NELSON LAB VOCABULARY Tyr WiesnerHanks December 12 2014

NELSON LAB VOCABULARY Tyr Wiesner-Hanks December 12, 2014

Corn physiology • Corn has a male inflorescence on the top (the tassel) and multiple female inflorescences along the stem (the ears) • Female flowers can be pollinated with pollen from the same plant (selfing) or other plants (outcrossing) • Crossing is the act of making a controlled pollination
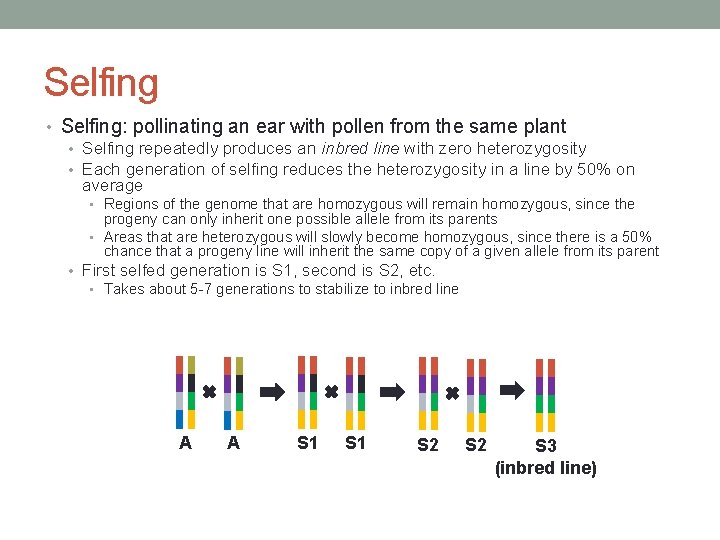
Selfing • Selfing: pollinating an ear with pollen from the same plant • Selfing repeatedly produces an inbred line with zero heterozygosity • Each generation of selfing reduces the heterozygosity in a line by 50% on average • Regions of the genome that are homozygous will remain homozygous, since the progeny can only inherit one possible allele from its parents • Areas that are heterozygous will slowly become homozygous, since there is a 50% chance that a progeny line will inherit the same copy of a given allele from its parent • First selfed generation is S 1, second is S 2, etc. • Takes about 5 -7 generations to stabilize to inbred line A A S 1 S 2 S 3 (inbred line)
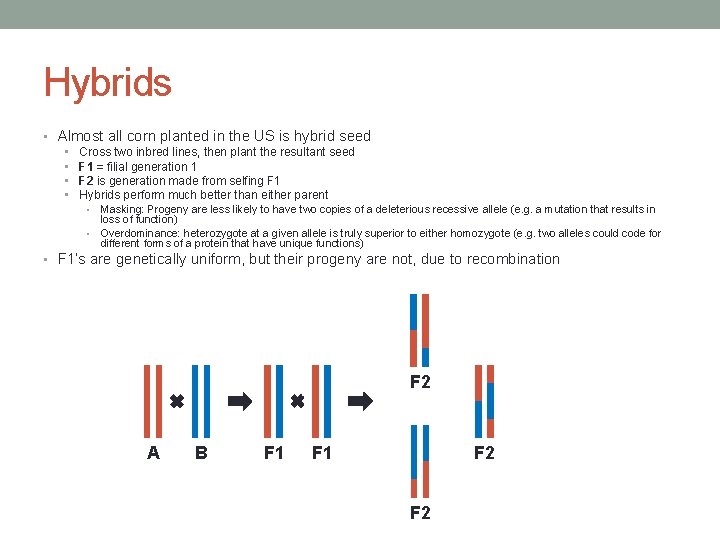
Hybrids • Almost all corn planted in the US is hybrid seed • • Cross two inbred lines, then plant the resultant seed F 1 = filial generation 1 F 2 is generation made from selfing F 1 Hybrids perform much better than either parent Masking: Progeny are less likely to have two copies of a deleterious recessive allele (e. g. a mutation that results in loss of function) • Overdominance: heterozygote at a given allele is truly superior to either homozygote (e. g. two alleles could code for different forms of a protein that have unique functions) • • F 1’s are genetically uniform, but their progeny are not, due to recombination F 2 A B F 1 F 2

