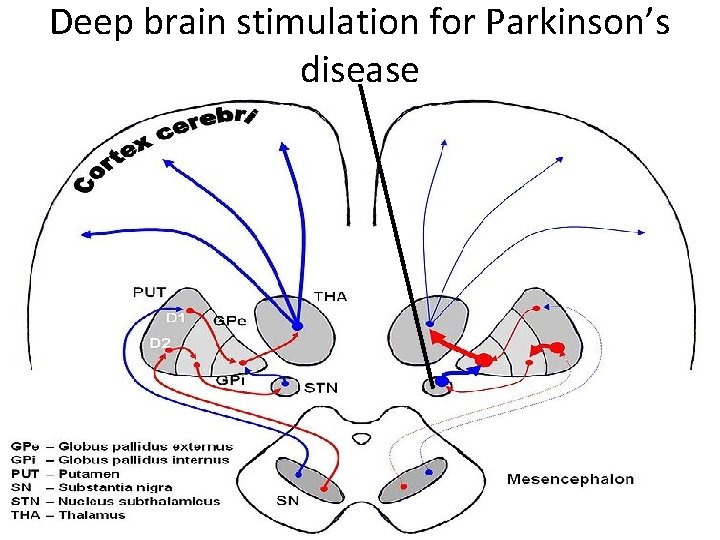
Deep brain stimulation for Parkinson’s disease
- Slides: 106