Nelarabine 506 U 78 Compound 506 GW 506

Nelarabine 506 U 78 Compound 506 GW 506 U 78 Nelarabine Arranon Lambe, Cancer Res. 1995

SELECTIVE CYTOTOXICITY OF ara-G T-Cell Specificity Cell Lines Leukemia Cells Associated with ara-GTP

CLINICAL DEVELOPMENT OF ara-G Problems Low solubility Difficulty in synthesis Solution Synthesis of prodrug Nelarabine
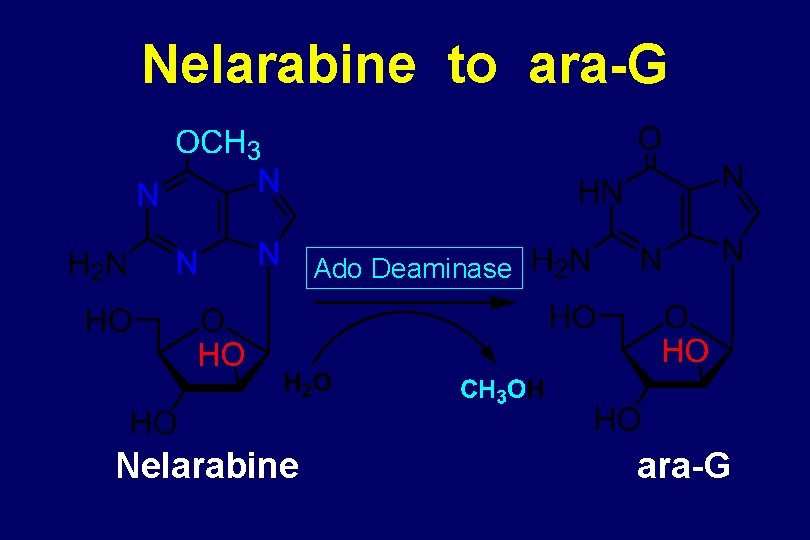
Nelarabine to ara-G Ado Deaminase Nelarabine ara-G

Phase I Study with Nelarabine Dose 5, 10, 20, 40, 60, and 75 mg/kg over 1 hr Frequency Daily x 5 (repeat every 28 days) MTD 30 mg/kg (adult), 40 mg/kg (Ped. ) 1. 2 g/m 2 DLT Neurotoxicity Kurtzberg, JCO 2005

Phase I Trial of Nelarabine –Pediatric and Adult with Hematologic Malignancies Total Patients: 93 Age (<21 yrs: >21 yrs) 34 : 59 Male : Female 66 : 27 Prior Rx: median (Range) 3 (1 -11) Prior Transplant: Auto Allo 15 11 Prior Vincristine 78 Prior high dose ara-C 11 Prior Intrathecal Rx 45
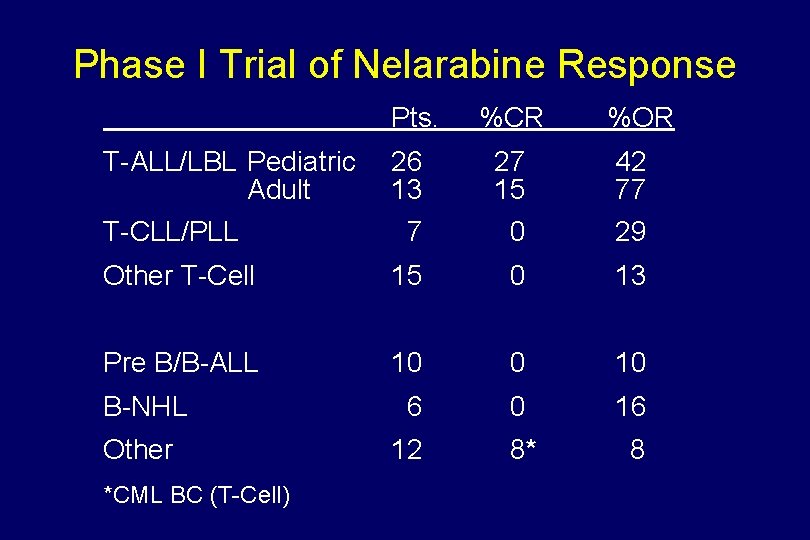
Phase I Trial of Nelarabine Response Pts. T-ALL/LBL Pediatric Adult %CR %OR 26 13 27 15 42 77 7 0 29 Other T-Cell 15 0 13 Pre B/B-ALL 10 0 10 B-NHL 6 0 16 Other 12 8* 8 T-CLL/PLL *CML BC (T-Cell)

