Neurobiology and aetiology Parkinsons disease 1 Neurobiology 2

Neurobiology and aetiology Parkinson’s disease 1

Neurobiology 2
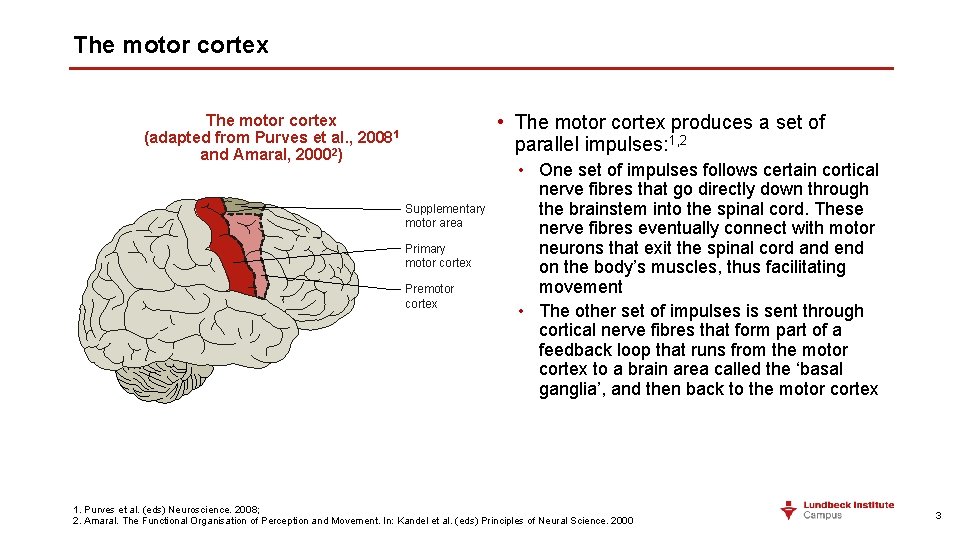
The motor cortex • The motor cortex produces a set of parallel impulses: 1, 2 The motor cortex (adapted from Purves et al. , 20081 and Amaral, 20002) Supplementary motor area Primary motor cortex Premotor cortex • One set of impulses follows certain cortical nerve fibres that go directly down through the brainstem into the spinal cord. These nerve fibres eventually connect with motor neurons that exit the spinal cord and end on the body’s muscles, thus facilitating movement • The other set of impulses is sent through cortical nerve fibres that form part of a feedback loop that runs from the motor cortex to a brain area called the ‘basal ganglia’, and then back to the motor cortex 1. Purves et al. (eds) Neuroscience. 2008; 2. Amaral. The Functional Organisation of Perception and Movement. In: Kandel et al. (eds) Principles of Neural Science. 2000 3
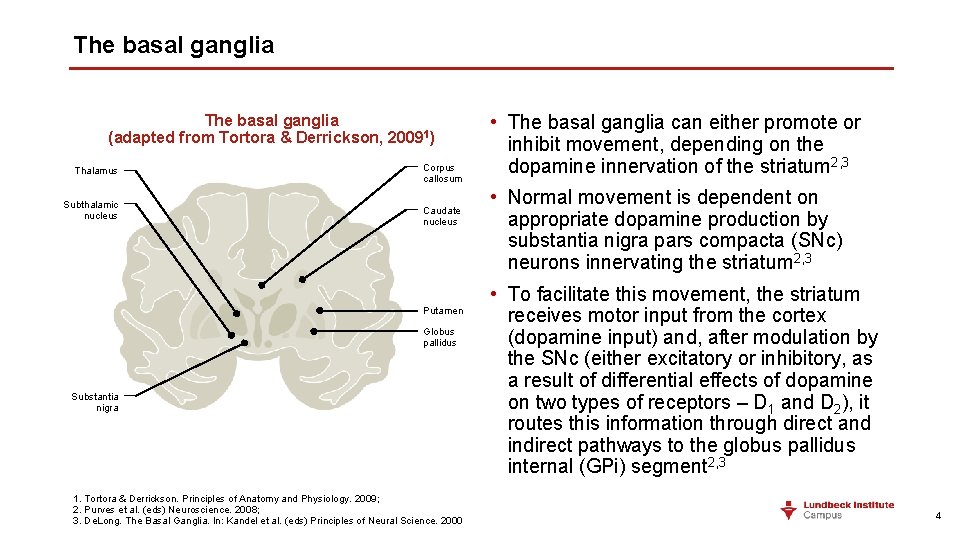
The basal ganglia (adapted from Tortora & Derrickson, 20091) Thalamus Subthalamic nucleus Corpus callosum Caudate nucleus Putamen Globus pallidus Substantia nigra 1. Tortora & Derrickson. Principles of Anatomy and Physiology. 2009; 2. Purves et al. (eds) Neuroscience. 2008; 3. De. Long. The Basal Ganglia. In: Kandel et al. (eds) Principles of Neural Science. 2000 • The basal ganglia can either promote or inhibit movement, depending on the dopamine innervation of the striatum 2, 3 • Normal movement is dependent on appropriate dopamine production by substantia nigra pars compacta (SNc) neurons innervating the striatum 2, 3 • To facilitate this movement, the striatum receives motor input from the cortex (dopamine input) and, after modulation by the SNc (either excitatory or inhibitory, as a result of differential effects of dopamine on two types of receptors – D 1 and D 2), it routes this information through direct and indirect pathways to the globus pallidus internal (GPi) segment 2, 3 4
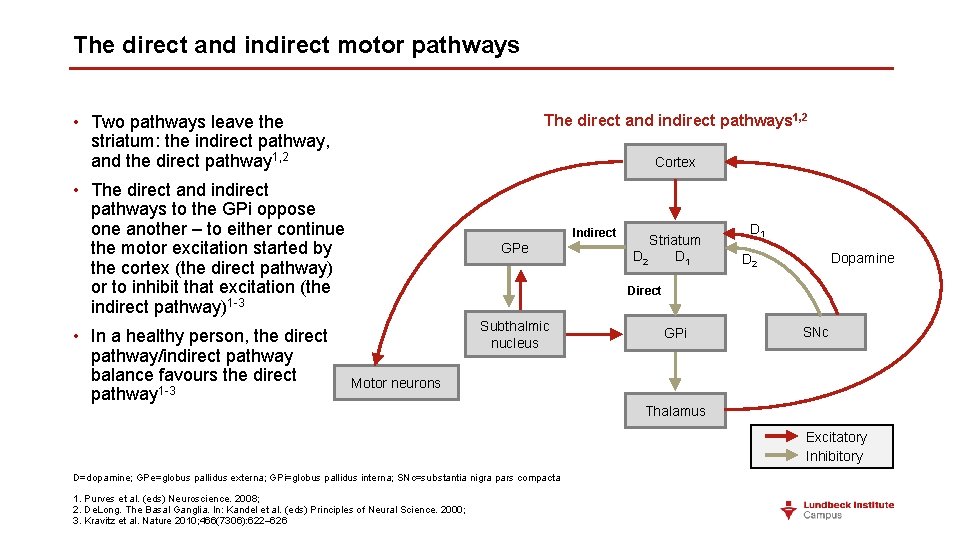
The direct and indirect motor pathways • Two pathways leave the striatum: the indirect pathway, and the direct pathway 1, 2 The direct and indirect pathways 1, 2 Cortex • The direct and indirect pathways to the GPi oppose one another – to either continue the motor excitation started by the cortex (the direct pathway) or to inhibit that excitation (the indirect pathway)1 -3 • In a healthy person, the direct pathway/indirect pathway balance favours the direct pathway 1 -3 Indirect GPe Striatum D 2 D 1 Dopamine D 2 Direct Subthalmic nucleus GPi SNc Motor neurons Thalamus Excitatory Inhibitory D=dopamine; GPe=globus pallidus externa; GPi=globus pallidus interna; SNc=substantia nigra pars compacta 1. Purves et al. (eds) Neuroscience. 2008; 2. De. Long. The Basal Ganglia. In: Kandel et al. (eds) Principles of Neural Science. 2000; 3. Kravitz et al. Nature 2010; 466(7306): 622– 626

