Network metaanalysis modelling in benefitrisk assessment Gert van

Network meta-analysis modelling in benefitrisk assessment Gert van Valkenhoef

Benefit-risk assessment • Regulatory BR “does not require a new drug to be assessed against other available treatments” 1 • Payers becoming more important for market access – Relative Efficacy / Relative Effectiveness – Better than placebo is not enough • Non-inferiority trials – difficult in current framework – Assay sensitivity – Drift into failure • Other decision makers (e. g. prescriber, patient) – Assess all available options 1. H-G Eichler et al. , Nat Rev Drug Discov. 2010; 9(4): 277 -291 (doi: 10. 1038/nrd 3079)

Network meta-analysis • “In the absence of direct head-to-head trails, a next best approach … is … common reference indirect comparison” 1 • Network meta-analysis 2, 3, 4: – Extension of normal meta-analysis – Compare ≥ 2 alternative treatments – Using an evidence network of trials – Combination of direct + indirect evidence – Assessment of consistency in network – Incorporates all available evidence 1. H-G Eichler et al. , Nat Rev Drug Discov. 2010; 9(4): 277 -291 (doi: 10. 1038/nrd 3079) 2. T Lumley, Stat in Med. 2002; 21(16): 2313– 2324 (doi: 10. 1002/sim. 1201) 3. G Lu & AE Ades, J Am Stat Assoc. 2006; 101(474): 447 -459 (doi: 10. 1198/016214505000001302) 4. G Salanti et al. , Stat Methods Med Res. 2008; 17(3): 279 -301 (doi: 10. 1177/0962280207080643)

Our proposal • BR assessment on basis of updating network meta-analysis • Indirect comparisons: assess all relevant options – e. g. indirect comparison to placebo • Interpretation of non-inferiority trials – Incorporate in network of trials – “Drift into