Phase I Trial of Nelarabine Neurotoxicity: Overall 50% <21 yrs; 85% >21 yrs Somnolence 3 -8 days, reversible, >21 yrs. Neuropathy Severe in 11%, resolution slow or not Hematologic Grade I-II neutropenia, thrombocytopenia in 50 -70%. No grade III-IV except BM involvement

Phase I Study with Nelarabine Dose Site MDACC 20, 40, and 60 mg/kg over 1 hr per d x 5 Patients n = 27 (2 = ped. ) Diagnoses Hematologic malignancies Gandhi, JCO 1998; Gandhi, JCO 2001
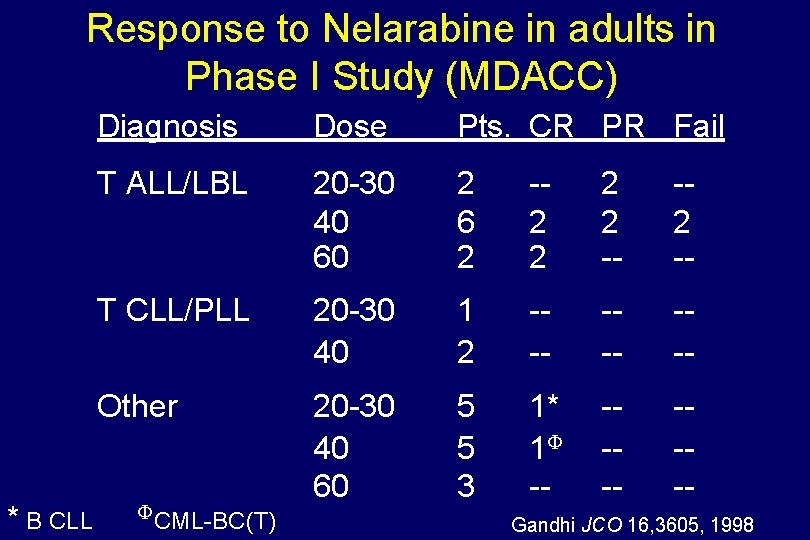
Response to Nelarabine in adults in Phase I Study (MDACC) * B CLL Diagnosis Dose Pts. CR PR Fail T ALL/LBL 20 -30 40 60 2 6 2 -2 2 -- -2 -- T CLL/PLL 20 -30 40 1 2 --- --- Other 20 -30 40 60 5 5 3 1* 1 -- ---- CML-BC(T) Gandhi JCO 16, 3605, 1998

Plasma Pharmacology Nelarabine and ara-G
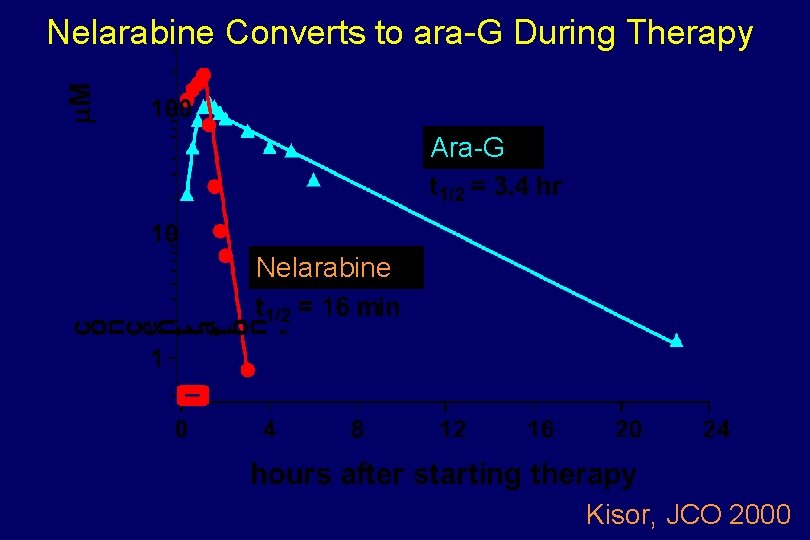
Nelarabine Converts to ara-G During Therapy Ara-G Nelarabine Kisor, JCO 2000

