Neuropeptide Y and corticotropinreleasing factor bidirectionally modulate inhibitory

Neuropeptide Y and corticotropinreleasing factor bi-directionally modulate inhibitory synaptic transmission in the bed nucleus of the stria terminalis Thomas L. Kash, Danny G. Winder
Introduction l. Neuropeptides (NPY) = potent neuromodulators in the CNS l. Involved in reward pathway ¡mediated via G-protein coupled receptors ¡released in a frequency dependent fashion ¡longer half-life of activity after release

Introduction l. Corticotropin Releasing Factor (CRF) involved in stress response ¡Mediated through the hypothalamus and the amygdala ¡Both pathways converge at the BNST

Introduction l. Life = Stress l. Repeated or severe stressors can produce behaviors such as posttraumatic stress disorder and generalized anxiety disorder l. BNST provides a substrate for interaction of CRF and NPY in regulating stress and anxiety

Introduction l. NPY Anxiety ↓ Reward pathway l. CRF Anxiety ↑ Stress response l. BNST acts as a scale to create a balance of CRF and NPY
Neves S, Ram P, Iyengar R. G protein pathways. Science 296, 1636 -1639 (2002)

Introduction l. BNST expresses both NPY/YRs and CRF/CRFRs l. Ce. A releases CRF and GABA to the BNST l. Both CRF and NPY modulate GABAergic transmissions

Introduction l. Study GABAergic influence on ventrolateral region of the BNST lvl. BNST projects to the VTA (reward) and the PVN (stress)

Introduction l IPSC= inhibitory post-synaptic current m = miniature e = evoked

Methods l Male C 57 B 1/6 J mice l Decapitated mice and placed brain in icecold sucrose a. CSF l Slices 300 um thick l Rostral slices contained anterior BNST ¡Stored in heated, oxygenated container w/ a. CSF ¡Transferred to submerged recording chamber l. Heated, O 2 a. CSF for 1 h ā experiments

Methods l Slices in chamber and neurons of vl. BNST visualized w/ infrared video microscopy l Analyzed e. IPSC & EPSC ¡Electrodes filled w/ p. H 7. 2 ¡Twisted nichrome wire ¡Placed in vl. BNST ¡Cells held @ -50 m. V & GABAAR-mediated IPSCs evoked @ 0. 2 Hz by fiber stim w/ bipolar electrodes

Methods l GABAA-IPSCs (& EPSC) isolated ¡ 3 m. M kynurenic acid (& 25 u. M picrotoxin) = block AMPA & NMDA receptor-dependent postsynaptic currents ¡ 1 u. M CGP 55845 = block GABABR l Signals acquired via Multiclamp 700 B amplifier l Input and series resistance continuously monitored

Methods l e. IPSC → measured peak amplitude of synaptic response normalized to baseline l Baseline period = 2 min period immediately preceding drug ¡Value is 2 min avg 15 min p neuropeptide B 0 3 * peptide 5 10 15 20 22

Methods l m. IPSC analysis ¡GABAAR-mediated IPSCs isolated → added 0. 5 u. M TTX ¡recorded in 120 s episodes ¡Ca 2+ influx on NPY → 100 u. M Cd 2+ + a. CSF ¡Amplitude and frequency determined from 120 s recording w/ cells held @ -70 m. V l. Multi-clamp

Methods l All drugs applied via bath l All peptides used were dissolved in d. H 2 O to 0. 1 m. M conc ¡Some stored @ -20°C

Results l. NPY and CRF influence on inhibitory synaptic transmission in vl. BNST ¡Whole-cell voltage clamp l. Local stimulation produced e. IPSC from GABAARs ¡SR 95531= GABAAR antagonist blocked response
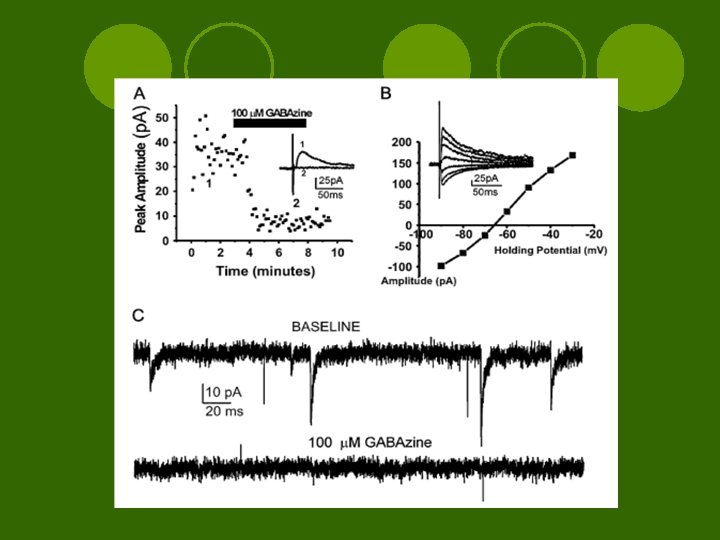

Results l. TTX elicited spontaneous m. IPSCs l. GABAzine = GABAAR antagonist ¡m. IPSCs blocked ¡GABAAR mediated

