NETCHEM Remote Access Laboratory Guide Exploring and observing


NETCHEM Remote Access Laboratory Guide Exploring and observing of yeasts from plant materials- endemic plants ëith industrial & medicinal importance Through this exercise, you will learn : ü To know and to be familiar with the techniques used in microbiology for the isolation of endemic microorganisms from medicinal plants and those added from environments and other pollution sources. ü To differentiate between techniques used for the isolation of bacteria and yeasts & molds. ü To evaluate the total number of microorganisms (CFU/unit) being familiar with a Colony Counter instrument. ü To select with screening methods yeasts with industrial importance. ü To be familiar with classical methods of their identification, morphological and physiological methods. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Background Yeasts are unicellular microorganisms, parts of three important families Endomycetaceae, Sporobolomycetaceae and Cryptococcaceae. ØThe ascosporogenous yeasts of Endomycetaceae family form asci & ascospores. ØThe yeasts grouped in Sporobolomycetaceae family forms ballistospores. ØThe asporogenous yeasts which form neither ascospores nor ballistospores are grouped in Cryptococcaceae family. The determination of the total number of microorganisms in plant substrates is an important step to identify their presence. The preparation of sampling to determine the total number of microorganisms grown in various anatomical parts of the plant in the study, is also the first step to a reliable result. The total number of microorganisms serves to find the yeasts colonies and to study with a microscopical examination their most important characteristics. This study is important because in identifying a yeast, the decision has first to be made to which of the three above families the yeast strain in question belongs (able to form ascospores or ballistospores). ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Background An efficient screening method serves to separate yeasts from other developed microorganisms. Selected strains are studied to determine: Ø macroscopical aspects of the culture, Øthe formation of pseudomycelium, Øvegetative and sexual reproduction Physiological properties for the differentiation of the strains are determined as well (assimilation of various types of glucides, glucides fermentation with ethanol and CO 2 formation, the ability to use various nitrogen compounds as N sources). The assimilation of glucose, galactose, sucrose, lactose, etc. , using auxanographic tests, is related with their fermentation because all fermentable sugars are also assimilated. Assimilation of N sources includes assimilation of nitrates (potassium nitrate). ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Materials For this lab exercise, you will need the following materials: Samples-Juniperus communis ; Rosa cannina Equipments Øcolony counter, Øan optical microscope to be familiar with the yeast cells, Øa microscope with a camera (or a microscope with a tablet in order to have the possibility to see and discuss the cell structure and respective characteristics). ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.
Consumables ØPetri dishes ØMicrobiology lab. supplies, etc. Media ØPlate Count Agar (PCA), Ø Sabouraud Dextrose Agar (SDA), ØPotato Dextrose Agar (PDA), ØCZAPEK Dox Agar. Reagents ØSugars-glucose, galactose, sucrose, lactose ØPotassium nitrate ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Procedure: i. Determination of TNM Ø Preparation of the samples- Prepare 10 g of fruits of Juniperus communis and 10 g of fruits of Rosa cannina, add them in 90 ml sterilized water and mixed them every 5 minutes for about 1 hour. Leave for a few minutes until decanting the solid part. Pipette 1 ml of solution and pour it into a Petri dish. Use PCA, prepared in advance according to a detailed work protocol, for the determination of the total number of microorganisms. Melt first the solid PCA media and pour it warm into Petri dishes. Ø Mix carefully the aliquot with the PCA media using parallel motion and then leave it for solidification. Petri dishes are incubated in a thermostat using growth temperature interval of 28 -30°C. Developed colonies are observed after 24, 48 and 72 hours. It is worked with parallel plates for each aliquot in order to calculate the average result for TNM-CFU (Colony-Forming Units) / ml. Ø Use a colony counter to determine TNM. The number of colonies is determined directly in the whole plate or using different techniques of enumeration as sector enumeration. www. dongnamlab. com ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.
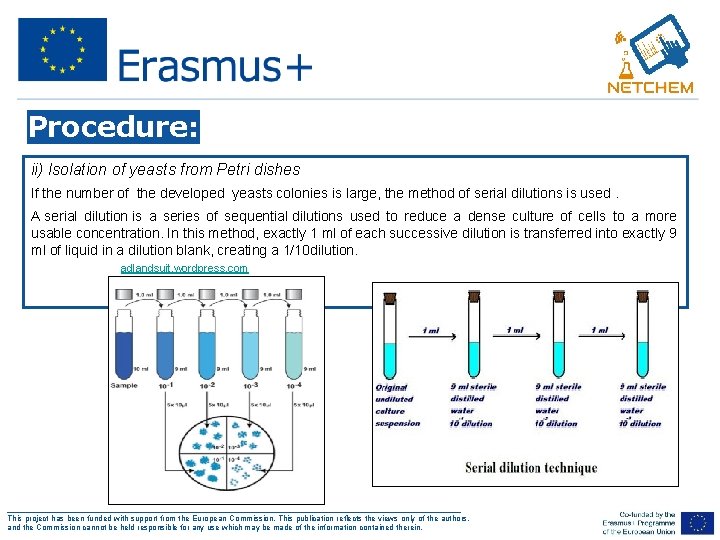
Procedure: ii) Isolation of yeasts from Petri dishes If the number of the developed yeasts colonies is large, the method of serial dilutions is used. A serial dilution is a series of sequential dilutions used to reduce a dense culture of cells to a more usable concentration. In this method, exactly 1 ml of each successive dilution is transferred into exactly 9 ml of liquid in a dilution blank, creating a 1/10 dilution. adlandsuit. wordpress. com ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Procedure: ii) Isolation of yeasts from Petri dishes ØDeveloped yeast colonies are identified using microscopic observation of the preparations. ØThe purification procedure is carried out with successive passages. ØSelected colonies are stored in microbiology glass test tubes with standard solid media culture for yeasts ( Sabouroaud Dextrose Agar-SDA, Potato Dextrose Agar -PDA) Ø 24 -48 hours cultures are used for microscopic observations for the determination of morphological characteristics (macro cultures, cell structure, reproduction, pseudomycelium formation, sporulation) and physiological characteristics (assimilation and fermentation of glucides and nitrate assimilation) ___________________________________________________ This project has been funded ëith support from the European Commission. This publication reflects the vieës only of the authors, and the Commission cannot be held responsible for any use ëhich may be made of the information contained therein.
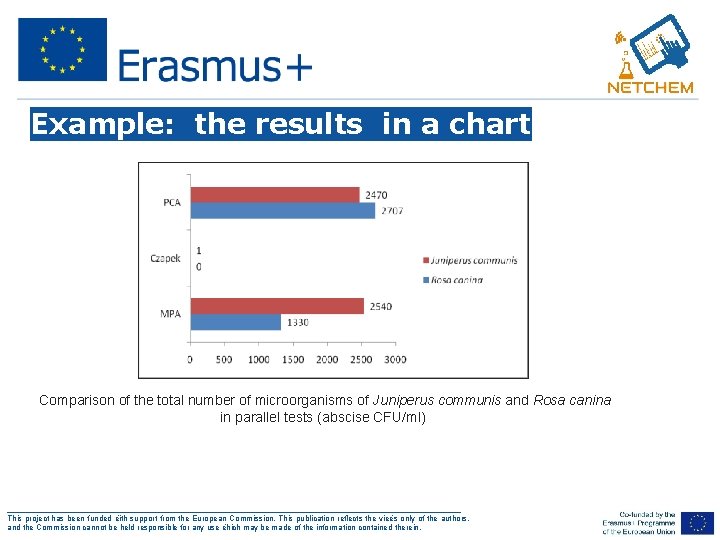
Example: the results in a chart Comparison of the total number of microorganisms of Juniperus communis and Rosa canina in parallel tests (abscise CFU/ml) ___________________________________________________ This project has been funded ëith support from the European Commission. This publication reflects the vieës only of the authors, and the Commission cannot be held responsible for any use ëhich may be made of the information contained therein.