Recombinant inbred lines • RILs = recombinant inbred lines • Self an F 2 repeatedly • Resultant lines are inbreds (close to fully homozygous), but have mix of original chromosomes from parents A B F 1 F 2 F 2 F 1
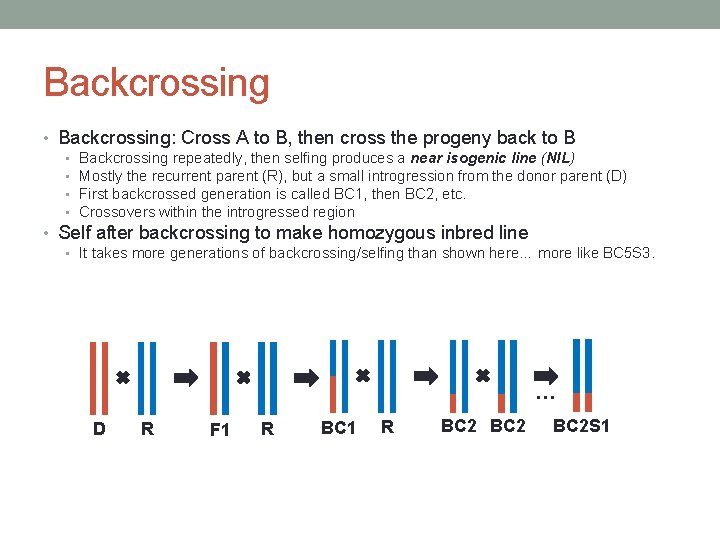
Backcrossing • Backcrossing: Cross A to B, then cross the progeny back to B • • Backcrossing repeatedly, then selfing produces a near isogenic line (NIL) Mostly the recurrent parent (R), but a small introgression from the donor parent (D) First backcrossed generation is called BC 1, then BC 2, etc. Crossovers within the introgressed region • Self after backcrossing to make homozygous inbred line • It takes more generations of backcrossing/selfing than shown here… more like BC 5 S 3. … D R F 1 R BC 2 S 1

Nested Association Mapping (NAM) • Combination of recombinant inbred lines and association mapping • Take 200 diverse founders • Cross each of them to B 73 (reference genome line)

Corn lines • Temperate lines • B 73: The first maize line to be sequenced, reference genome for the species. Released by Iowa State in the 70 s, used in many breeding programs. Also the common parent in NAM. • Tropical lines • CMLs: CIMMYT maize lines. • 282 Diversity panel

Genes and such • Quantitative resistance: resistance to all races of the pathogen, conditioned by many genes • Quantitative trait locus (QTL): Any locus contributing to a quantitative trait • Single Nucleotide Polymorphism (SNP, “snip”) • Point in the genome where a single base varies between individuals (e. g. ATCGCT vs. ATCACT) • Most common genetic marker nowadays • Genotyping-by-sequencing (GBS) • Sequence portions of the genome, then look for SNPs within sequenced regions • Cheap, easy way to get 100 k to millions of SNPs

GWAS • Genome-wide association study (GWAS) • Looking for genetic markers that are associated with variation in a phenotype • For every locus: Test the null hypothesis that “the genotype at this locus has no effect on phenotype” • GWAS-specific lingo: • Manhattan plot: Plot the physical location of all SNPs versus the -log 10(pvalue). A high –log 10(p) → low p-value → highly significant predictor of phenotype • QQ plot: plot the observed p-values for all SNPs vs. the expected p-values. If you account for covariates, they should match up well for 99% of SNPs • Bonferroni correction: Divide the significant p-value cutoff by the number of hypothesis tests performed. Since we perform thousands-millions of tests, a p < 0. 05 cutoff would still give us thousands of false positives. • False discovery rate (FDR)/Benjamini-Hochberg correction: another way to control false positives with many hypothesis tests. Less conservative than the Bonferroni correction.