Neurotransmitters and the presynaptic and postsynaptic neurons The synapse (adapted from Purves et al. , 20081 and Kandel & Siegelbaum, 20002) • Neurons are separated from each other by a gap, called a synapse 1, 2 Electrical impulse • Messages reaching the presynaptic neuron are transmitted to the postsynaptic neuron via chemical messengers, called neurotransmitters 1, 2 • Some neurotransmitters cause the postsynaptic neuron to become activated, and some inhibit it – the good functioning of the brain requires both activation and inhibition when required, in a dynamic balance 1, 2 Presynaptic terminal of (presynaptic) neuron Synaptic vesicle (‘packet‘ of neurotransmitter) Vesicle fuses with membrane to release neurotransmitter into synapse Synapse Postsynaptic receptor Postsynaptic neuron Neurotransmitter binds to postsynaptic receptor Impulse triggered in postsynaptic neuron 1. Purves et al. (eds) Neuroscience. 2008; 2. Kandel & Siegelbaum. Overview of Synaptic Transmission. In: Kandel et al. (eds) Principles of Neural Science. 2000 6
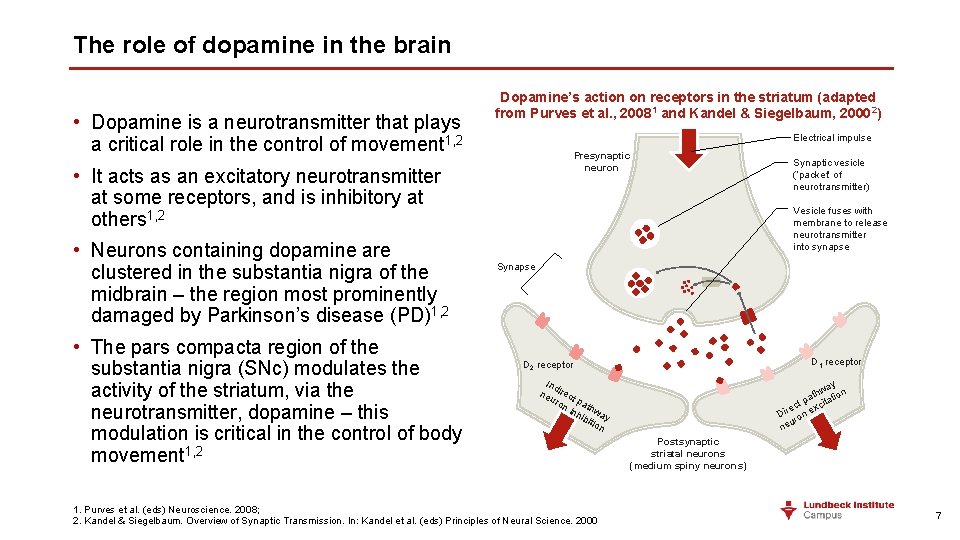
The role of dopamine in the brain • Dopamine is a neurotransmitter that plays a critical role in the control of movement 1, 2 Dopamine’s action on receptors in the striatum (adapted from Purves et al. , 20081 and Kandel & Siegelbaum, 20002) Electrical impulse Presynaptic neuron • It acts as an excitatory neurotransmitter at some receptors, and is inhibitory at others 1, 2 • Neurons containing dopamine are clustered in the substantia nigra of the midbrain – the region most prominently damaged by Parkinson’s disease (PD)1, 2 • The pars compacta region of the substantia nigra (SNc) modulates the activity of the striatum, via the neurotransmitter, dopamine – this modulation is critical in the control of body movement 1, 2 Synaptic vesicle (‘packet‘ of neurotransmitter) Vesicle fuses with membrane to release neurotransmitter into synapse Synapse D 1 receptor D 2 receptor Ind ne irect uro pa ni nh thwa y ibi tio n 1. Purves et al. (eds) Neuroscience. 2008; 2. Kandel & Siegelbaum. Overview of Synaptic Transmission. In: Kandel et al. (eds) Principles of Neural Science. 2000 y wa on h t a ti t p xcita c e Dir ron e u ne Postsynaptic striatal neurons (medium spiny neurons) 7