Results l. NPY depresses GABA through Y 2 R l. Baseline recordings revealed decreased peak amplitude of e. IPSC ¡Observed in all cells l. NPY-induced depression = concentration dependent
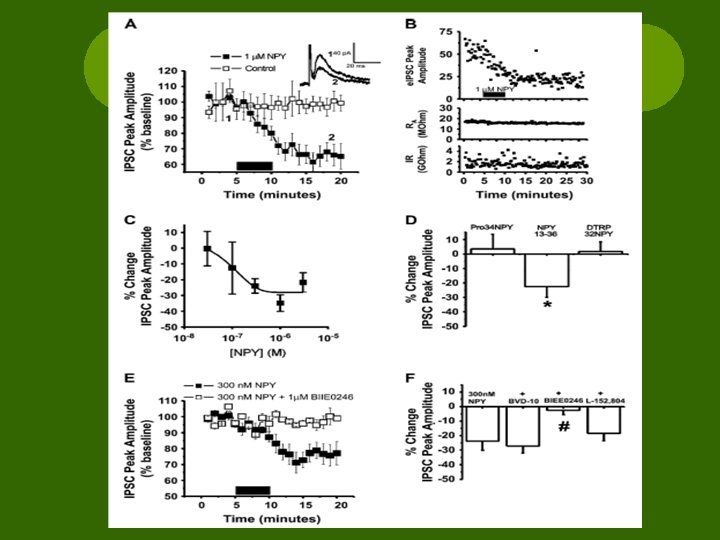

Results l. NPY 13 -36 = Y 2 R agonist ¡ ↓ peak amplitude l[Pro 34]-NPY = Y 1 R agonist & [D-Trp 32]-NPY = Y 5 R agonist ¡No change
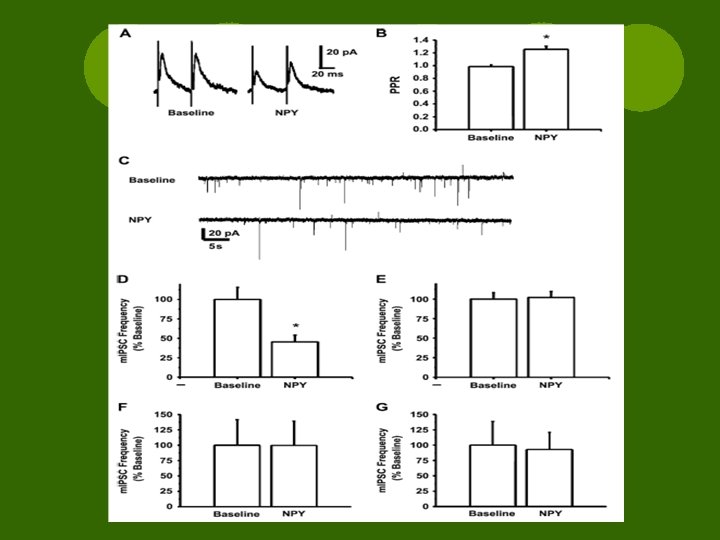

Results l. Antagonist testing was preceded by an exposure to NPY (10 -15 min) l. Agonist and antagonist co-applied (5 min) l. NONE had significant effects on e. IPSC without agonist

Results l. Non-peptide Y 2 R antagonist blocked NPY actions l. Peptide Y 1 R antagonist had no effect l. Non-peptide Y 5 R antagonist had no effect l. Y 2 Rs activated NPY-induced e. IPSC depression

Results l. Paired Pulse Ratio experiments ¡Pair of e. IPSCs w/ 50 ms between ¡Ratio of amplitudes determined l. NPY ↑ PPR of e. IPSCs ¡Suggest ↓ release of GABA l. NPY ↓ frequency but not amplitude

Results l. Presynaptic inhibition of NT release ¡Modulate Ca 2+ entry ¡Regulate release machinery l. At CNS synapse basal m. IPSC freq ↓ by using Cd 2+ to block Vg Ca 2+ channels l. Cd 2+ + NPY = restores m. IPSCs l. NPY inhibits GABA via Y 2 R regulation of Ca 2+ influx

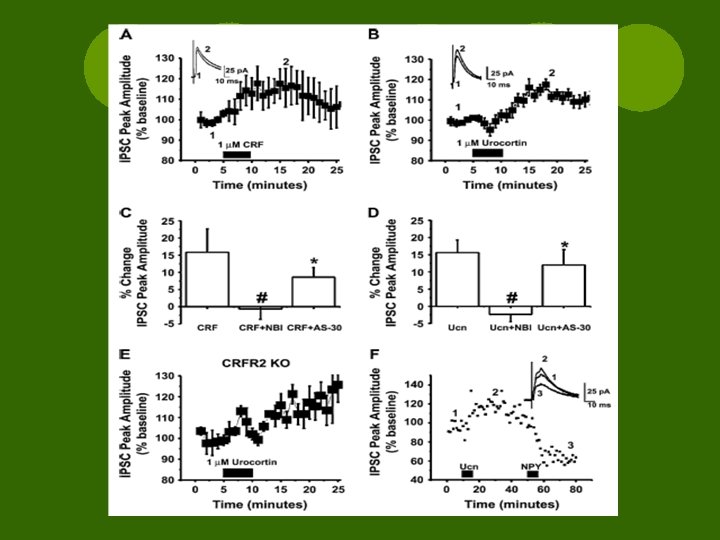
Results l 5 min bath of 1 um CRF sig ↑ peak amp of e. IPSC l 1 um Urocortin (CRFR agonist) → similar results l. CRF results were concentration dependent [100 n. M vs 10 n. M] l. CRF antagonist had no sig effects on e. IPSC in absence of agonist