failure” detectable – RE, not the chosen margin, is central • Network meta-analysis is (meta-)observational – There may be unknown confounding

Example: anti-depressants Fluoxetine Paroxetine Placebo Sertraline Venlafaxine HAM-D response
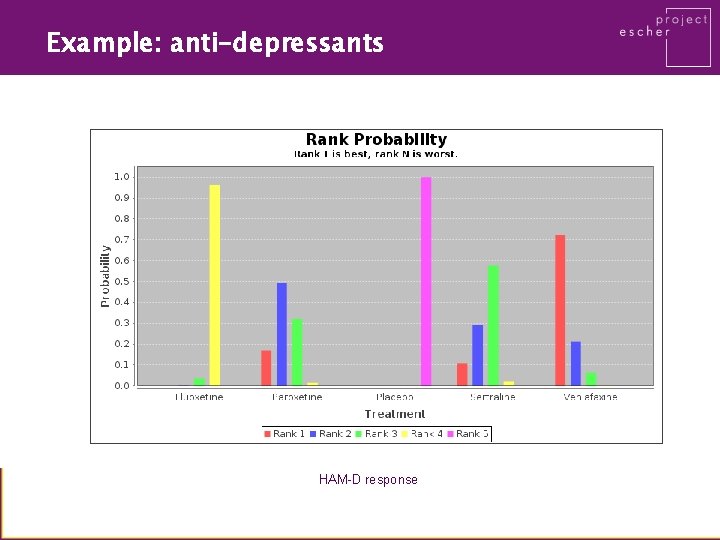
Example: anti-depressants HAM-D response
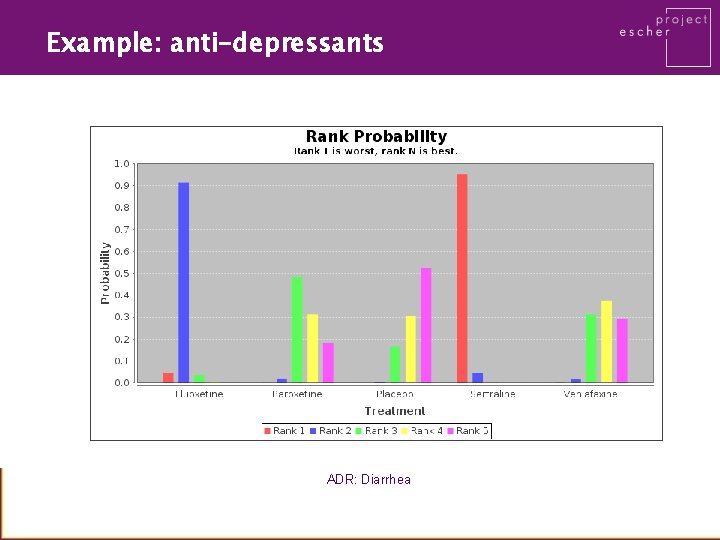
Example: anti-depressants ADR: Diarrhea
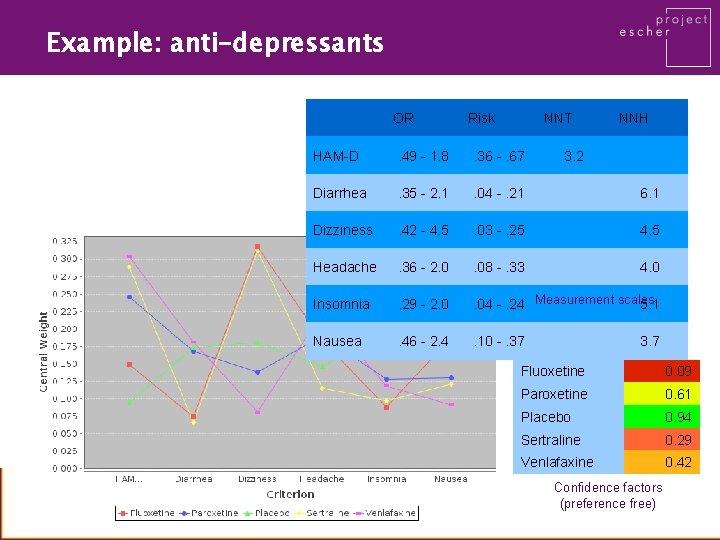
Example: anti-depressants OR Risk NNT NNH HAM-D . 49 - 1. 8 . 36 -. 67 3. 2 Diarrhea . 35 - 2. 1 . 04 -. 21 6. 1 Dizziness . 42 - 4. 5 . 03 -. 25 4. 5 Headache . 36 - 2. 0 . 08 -. 33 4. 0 Insomnia . 29 - 2. 0 . 04 -. 24 Measurement scales 5. 1 Nausea . 46 - 2. 4 . 10 -. 37 3. 7 Fluoxetine 0. 09 Paroxetine 0. 61 Placebo 0. 94 Sertraline 0. 29 Venlafaxine 0. 42 Confidence factors (preference free)