The pathogenesis of Parkinson’s disease 8
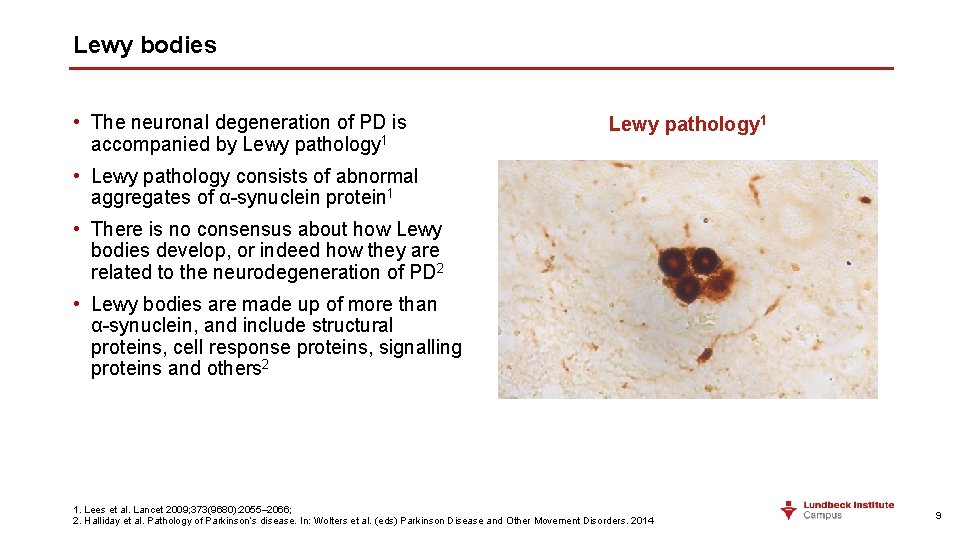
Lewy bodies • The neuronal degeneration of PD is accompanied by Lewy pathology 1 • Lewy pathology consists of abnormal aggregates of α-synuclein protein 1 • There is no consensus about how Lewy bodies develop, or indeed how they are related to the neurodegeneration of PD 2 • Lewy bodies are made up of more than α-synuclein, and include structural proteins, cell response proteins, signalling proteins and others 2 1. Lees et al. Lancet 2009; 373(9680): 2055– 2066; 2. Halliday et al. Pathology of Parkinson’s disease. In: Wolters et al. (eds) Parkinson Disease and Other Movement Disorders. 2014 9
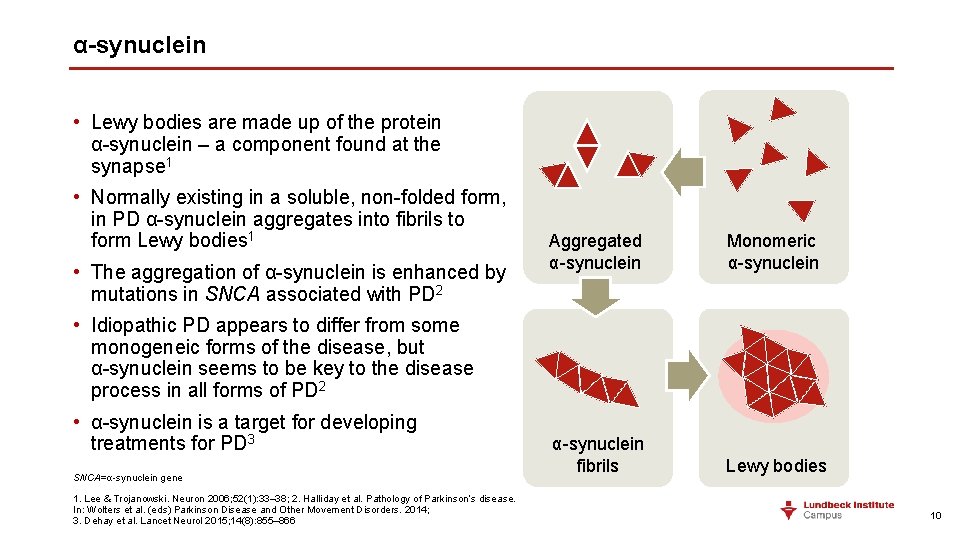
α-synuclein • Lewy bodies are made up of the protein α-synuclein – a component found at the synapse 1 • Normally existing in a soluble, non-folded form, in PD α-synuclein aggregates into fibrils to form Lewy bodies 1 • The aggregation of α-synuclein is enhanced by mutations in SNCA associated with PD 2 Aggregated α-synuclein Monomeric α-synuclein fibrils Lewy bodies • Idiopathic PD appears to differ from some monogeneic forms of the disease, but α-synuclein seems to be key to the disease process in all forms of PD 2 • α-synuclein is a target for developing treatments for PD 3 SNCA=α-synuclein gene 1. Lee & Trojanowski. Neuron 2006; 52(1): 33– 38; 2. Halliday et al. Pathology of Parkinson’s disease. In: Wolters et al. (eds) Parkinson Disease and Other Movement Disorders. 2014; 3. Dehay et al. Lancet Neurol 2015; 14(8): 855– 866 10
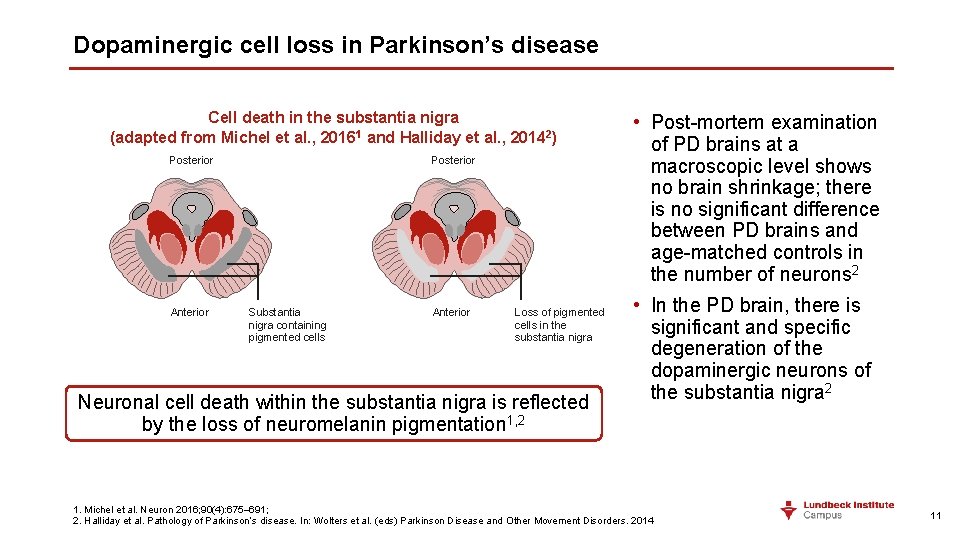
Dopaminergic cell loss in Parkinson’s disease Cell death in the substantia nigra (adapted from Michel et al. , 20161 and Halliday et al. , 20142) Posterior Anterior Posterior Substantia nigra containing pigmented cells Anterior Loss of pigmented cells in the substantia nigra Neuronal cell death within the substantia nigra is reflected by the loss of neuromelanin pigmentation 1, 2 • Post-mortem examination of PD brains at a macroscopic level shows no brain shrinkage; there is no significant difference between PD brains and age-matched controls in the number of neurons 2 • In the PD brain, there is significant and specific degeneration of the dopaminergic neurons of the substantia nigra 2 1. Michel et al. Neuron 2016; 90(4): 675– 691; 2. Halliday et al. Pathology of Parkinson’s disease. In: Wolters et al. (eds) Parkinson Disease and Other Movement Disorders. 2014 11
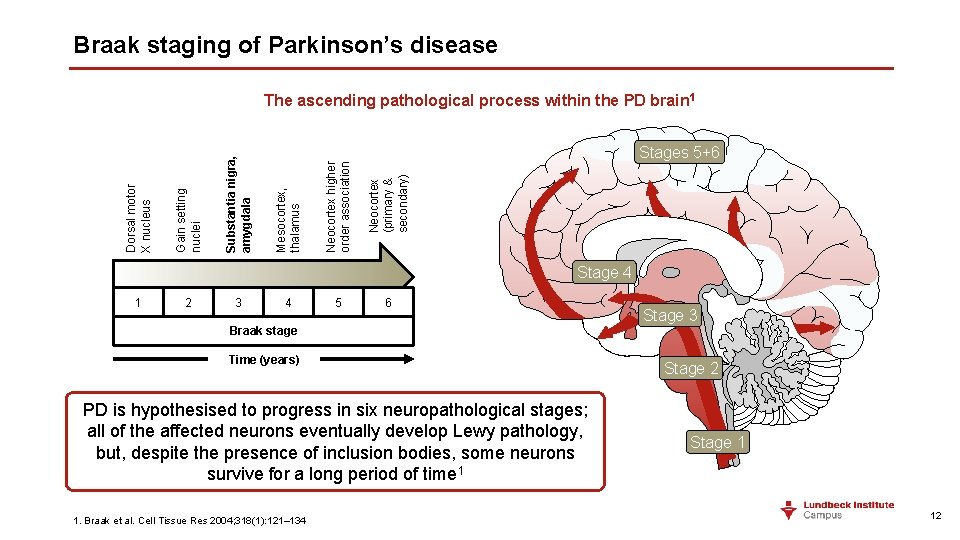
Braak staging of Parkinson’s disease Neocortex (primary & secondary) Neocortex higher order association Stages 5+6 Mesocortex, thalamus Substantia nigra, amygdala Gain setting nuclei Dorsal motor X nucleus The ascending pathological process within the PD brain 1 Stage 4 1 2 3 4 5 6 Stage 3 Braak stage Time (years) PD is hypothesised to progress in six neuropathological stages; all of the affected neurons eventually develop Lewy pathology, but, despite the presence of inclusion bodies, some neurons survive for a long period of time 1 1. Braak et al. Cell Tissue Res 2004; 318(1): 121– 134 Stage 2 Stage 1 12
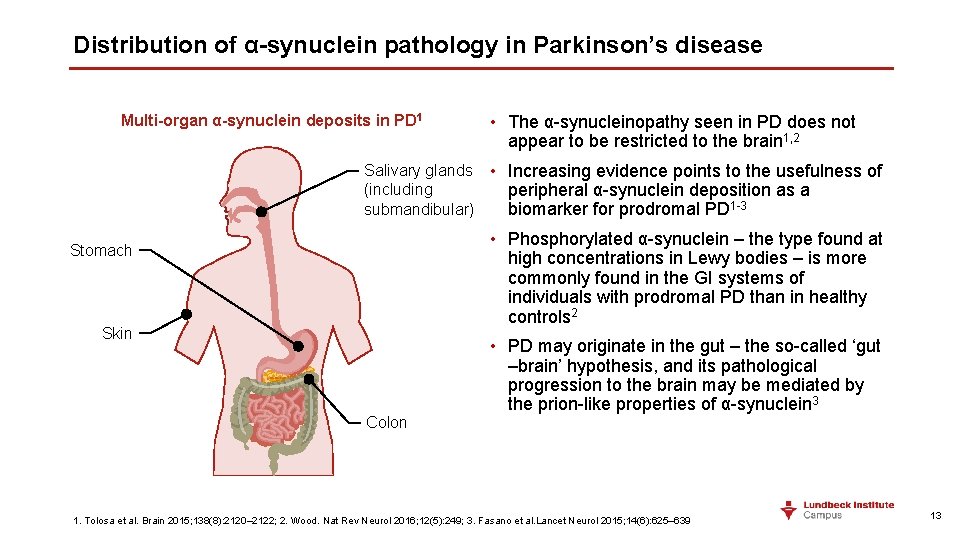
Distribution of α-synuclein pathology in Parkinson’s disease Multi-organ α-synuclein deposits in PD 1 • The α-synucleinopathy seen in PD does not appear to be restricted to the brain 1, 2 Salivary glands • Increasing evidence points to the usefulness of (including peripheral α-synuclein deposition as a submandibular) biomarker for prodromal PD 1 -3 • Phosphorylated α-synuclein – the type found at high concentrations in Lewy bodies – is more commonly found in the GI systems of individuals with prodromal PD than in healthy controls 2 Stomach Skin Colon • PD may originate in the gut – the so-called ‘gut –brain’ hypothesis, and its pathological progression to the brain may be mediated by the prion-like properties of α-synuclein 3 1. Tolosa et al. Brain 2015; 138(8): 2120– 2122; 2. Wood. Nat Rev Neurol 2016; 12(5): 249; 3. Fasano et al. Lancet Neurol 2015; 14(6): 625– 639 13
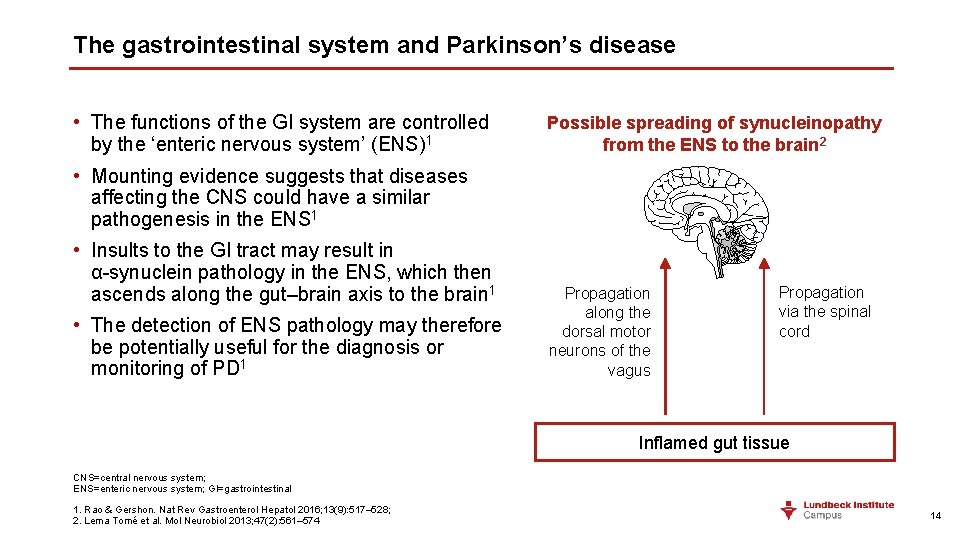
The gastrointestinal system and Parkinson’s disease • The functions of the GI system are controlled by the ‘enteric nervous system’ (ENS)1 Possible spreading of synucleinopathy from the ENS to the brain 2 • Mounting evidence suggests that diseases affecting the CNS could have a similar pathogenesis in the ENS 1 • Insults to the GI tract may result in α-synuclein pathology in the ENS, which then ascends along the gut–brain axis to the brain 1 • The detection of ENS pathology may therefore be potentially useful for the diagnosis or monitoring of PD 1 Propagation along the dorsal motor neurons of the vagus Propagation via the spinal cord Inflamed gut tissue CNS=central nervous system; ENS=enteric nervous system; GI=gastrointestinal 1. Rao & Gershon. Nat Rev Gastroenterol Hepatol 2016; 13(9): 517– 528; 2. Lema Tomé et al. Mol Neurobiol 2013; 47(2): 561– 574 14