Cellular Pharmacology ara-G triphosphate
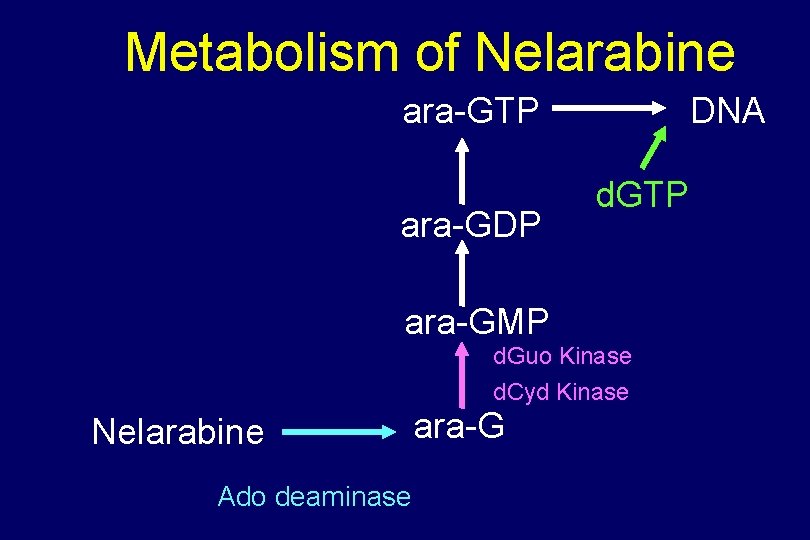
Metabolism of Nelarabine ara-GTP ara-GDP DNA d. GTP ara-GMP d. Guo Kinase d. Cyd Kinase Nelarabine Ado deaminase ara-G
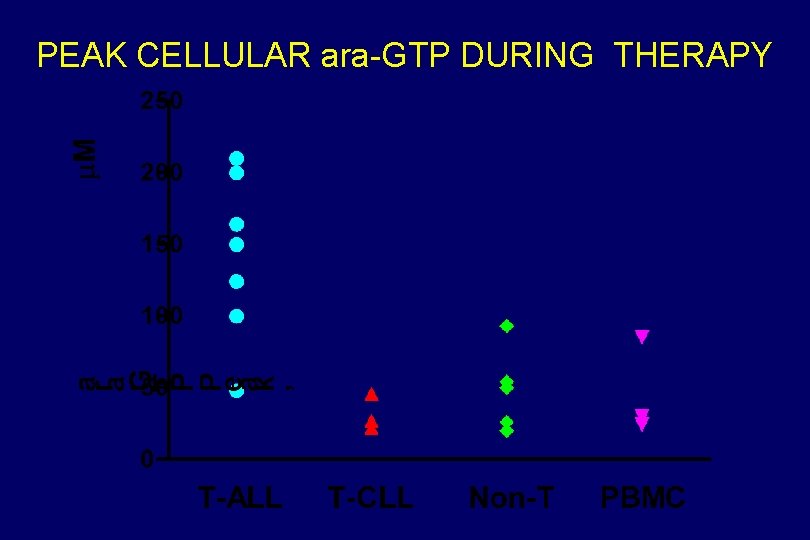
PEAK CELLULAR ara-GTP DURING THERAPY
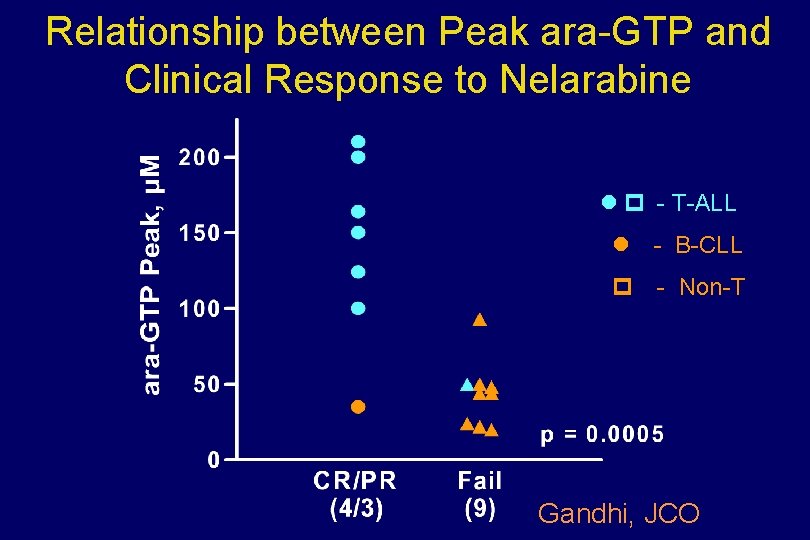
Relationship between Peak ara-GTP and Clinical Response to Nelarabine - T-ALL - B-CLL - Non-T Gandhi, JCO