Performing: 1. Microbiological analysis will be performed after the program of Microbiology for the students of diploma of second level and for those students of third level, working in the field of medicinal plants. 2. Before, the students will have to be familiar with theoretical aspects of Industry of Plant Materials-medicinal plants (text books, former scientific works and the experience of other researchers). 3. Students will be in the laboratory together with the performer or in the classroom and the performer in the microbiological laboratory. 4. The performer will introduce students to the basic principles of good laboratory practice, equipments, media and techniques of manipulation.

DESCRIPTION OF THE EDUCATIONAL ELEMENT Yeasts in plant materials (medicinal plants 0 Educational element title Educational field Level of study Industrial Microbiology Studies of second level (Master of Sciences) and Third level (Ph. D studies) Industry of Plant Materials Title of course in which Educational element is implemented (lecture or lab exercise) Title of teaching unit Teacher Target group (study program, year of study) Educational objectives of educational element Required preliminary knowledge and skills Material available at Moodle platform for the educational element: -Type (. mp 4/. avi/. ppt/. pdf/. doc/. jpeg …): -Size (MB): -Used language in the material: Microbiological Analysis of Medicinal Plants Prof. Dr. Rozana Troja Msc in Food Sciences, Food Technology and Microbiology The students will be prepared how to perform the determination of the total number of microorganisms from medicinal plants and how to use screening methods and the microbiological methods of identification to determine yeasts presence Basic knowledge of chemistry, industrial microbiology and microbiological methods of analysis pp presentations English ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, Not always and the Commission cannot be held responsible for any use which may be made of the information contained therein. Remote access classroom-laboratory

___________________________________________________ This project has been funded ëith support from the European Commission. This publication reflects the vieës only of the authors, and the Commission cannot be held responsible for any use ëhich may be made of the information contained therein.

Author, Editor and Referee References This remote access laboratory was created thanks to work done primarily at University of Niš. Contributors to this material were: Rozana Troja Date of creation: September, 2018. Refereeing of this material was done by: Editing into NETCHEM Format and onto NETCHEM platform was completed by: __________________________________________________________ This project has been funded ëith support from the European Commission. This publication reflects the vieës only of the authors, and the Commission cannot be held responsible for any use ëhich may be made of the information contained therein.

References and Supplemental Material The NETCHEM platform was established at the University of Nis in 2016 -2019 through the Erasmus Programme. Please contact a NETCHEM representatives at your institution or visit our website for an expanded contact list. The work included had been led by the NETCHEM staff at your institution. ___________________________________________________ This project has been funded ëith support from the European Commission. This publication reflects the vieës only of the authors, and the Commission cannot be held responsible for any use ëhich may be made of the information contained therein.
- Slides: 15