α-synuclein and the ‘prion hypothesis’ of PD 15

Aggregation of misfolded proteins • At the heart of the PD disease process is the misfolding and propagation of the protein α-synuclein 1 • The pathology typical of PD is first seen, within the brain, in the dorsal motor nucleus, and spreads upwards from there 2 • A milestone in understanding came in 2008, when Lewy body-like inclusions were observed in neurons that had been grafted (from foetal tissue) into the brains of patients with PD, 11– 16 years after the grafts were performed. 1 This observation was ground-breaking, because it suggested host-to-graft transmission of PD pathology 1 1. Hauser. Lancet Neurol 2015; 14(8): 785– 786; 2. Braak et al. Cell Tissue Res 2004; 318(1): 121– 134 16
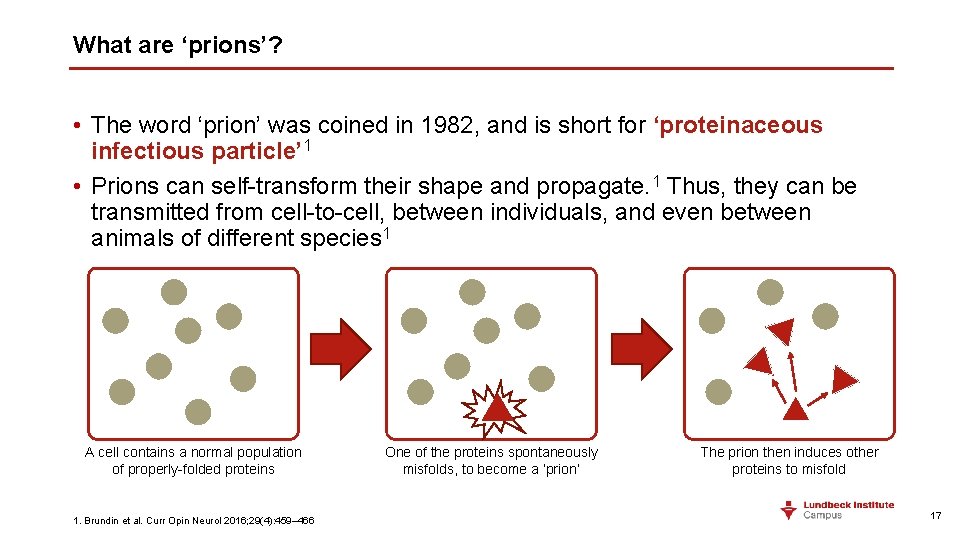
What are ‘prions’? • The word ‘prion’ was coined in 1982, and is short for ‘proteinaceous infectious particle’ 1 • Prions can self-transform their shape and propagate. 1 Thus, they can be transmitted from cell-to-cell, between individuals, and even between animals of different species 1 A cell contains a normal population of properly-folded proteins 1. Brundin et al. Curr Opin Neurol 2016; 29(4): 459– 466 One of the proteins spontaneously misfolds, to become a ‘prion’ The prion then induces other proteins to misfold 17

How strong is the evidence that Parkinson’s disease is a prion disease? Evidence for the prion hypothesis 1 • Neurons can both secrete α-synuclein and take it up from the extracellular space • α-synuclein added to the medium of cultured neurons is taken up by the cells • Once taken up, the α-synuclein oligomerises (bonds chemically) with endogenous α-synuclein to form aggregates • An obvious question arises: is PD a communicable disease? • Although it is hard to be certain, studies looking for epidemiological evidence have not found any evidence that PD can be transmitted from person to person 1 • There are known prion disorders that are not communicable – such as sporadic Creutzfeldt–Jakob disease 1 • Animal studies have supported the cell culture observations outlined above Experimental studies demonstrating cell-to-cell transfer of α-synuclein pathology firmly establish that, in addition to its normal folding, α-synuclein can fold in an altered, self-propagating manner 1 1. Brundin et al. Curr Opin Neurol 2016; 29(4): 459– 466 18

Prions and dementia in Parkinson’s disease • The spread of α-synuclein pathology from the brainstem to certain cortical structures of the brain appears to be the strongest correlate of dementia in patients with PD 1 • Furthermore, up to 50% of patients with PD dementia go on to develop sufficient numbers of the amyloid-β plaques and tau tangles for a secondary diagnosis of Alzheimer’s disease 1 • The plaques and tangles may act synergistically with α-synuclein pathology to confer a worse prognosis in those patients with underlying pathologies 1 1. Irwin et al. Nat Rev Neurosci 2013; 14(9): 626– 636 19