Results l. Non-peptide CRFR 1 antagonist (NBI 27914) blocked both CRF and Ucn I actions l. Peptide CRFR 2 antagonist (anti. Sauvagine-30) had no sig effect l. Ucn I enhanced e. IPSCs in CRFR 2 knockout mice l. So, CRF/Ucn I induced enhancement of e. IPSC is d/t CRFR 1 activation
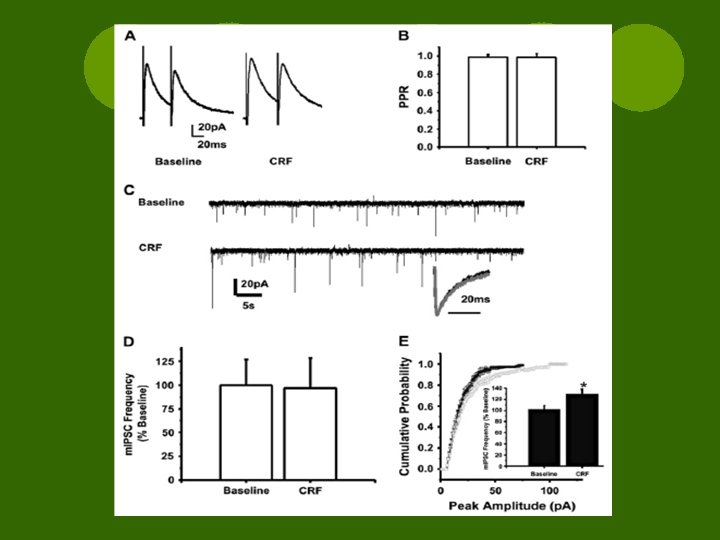

Results l. CRF ¡did not alter PPR or m. IPSC kinetics ¡no effect on freq of m. IPSCs ¡Mean amplitude ↑↑ ¡Shifted cum. amplitude curve to the right l. CRF enhanced GABAergic postsynaptic transmission l. Change in IPSCs d/t non-specific enhancement via synaptic excitability

Discussion l NPY suppresses GABAergic transmission in vl. BNST via Y 2 R: 1. NPY effect mimicked by NPY 13 -36, not by [Pro 34]-NPY (Y 1 agonist) or [DTrp 32]-NPY (Y 5 agonist) 2. NPY antagonized by Y 2 R antagonist (BIEE 0246), not by BVD-10 (Y 1 R antagonist) or L-152804 (Y 5 R antagonist)

Discussion l. Data consistent w/ NPY actions in thalamus & PVN and Y 2 R expression in BNST l. Y 2 R ↓ GABA release: 1. NPY ↑ PPR of e. IPSCs → ↓ release probability 2. NPY ↓ m. IPSC frequency not amplitude 3. Cd+2 effect → NPY via Y 2 R inhibit GABA via presynaptic Ca+2 influx

Discussion l. NPY via Y 2 R → heterocepter on GABAergic terminals l. Which YR is activated determines the behavioral outcome ¡Y 1 R and maybe Y 5 R → NPY anxiolytic response ¡Y 2 R → anxiogenic response l. But, Y 2 R activation in LC → NPY anxiolytic effects

Discussion l. Suppose region-specific activation of YR subtypes will evoke distinct behavioral phenotypes, OR… l. Autoreceptor-like functions are anxiolytic

Discussion l. Based on evidence that inhibitory projections from the vl. BNST contact PVN neurons: ↓ GABAergic input to vl. BNST → ↑ GABAergic output to PVN → ↓ stress response

Discussion l. In the vl. BNST, CRF and Ucn I enhance GABAergic transmission via CRFR 1: 1. CRF & Ucn I antagonized by NBI 27914 (CRFR 1), not AS 30 (CRFR 2) 2. Ucn I effects observed in mice lacking CRFR 2 3. CRFR 1 enhance postsynaptic GABAA-R response

Discussion l. CRF ↑ m. IPSC amplitude not frequency or PPR l. Glutamatergic transmission in vl. BNST not affected by Ucn I l. Presynaptic CRFR 1 mediates GABAergic transmission in Ce. A l. Altering m. IPSC frequency modulates GABA release → now amplitude as well

Discussion l. CRFR 1 mediates CRF anxiogenic effects in the BNST l. CRF enhancing GABAergic transmission in BNST could potentially reactivate the PVN → stress response
- Slides: 39