Summary of Clinical Trial • Nelarabine an effective prodrug • ara-G readily phosphorylated • Prolonged retention of triphosphate • Cellular pharmacokinetics predict clinical response • Active in T-ALL

Regimen Used in Nelarabine for Mature Lymphoid Leukemia Dose Days Patients Nelarabine 20 -60 mg/Kg/day 1 -5 6 Nelarabine 1. 5 -2. 9 G/m 2 1, 3, 5 22 1, 3, 5 9 Nelarabine + 1. 2 G/m 2 Fludarabine 30 mg/m 2

Nelarabine in Mature Lymphoid Leukemia Characteristics Total Patients 37 Male/Female 25 / 12 Age: 64 (27 – 83) Median (range) Prior Rx: Median (range) 2 (0 – 7) Hgb <11 G% 25 (68%) Platelets <100 x 103/ 25 (68%)
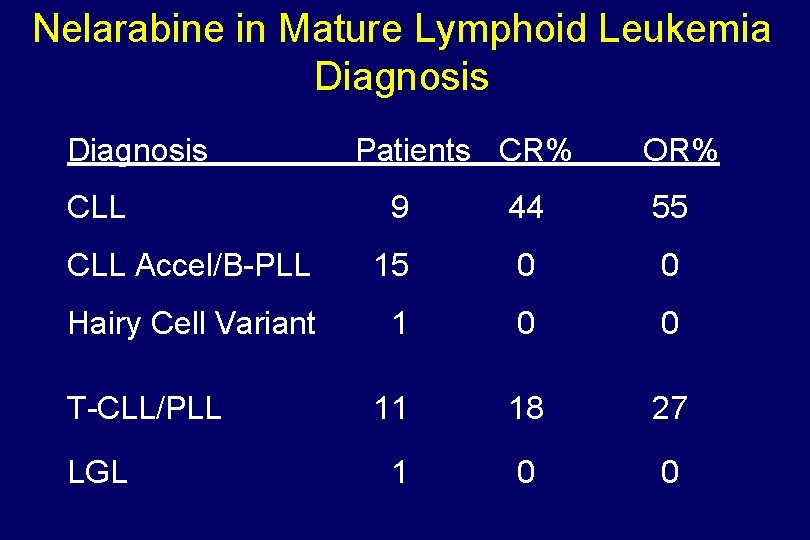
Nelarabine in Mature Lymphoid Leukemia Diagnosis CLL Patients CR% OR% 9 44 55 CLL Accel/B-PLL 15 0 0 Hairy Cell Variant 1 0 0 11 18 27 1 0 0 T-CLL/PLL LGL