Risk factors for Parkinson’s disease 20
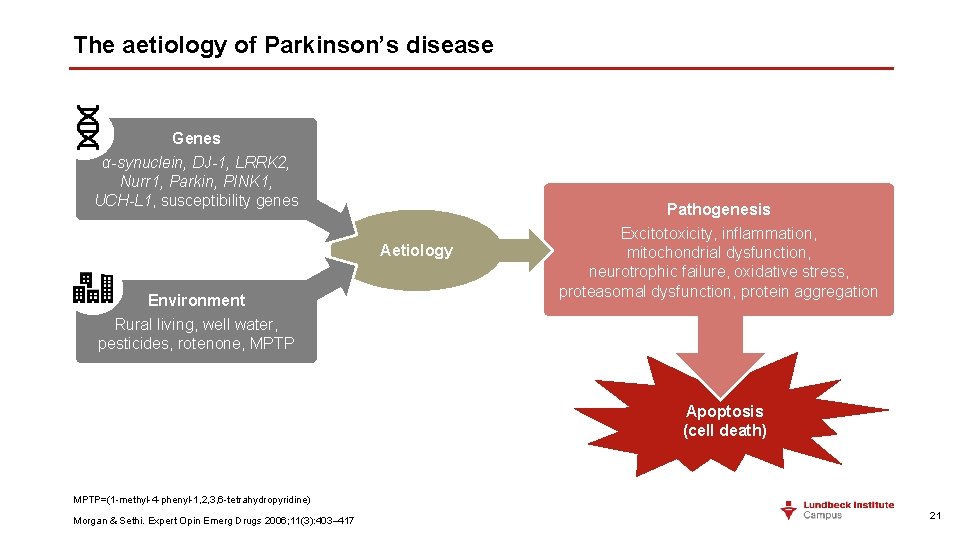
The aetiology of Parkinson’s disease Genes α-synuclein, DJ-1, LRRK 2, Nurr 1, Parkin, PINK 1, UCH-L 1, susceptibility genes Aetiology Environment Rural living, well water, pesticides, rotenone, MPTP Pathogenesis Excitotoxicity, inflammation, mitochondrial dysfunction, neurotrophic failure, oxidative stress, proteasomal dysfunction, protein aggregation Apoptosis (cell death) MPTP=(1 -methyl-4 -phenyl-1, 2, 3, 6 -tetrahydropyridine) Morgan & Sethi. Expert Opin Emerg Drugs 2006; 11(3): 403– 417 21

Environmental risk factors for Parkinson’s disease Risk factors for the development of PD 1 Level of evidence Environmental risk factor Increased risk (robust evidence) • Pesticides Increased risk (weak evidence) • High milk consumption (men) • High iron intake • Chronic anaemia • Hyperuricaemia • Tobacco use • Coffee consumption • High vitamin E intake • Moderate alcohol consumption • Tea consumption • Vigorous physical exercise • NSAID Reduced risk (robust evidence) Reduced risk (weak evidence) • One of the most well elucidated risk factors for PD is exposure to pesticides 1 • Certain pesticides are known to destroy dopamine neurons, which is why they are used in animal models of PD 1 • Other factors are known to reduce the risk of developing PD, these include caffeine intake 1 • Caffeine is known to activate adenosine receptors, which is thought to be neuroprotective 1 NSAID=non-steroidal anti-inflammatory drug 1. Campdelacreu. Neurología 2014; 29(9): 541– 549 22

Susceptibility genes • There are genes which, rather than causing PD, confer an increased risk of developing PD – these are known as susceptibility genes 1 The genetic architecture of PD 1 PLA 2 G 6 Very high risk (causal) ATP 13 A 2 PINK 1 FBXO 7 SNCA mutations VPS 35 DJ-1 Parkin GBA LRRK 2 SNCA MAPT Autosomal recessive Autosomal dominant Low risk Susceptibility genes Juvenile onset Early onset SYT 11/RAB 25, BST 1, CCDC 62/HIP 1 R, PARK 16, DGKQ/GAK, ITGA 8, MCCC 1/LAMP 3, STK 39, ACMSD/TMTM 163, HLA, STBD 1, GPNMB, FGF 20, STX 1 B • There is an increasing number of genes that are known to be related to the risk of developing PD 2 • The genes identified thus far are involved in pathways such as: 2 • Those involving the mitochondria, (mitochondrial dysfunction, oxidative stress, mitochondrial fission and fusion, and mitophagy) • Those involved in aberrant protein degradation, and proteasomal and lysosomal dysfunction Late onset The circle size represents an approximation of the percent of cases due to known genetic causes 1. Clarimón & Kulisevsky. Curr Genomics 2013; 14(8): 560– 567; 2. Schulte & Gasser. Appl Clin Genet 2011; 4: 67– 80 23
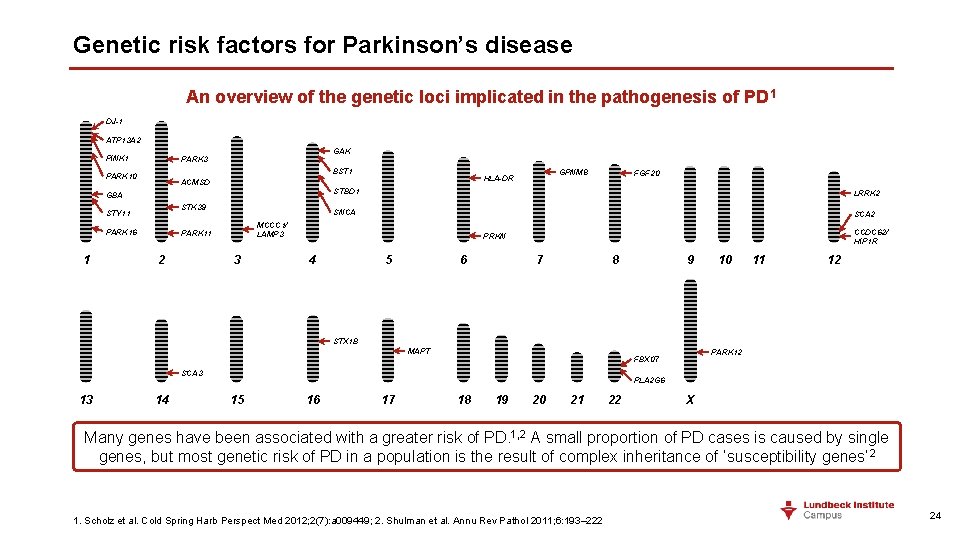
Genetic risk factors for Parkinson’s disease An overview of the genetic loci implicated in the pathogenesis of PD 1 DJ-1 ATP 13 A 2 PINK 1 GAK PARK 3 BST 1 PARK 10 GBA STK 39 STY 11 PARK 16 1 2 3 FGF 20 STBD 1 LRRK 2 SNCA SCA 2 MCCC 1/ LAMP 3 PARK 11 GPNM 8 HLA-DR ACMSD CCDC 62/ HIP 1 R PRKN 4 5 6 7 8 9 10 11 12 STX 1 B MAPT SCA 3 13 14 PARK 12 FBX 07 PLA 2 G 6 15 16 17 18 19 20 21 22 X Many genes have been associated with a greater risk of PD. 1, 2 A small proportion of PD cases is caused by single genes, but most genetic risk of PD in a population is the result of complex inheritance of ‘susceptibility genes’ 2 1. Scholz et al. Cold Spring Harb Perspect Med 2012; 2(7): a 009449; 2. Shulman et al. Annu Rev Pathol 2011; 6: 193– 222 24
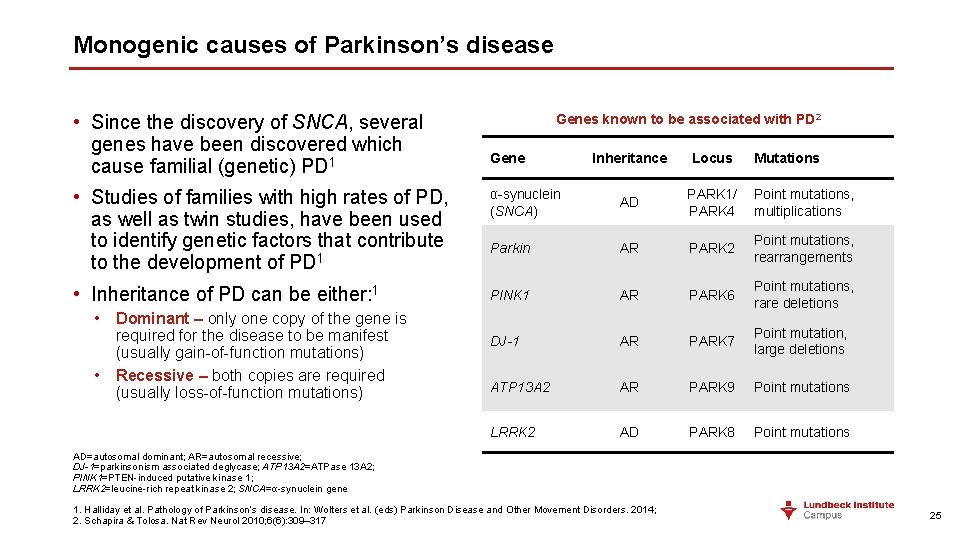
Monogenic causes of Parkinson’s disease • Since the discovery of SNCA, several genes have been discovered which cause familial (genetic) PD 1 Genes known to be associated with PD 2 Gene Inheritance Locus Mutations • Studies of families with high rates of PD, as well as twin studies, have been used to identify genetic factors that contribute to the development of PD 1 α-synuclein (SNCA) AD PARK 1/ PARK 4 Point mutations, multiplications Parkin AR PARK 2 Point mutations, rearrangements • Inheritance of PD can be either: 1 PINK 1 AR PARK 6 Point mutations, rare deletions DJ-1 AR PARK 7 Point mutation, large deletions ATP 13 A 2 AR PARK 9 Point mutations LRRK 2 AD PARK 8 Point mutations • Dominant – only one copy of the gene is required for the disease to be manifest (usually gain-of-function mutations) • Recessive – both copies are required (usually loss-of-function mutations) AD=autosomal dominant; AR=autosomal recessive; DJ-1=parkinsonism associated deglycase; ATP 13 A 2=ATPase 13 A 2; PINK 1=PTEN-induced putative kinase 1; LRRK 2=leucine-rich repeat kinase 2; SNCA=α-synuclein gene 1. Halliday et al. Pathology of Parkinson’s disease. In: Wolters et al. (eds) Parkinson Disease and Other Movement Disorders. 2014; 2. Schapira & Tolosa. Nat Rev Neurol 2010; 6(6): 309– 317 25