Conclusions • Network meta-analysis useful in BR assessment – When placebo trials are not available – To detect “drift into failure” – To use all available evidence – Give insight for > 2 alternatives • Network meta-analysis + multi-criteria model – Makes trade-offs explicit – Takes into account full uncertainty – Some options are bad, regardless of preference
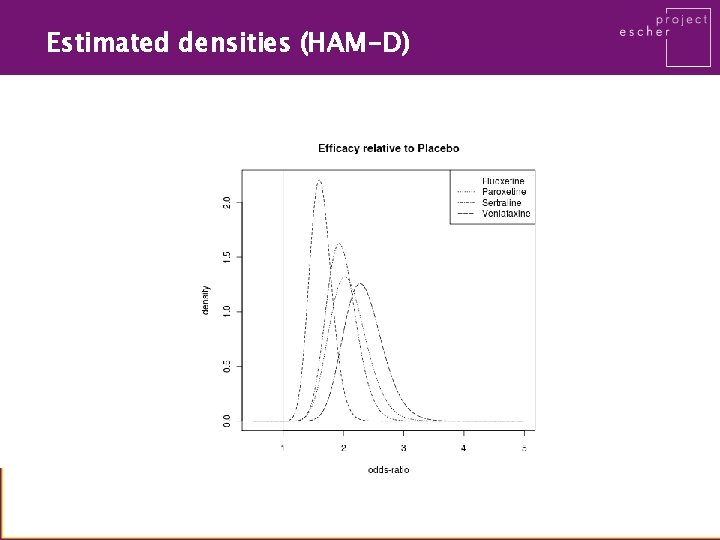
Estimated densities (HAM-D)
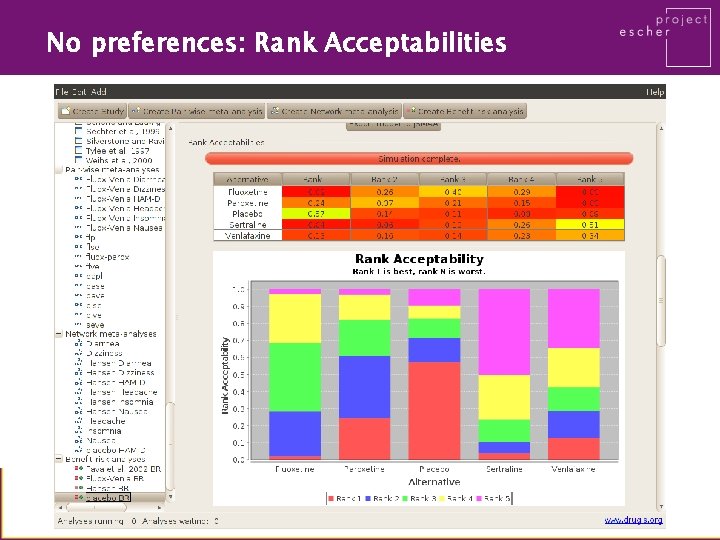
No preferences: Rank Acceptabilities
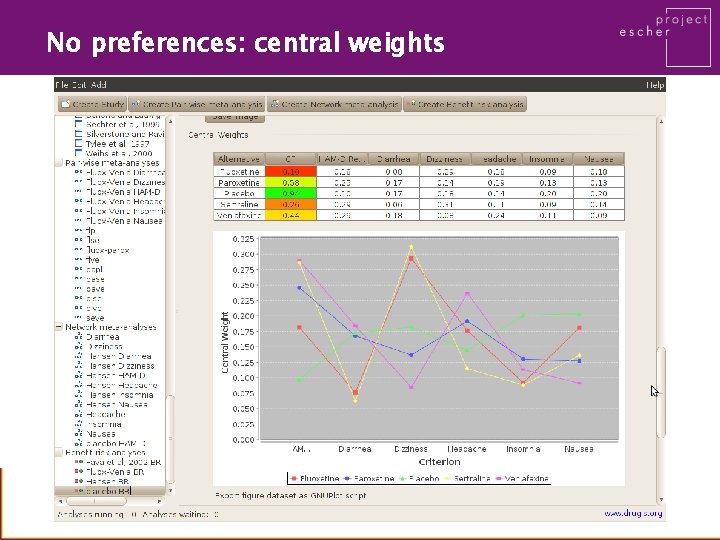
No preferences: central weights
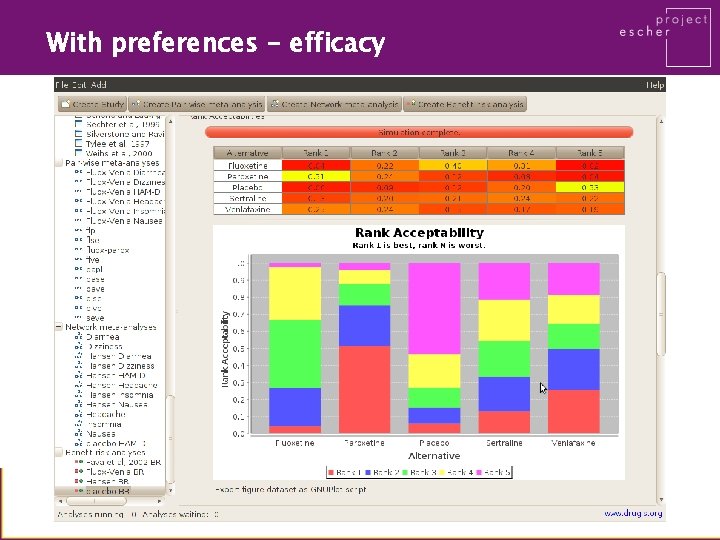
With preferences - efficacy
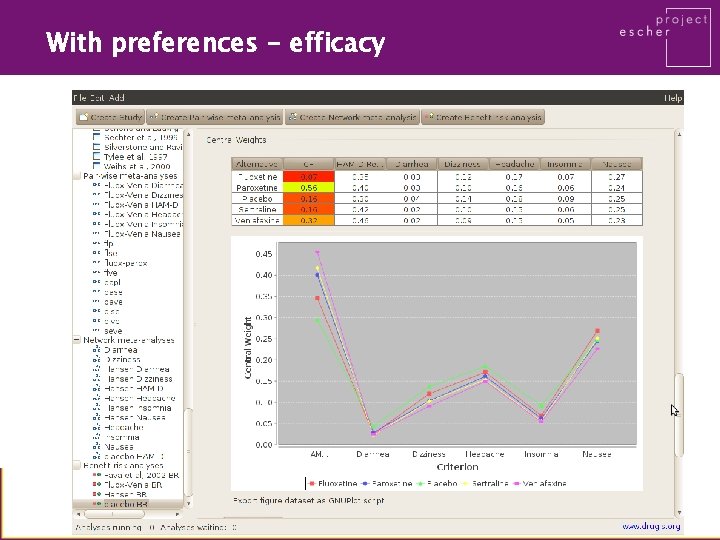
With preferences - efficacy
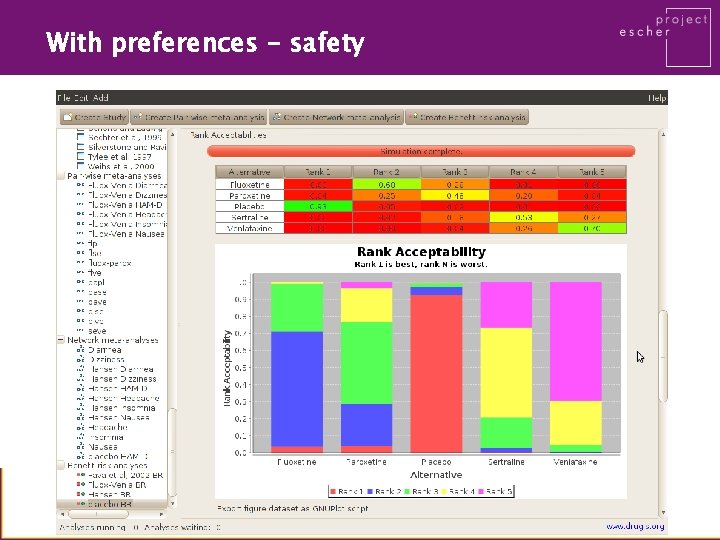
With preferences - safety
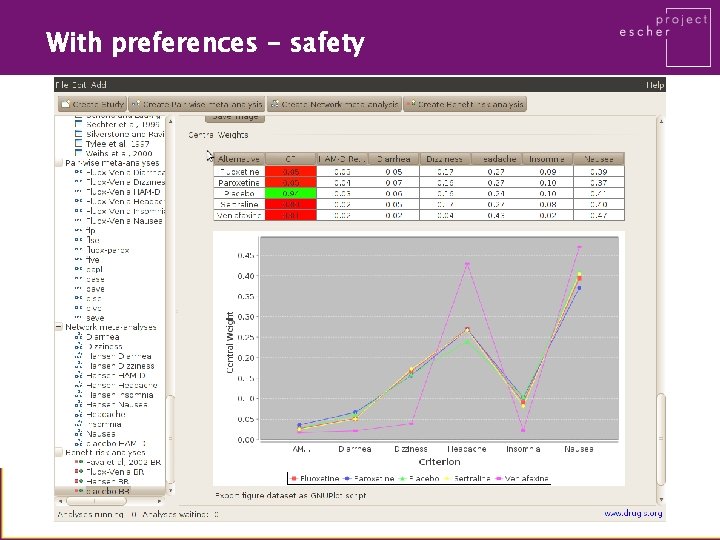
With preferences - safety
- Slides: 16