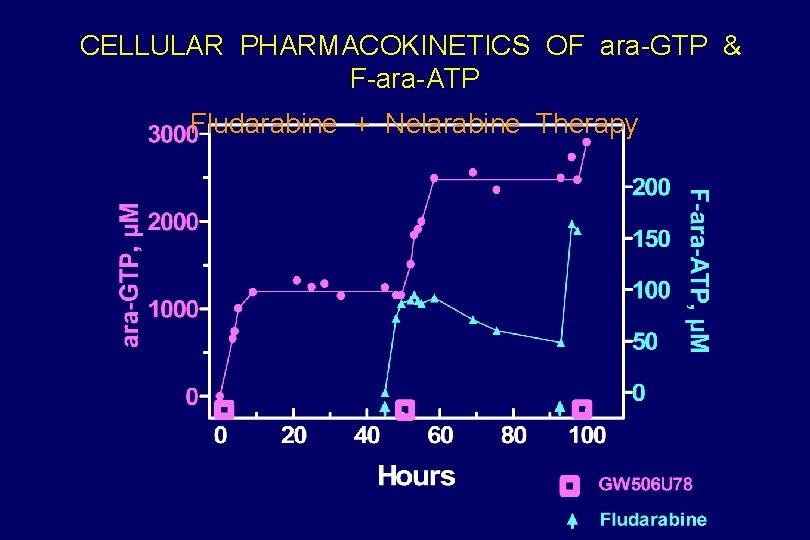
CELLULAR PHARMACOKINETICS OF ara-GTP & F-ara-ATP Fludarabine + Nelarabine Therapy
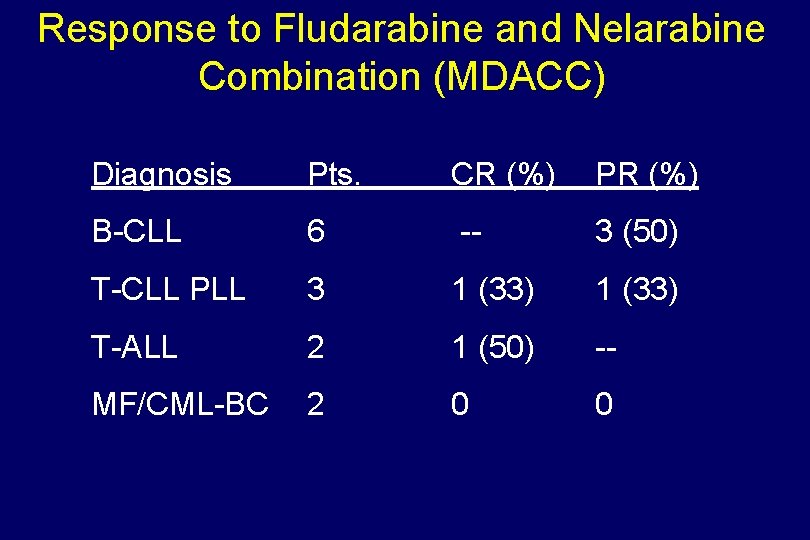
Response to Fludarabine and Nelarabine Combination (MDACC) Diagnosis Pts. CR (%) PR (%) B-CLL 6 -- 3 (50) T-CLL PLL 3 1 (33) T-ALL 2 1 (50) -- MF/CML-BC 2 0 0
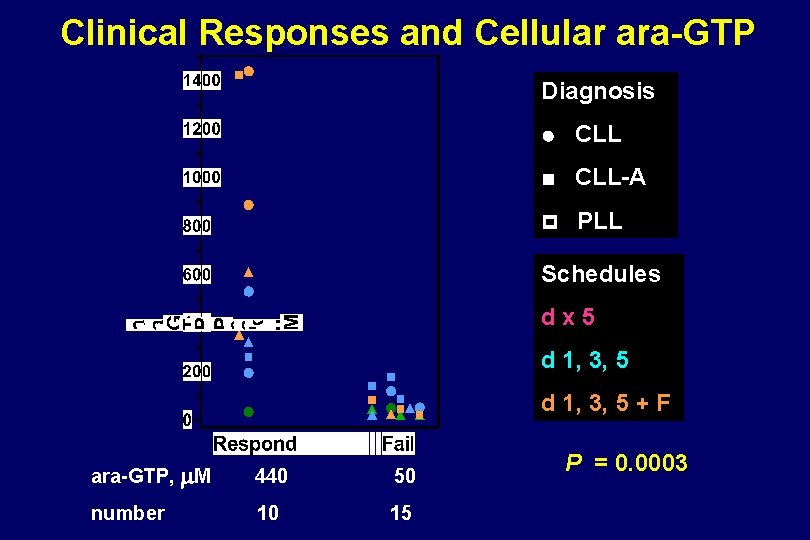
Clinical Responses and Cellular ara-GTP Diagnosis CLL-A PLL Schedules dx 5 d 1, 3, 5 + F ara-GTP, m. M 440 50 number 10 15 P = 0. 0003
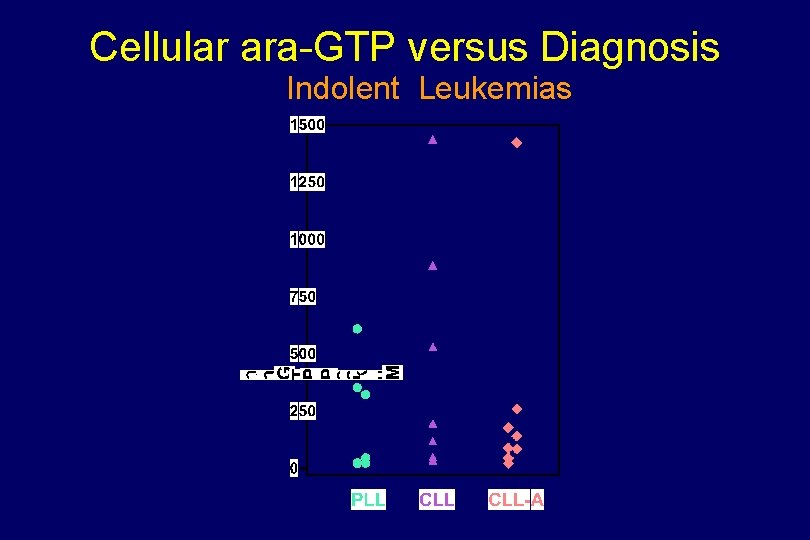
Cellular ara-GTP versus Diagnosis Indolent Leukemias
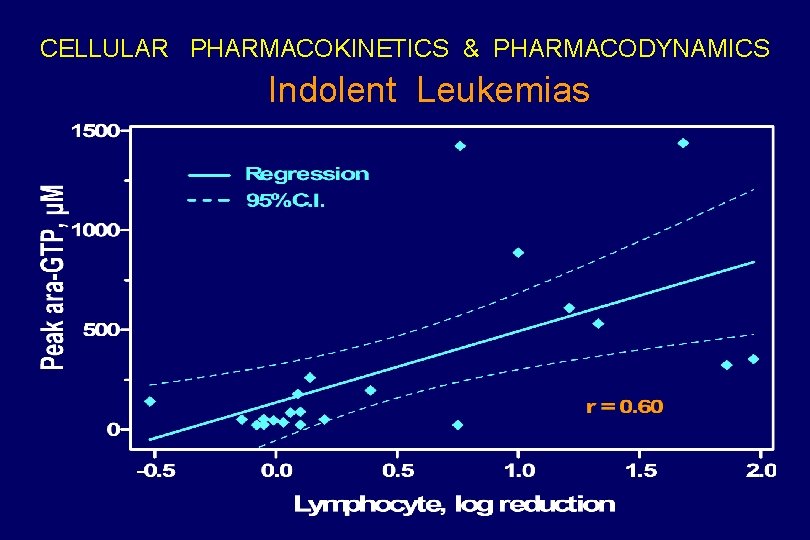
CELLULAR PHARMACOKINETICS & PHARMACODYNAMICS Indolent Leukemias