Genetic factors in Parkinson’s disease varying by ethnicity • It is increasingly being recognised that ethnic background should be taken into account when considering the genetic risk for developing PD 1 • When the results of GWAS studies conducted in different ethic groups have been compared, differences in allele frequencies and effect sizes have been observed 1 • This means that a patient may be wrongly characterised as having an increased risk of developing PD, because they are positive for a genetic association that is only a risk factor for individuals of a different ethnic group 1 1. Foo et al. Nat Rev Neurol 2012; 8: 508– 517 26

Common pathways underlying PD pathogenesis 27
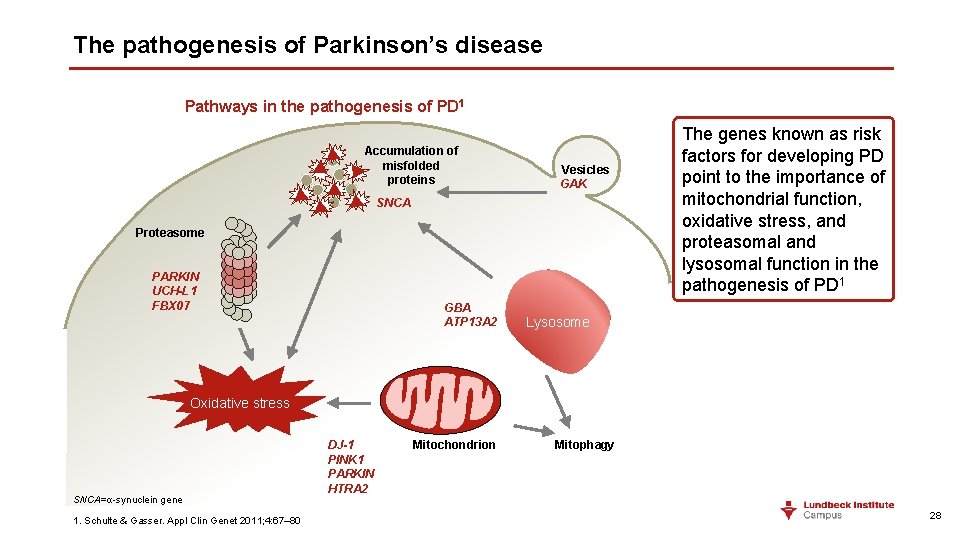
The pathogenesis of Parkinson’s disease Pathways in the pathogenesis of PD 1 Accumulation of misfolded proteins Vesicles GAK SNCA Proteasome PARKIN UCH-L 1 FBX 07 GBA ATP 13 A 2 The genes known as risk factors for developing PD point to the importance of mitochondrial function, oxidative stress, and proteasomal and lysosomal function in the pathogenesis of PD 1 Lysosome Oxidative stress SNCA=α-synuclein gene 1. Schulte & Gasser. Appl Clin Genet 2011; 4: 67– 80 DJ-1 PINK 1 PARKIN HTRA 2 Mitochondrion Mitophagy 28
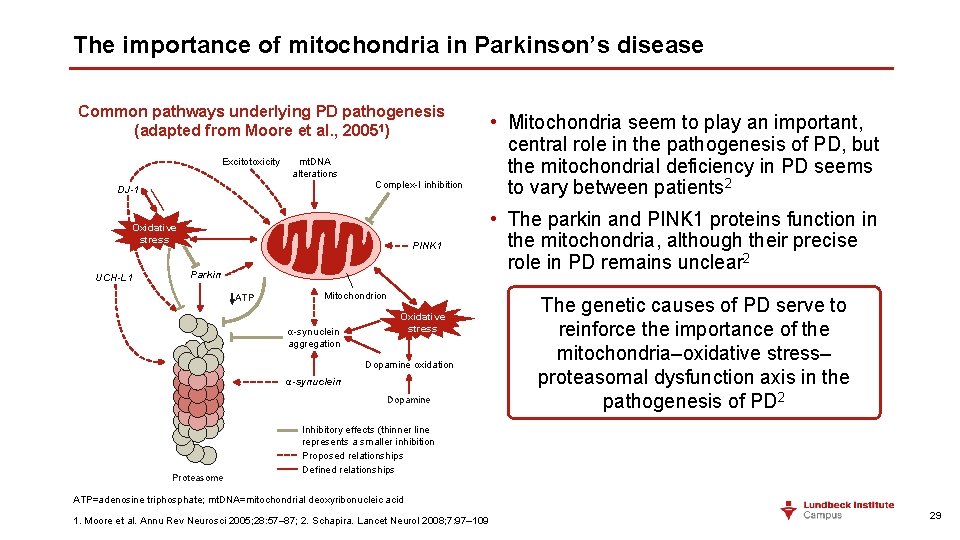
The importance of mitochondria in Parkinson’s disease Common pathways underlying PD pathogenesis (adapted from Moore et al. , 20051) Excitotoxicity mt. DNA alterations DJ-1 Complex-I inhibition Oxidative stress UCH-L 1 PINK 1 Parkin ATP Mitochondrion α-synuclein aggregation Oxidative stress Dopamine oxidation α-synuclein Dopamine Proteasome • Mitochondria seem to play an important, central role in the pathogenesis of PD, but the mitochondrial deficiency in PD seems to vary between patients 2 • The parkin and PINK 1 proteins function in the mitochondria, although their precise role in PD remains unclear 2 The genetic causes of PD serve to reinforce the importance of the mitochondria–oxidative stress– proteasomal dysfunction axis in the pathogenesis of PD 2 Inhibitory effects (thinner line represents a smaller inhibition Proposed relationships Defined relationships ATP=adenosine triphosphate; mt. DNA=mitochondrial deoxyribonucleic acid 1. Moore et al. Annu Rev Neurosci 2005; 28: 57– 87; 2. Schapira. Lancet Neurol 2008; 7: 97– 109 29