Diseases we work on • Foliar diseases • Northern leaf blight (NLB): caused by Setosphaeria turcica, greybrown cigar-shaped lesions • Southern leaf blight (SLB) • Grey leaf spot (GLS) • Ear diseases • Fusarium ear rot: caused by Fusarium verticillioides, can lead to accumulation of fumonisin, a carcinogenic mycotoxin • Aspergillus ear rot: caused by Aspergillus flavus, can lead to accumulation of aflatoxin, a carcinogenic mycotoxin
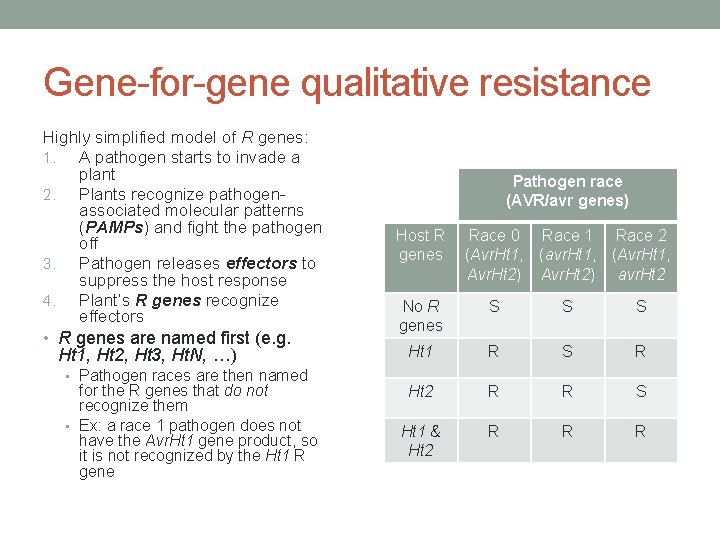
Gene-for-gene qualitative resistance Highly simplified model of R genes: 1. A pathogen starts to invade a plant 2. Plants recognize pathogenassociated molecular patterns (PAMPs) and fight the pathogen off 3. Pathogen releases effectors to suppress the host response 4. Plant’s R genes recognize effectors • R genes are named first (e. g. Ht 1, Ht 2, Ht 3, Ht. N, …) • Pathogen races are then named for the R genes that do not recognize them • Ex: a race 1 pathogen does not have the Avr. Ht 1 gene product, so it is not recognized by the Ht 1 R gene Pathogen race (AVR/avr genes) Host R genes Race 0 Race 1 Race 2 (Avr. Ht 1, (avr. Ht 1, (Avr. Ht 1, Avr. Ht 2) avr. Ht 2 No R genes S S S Ht 1 R S R Ht 2 R R S Ht 1 & Ht 2 R R R

NLB vocab • NY 001 (“New York 1”) • An isolate of S. turcica taken locally • Race 1 (the race native to New York State; not recognized by Ht 1) • Sequence coming soon • 28 A • An isolate of S. turcica taken by Gillian Turgeon’s Lab, selected because it tolerated genetic transformation protocols • Race 23 N (not recognized by Ht 2, Ht 3, Ht. N) • The first S. turcica line sequenced. • Phenotypes • DLA (diseased leaf area): Percentage of leaf covered by lesions, estimated by eye on a weekly basis • IP (incubation period): Number of days between inoculation and first lesion.

Etc. • Internal transcribed spacer (ITS): • Portion of ribosomal RNA sequence that is later excised out • Often sequenced to ID fungal species • Flowering time • Measure of time to maturity • Typically days to anthesis- number of days after planting that 50% of row is shedding pollen

Other experimental terms • ELISA: • RNA-seq: Sequencing of the transcriptome (all the RNA present in the host) to quantify how highly each gene is expressed at certain conditions/times. • Histopathology: Stain different materials in different ways for microscopy. We use this to differentiate fungal/plant material and study pathogenesis.
- Slides: 15