Phase II Study of Nelarabine in Refractory Hematologic Malignancies (Age <21 yrs) Schedule: 1200 mg/m 2/day Day 1 -5 Reduced to 900 mg/m 2/day then to 650 mg/m 2/day For CNS or extramedullary relapse 400 mg/m 2/day Berg et al, JCO 23; 3376; 2005
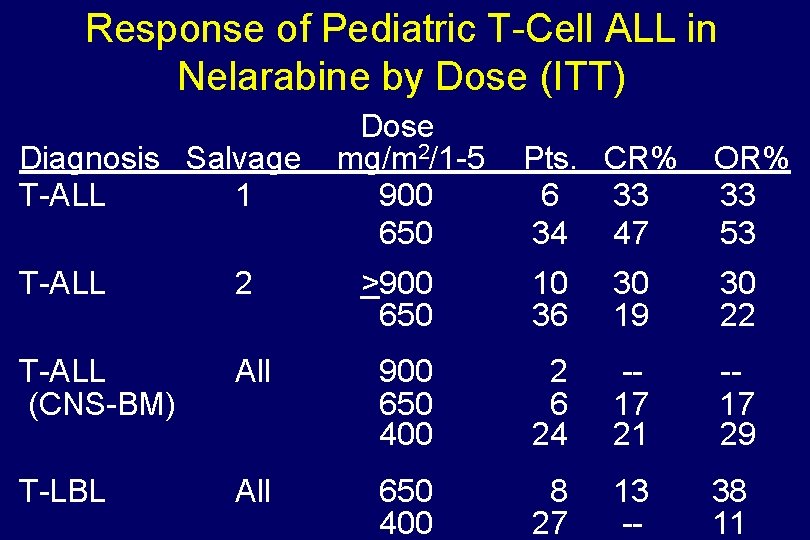
Response of Pediatric T-Cell ALL in Nelarabine by Dose (ITT) Diagnosis Salvage T-ALL 1 Dose mg/m 2/1 -5 900 650 Pts. CR% 6 33 34 47 OR% 33 53 T-ALL 2 >900 650 10 36 30 19 30 22 T-ALL (CNS-BM) All 900 650 400 2 6 24 -17 21 -17 29 T-LBL All 650 400 8 27 13 -- 38 11
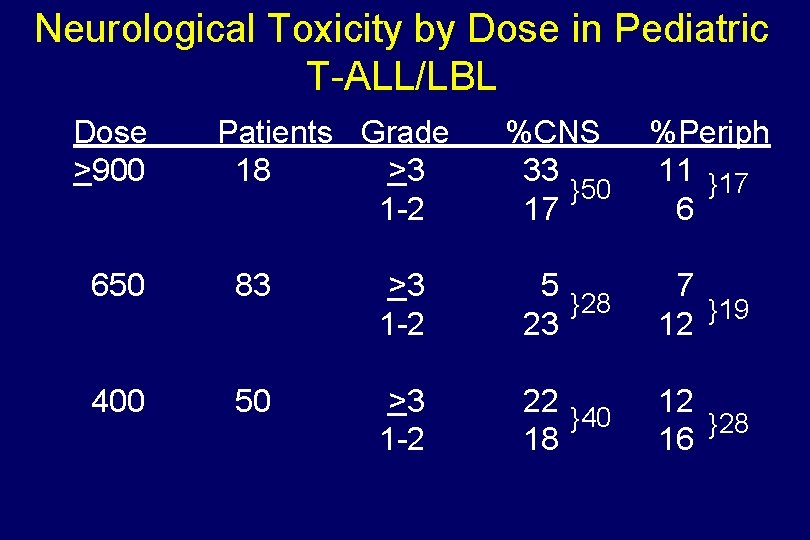
Neurological Toxicity by Dose in Pediatric T-ALL/LBL Dose >900 Patients Grade 18 >3 1 -2 %CNS 33 }50 17 %Periph 11 }17 6 650 83 >3 1 -2 5 }28 23 7 }19 12 400 50 >3 1 -2 22 }40 18 12 }28 16