Mitochondrial dysfunction plays a key role in Parkinson’s disease • Mitochondrial dysfunction has been observed in cells of the substantia nigra in patients with PD 1 • These cells show impaired mitochondrial complex-I activity 1 • Similar dysfunction is also seen in platelets and in skeletal muscle cells of patients with PD 1 • Several mutations in mitochondrial DNA have been associated with PD 1 • Cybrid cells (human cytoplasmic hybrid cells that lack their own mitochondrial DNA) containing mitochondrial DNA from patients with PD have shown reduced levels of mitochondrial complex-I activity 1, 2 Mitochondrial dysfunction, which occurs as a result of mutations in mitochondrial DNA, appears to play a key role in the pathogenesis of PD 1, 2 DNA=deoxyribonucleic acid 1. Chaturvedi & Beal. Ann N Y Acad Sci 2008; 1147: 395– 412; 2. Trimmer et al. J Neurochem 2004; 88(4): 800– 812 30
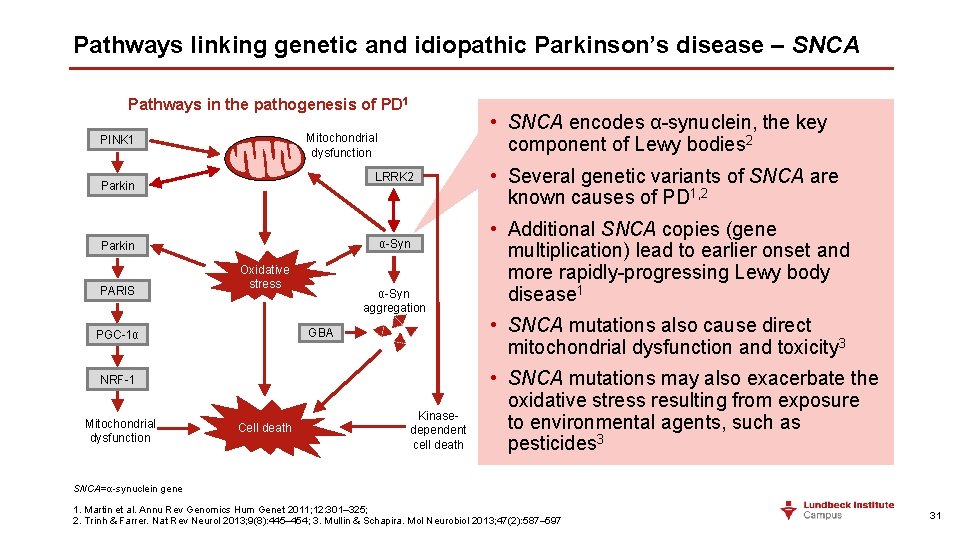
Pathways linking genetic and idiopathic Parkinson’s disease – SNCA Pathways in the pathogenesis of PD 1 PINK 1 LRRK 2 Parkin α-Syn Parkin PARIS • SNCA encodes α-synuclein, the key component of Lewy bodies 2 Mitochondrial dysfunction Oxidative stress α-Syn aggregation GBA PGC-1α NRF-1 Mitochondrial dysfunction Cell death Kinasedependent cell death • Several genetic variants of SNCA are known causes of PD 1, 2 • Additional SNCA copies (gene multiplication) lead to earlier onset and more rapidly-progressing Lewy body disease 1 • SNCA mutations also cause direct mitochondrial dysfunction and toxicity 3 • SNCA mutations may also exacerbate the oxidative stress resulting from exposure to environmental agents, such as pesticides 3 SNCA=α-synuclein gene 1. Martin et al. Annu Rev Genomics Hum Genet 2011; 12: 301– 325; 2. Trinh & Farrer. Nat Rev Neurol 2013; 9(8): 445– 454; 3. Mullin & Schapira. Mol Neurobiol 2013; 47(2): 587– 597 31
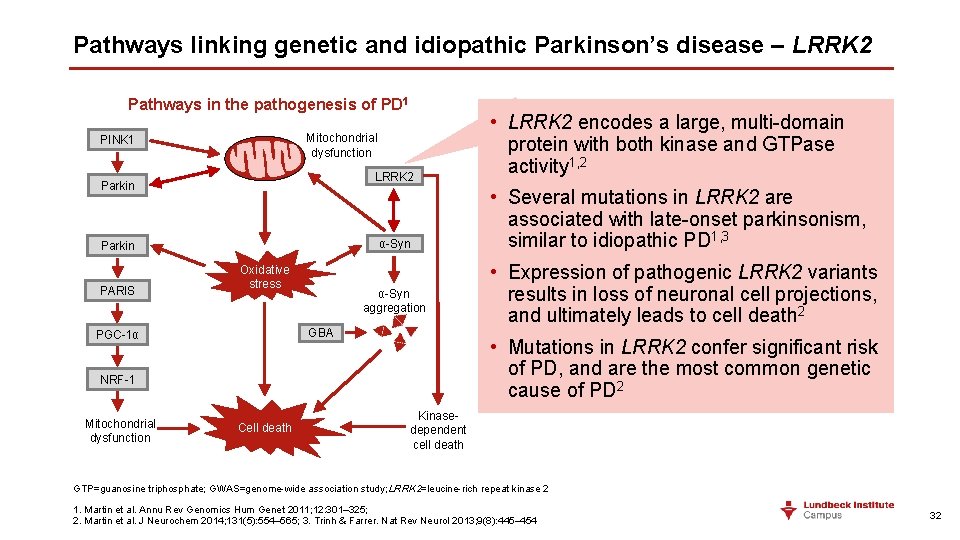
Pathways linking genetic and idiopathic Parkinson’s disease – LRRK 2 Pathways in the pathogenesis of PD 1 Mitochondrial dysfunction PINK 1 LRRK 2 Parkin α-Syn Parkin PARIS Oxidative stress α-Syn aggregation GBA PGC-1α Mitochondrial dysfunction Cell death • Several mutations in LRRK 2 are associated with late-onset parkinsonism, similar to idiopathic PD 1, 3 • Expression of pathogenic LRRK 2 variants results in loss of neuronal cell projections, and ultimately leads to cell death 2 • Mutations in LRRK 2 confer significant risk of PD, and are the most common genetic cause of PD 2 NRF-1 Cell death • LRRK 2 encodes a large, multi-domain protein with both kinase and GTPase activity 1, 2 Kinasedependent cell death GTP=guanosine triphosphate; GWAS=genome-wide association study; LRRK 2=leucine-rich repeat kinase 2 1. Martin et al. Annu Rev Genomics Hum Genet 2011; 12: 301– 325; 2. Martin et al. J Neurochem 2014; 131(5): 554– 565; 3. Trinh & Farrer. Nat Rev Neurol 2013; 9(8): 445– 454 32
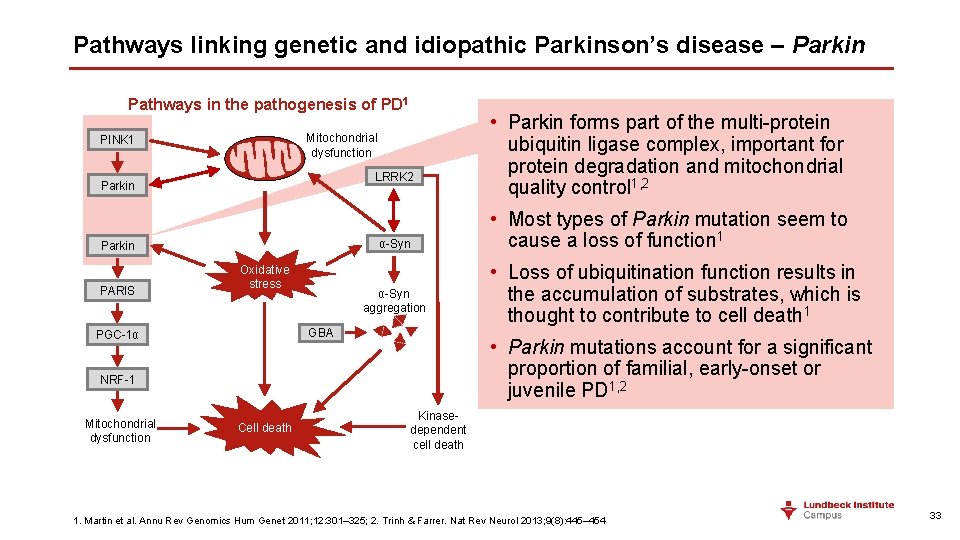
Pathways linking genetic and idiopathic Parkinson’s disease – Parkin Pathways in the pathogenesis of PD 1 Mitochondrial dysfunction PINK 1 LRRK 2 Parkin α-Syn Parkin PARIS Oxidative stress α-Syn aggregation GBA PGC-1α Mitochondrial dysfunction Cell death • Most types of Parkin mutation seem to cause a loss of function 1 • Loss of ubiquitination function results in the accumulation of substrates, which is thought to contribute to cell death 1 • Parkin mutations account for a significant proportion of familial, early-onset or juvenile PD 1, 2 NRF-1 Cell death • Parkin forms part of the multi-protein ubiquitin ligase complex, important for protein degradation and mitochondrial quality control 1, 2 Kinasedependent cell death 1. Martin et al. Annu Rev Genomics Hum Genet 2011; 12: 301– 325; 2. Trinh & Farrer. Nat Rev Neurol 2013; 9(8): 445– 454 33