Nelarabine in T-ALL, T-LBL –Adult (CALGB-SWOG) Total patients 40 T-ALL 22 T-LBL 18 Age (range) 16 – 66 Male/Female 33 / 7 All refractory / Relapses Dose 1. 56/m/day Day 1, 3, 5
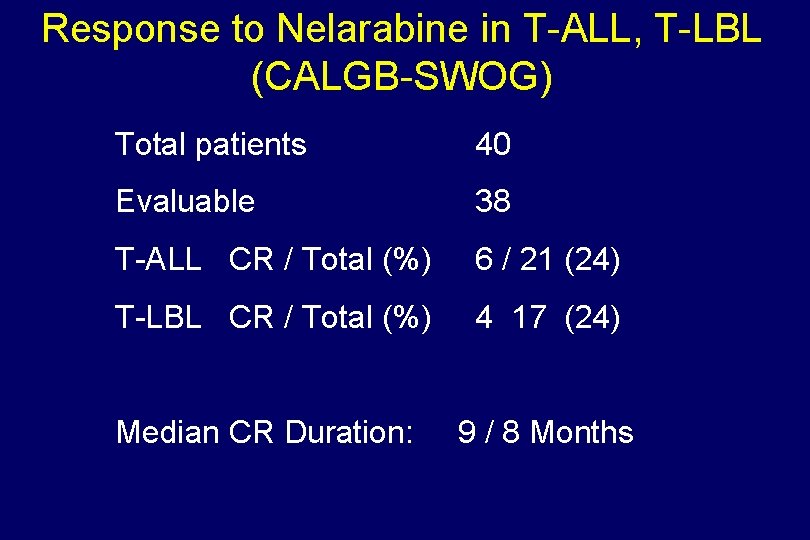
Response to Nelarabine in T-ALL, T-LBL (CALGB-SWOG) Total patients 40 Evaluable 38 T-ALL CR / Total (%) 6 / 21 (24) T-LBL CR / Total (%) 4 17 (24) Median CR Duration: 9 / 8 Months

Conclusions regarding Nelarabine • Active in and approved for T-Cell ALL/LBL • Active in T-Cell CLL/PLL • Active in B-CLL-indolent • Response related to intracellular ara-GTP level • Dose limited by neurotoxicity • Minimal hematologic toxicity • Optimal dose/schedule still to be explored as single agent/combination
- Slides: 31