The role of neuroinflammation in Parkinson’s disease • The presence of aggregated α-synuclein triggers the activation of microglia, causing them to: 1 • Change their morpho-functional characteristics • Increase rates of proliferation • Release inflammatory mediators • Inflammatory molecules recruit peripheral leukocytes into the CNS, which promotes clearance of cell debris 1 • Inflammatory mediators can also lead to neurodegeneration 1 • Neurodegeneration further activates inflammatory mechanisms, resulting in a vicious cycle of inflammation and neuronal death 1 CNS=central nervous system 1. Rocha et al. Biomed Res Int 2015; 2015: 628192 34

Gaucher disease – glucocerebrosidase • Mutations in the glucocerebrosidase gene (GBA 1) cause the autosomal-recessive lysosomal storage disorder ‘Gaucher disease’ 1 • Patients with GBA 1 -PD still display the cardinal symptoms of PD, but they tend towards younger onset, and have a greater risk of cognitive impairment 1 • Homozygous individuals with Gaucher disease and their asymptomatic heterozygous relatives have a higher risk of PD than the general population 1 • Individuals with the GBA 1 mutation without clinical evidence of PD still exhibit the prodromal characteristics of the disease, such as olfactory dysfunction 1 • The lifetime risk of developing PD in individuals with GBA 1 mutations is up to 20% at 70 years, and 30% at 80 years 1 • The proportion of PD patients with mutations in GBA 1 is estimated to be ~5– 10%1 Mutations in GBA 1 represent the most important risk factor for PD identified to date 1 1. Schapira. Mol Cell Neurosci 2015; 66(Pt A): 37– 42 • The response of patients with GBA 1 -PD to dopaminergic medication appears to mimic that of individuals with idiopathic PD 1 • Given its importance as a risk factor for PD, GBA 1 is being examined for its therapeutic potential 1 35
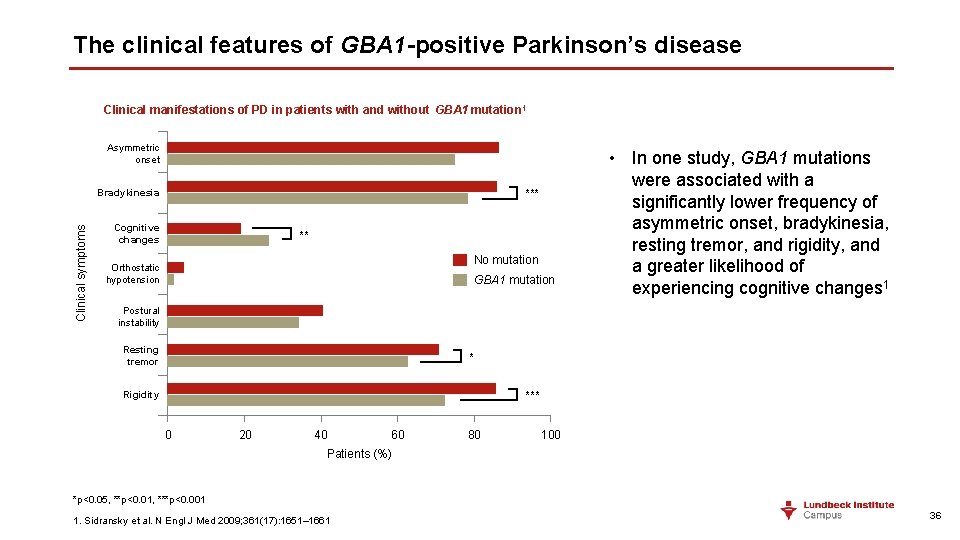
The clinical features of GBA 1 -positive Parkinson’s disease Clinical manifestations of PD in patients with and without GBA 1 mutation 1 Asymmetric onset Clinical symptoms Bradykinesia *** Cognitive changes ** No mutation Orthostatic hypotension GBA 1 mutation • In one study, GBA 1 mutations were associated with a significantly lower frequency of asymmetric onset, bradykinesia, resting tremor, and rigidity, and a greater likelihood of experiencing cognitive changes 1 Postural instability Resting tremor * Rigidity *** 0 20 40 60 80 100 Patients (%) *p<0. 05, **p<0. 01, ***p<0. 001 1. Sidransky et al. N Engl J Med 2009; 361(17): 1651– 1661 36

Designing treatments based on the pathogenesis model; where to target 37
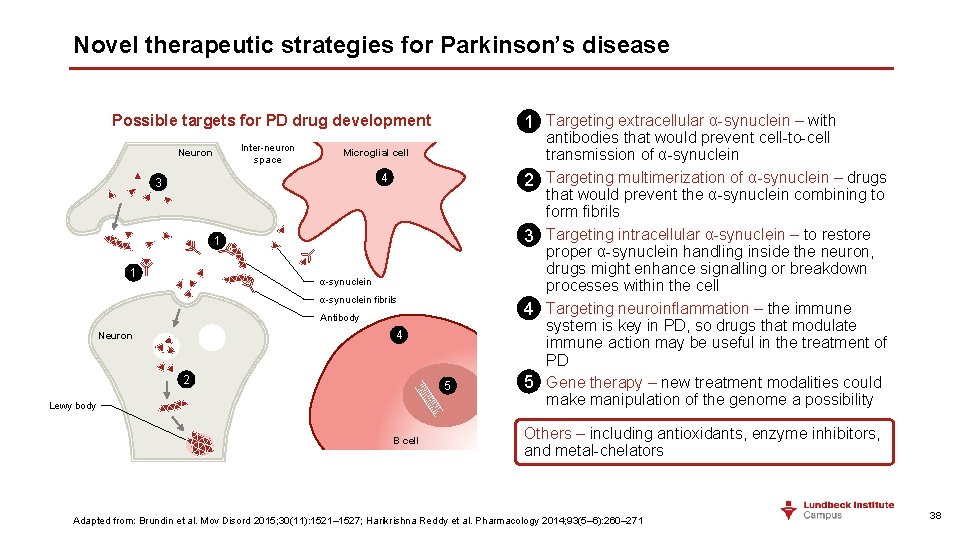
Novel therapeutic strategies for Parkinson’s disease Possible targets for PD drug development Inter-neuron space Neuron Microglial cell 4 3 1 1 α-synuclein fibrils Antibody 4 Neuron 2 5 Lewy body B cell 1. 1 Targeting extracellular α-synuclein – with antibodies that would prevent cell-to-cell transmission of α-synuclein 2. 2 Targeting multimerization of α-synuclein – drugs that would prevent the α-synuclein combining to form fibrils 3. 3 Targeting intracellular α-synuclein – to restore proper α-synuclein handling inside the neuron, drugs might enhance signalling or breakdown processes within the cell 4. 4 Targeting neuroinflammation – the immune system is key in PD, so drugs that modulate immune action may be useful in the treatment of PD 5. 5 Gene therapy – new treatment modalities could make manipulation of the genome a possibility Others – including antioxidants, enzyme inhibitors, and metal-chelators Adapted from: Brundin et al. Mov Disord 2015; 30(11): 1521– 1527; Harikrishna Reddy et al. Pharmacology 2014; 93(5– 6): 260– 271 38

The future of Parkinson’s disease treatments • Researchers are perennially humbled by the challenges of finding disease-modifying strategies for neurodegenerative diseases 1, 2 • These disease-modifying strategies, once discovered, may be best used by: 1, 2 • Combining drugs with different mechanisms of action (for example, as is routinely done in the treatment of cancers) • Appreciating that PD is a collection of diseases, and that each phenotype may need to be treated differently 1. Brundin et al. Mov Disord 2015; 30(11): 1521– 1527; 2. Kalia et al. Mov Disord 2015; 30(11): 1442– 1450 39
- Slides: 39