Neglected Diseases a special case Isabela Ribeiro VI

Neglected Diseases – a special case Isabela Ribeiro VI Pan American Network for Drug Regulatory Harmonization Conference (PANDRH) Strengthening of the National Regulatory Authorities in the context of Health Systems Brasilia, Brazil, 6 to 8 July, 2011
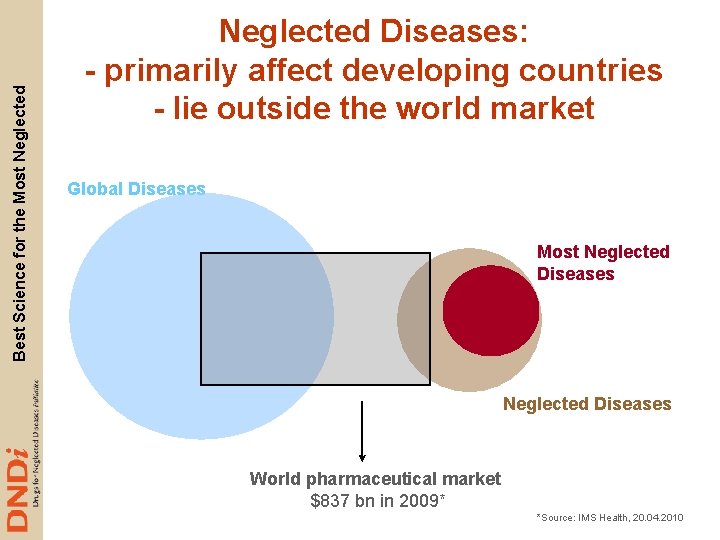
Best Science for the Most Neglected Diseases: - primarily affect developing countries - lie outside the world market Global Diseases Most Neglected Diseases World pharmaceutical market $837 bn in 2009* *Source: IMS Health, 20. 04. 2010
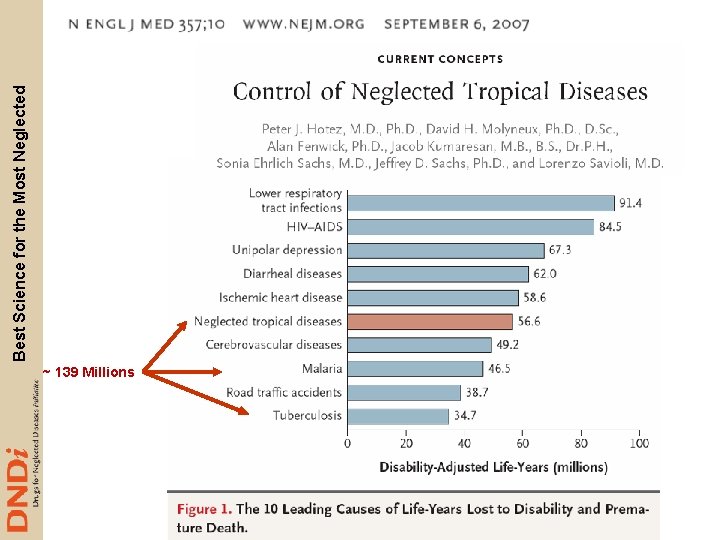
~ 139 Millions Best Science for the Most Neglected

Best Science for the Most Neglected What’s Needed to Combat NTDs? Hotez P, Pecoul B. "Manifesto" for Advancing the Control and Elimination of Neglected Tropical Diseases, PLo. S NTDs, May 2010, Vol 4, 5, e 718
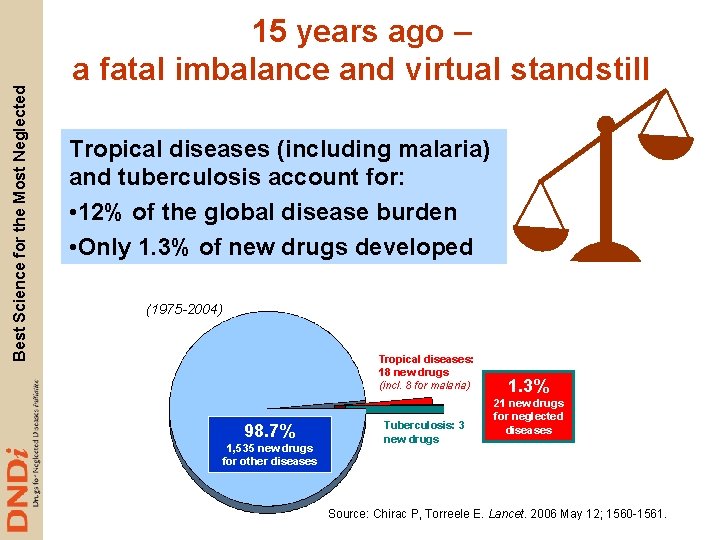
Best Science for the Most Neglected 15 years ago – a fatal imbalance and virtual standstill Tropical diseases (including malaria) and tuberculosis account for: • 12% of the global disease burden • Only 1. 3% of new drugs developed (1975 -2004) Tropical diseases: 18 new drugs (incl. 8 for malaria) 98. 7% 1, 535 new drugs for other diseases Tuberculosis: 3 new drugs 1. 3% 21 new drugs for neglected diseases Source: Chirac P, Torreele E. Lancet. 2006 May 12; 1560 -1561.

Best Science for the Most Neglected Product Development Partnerships (PDPs): Filling the Gaps in Translational Research and Product Development
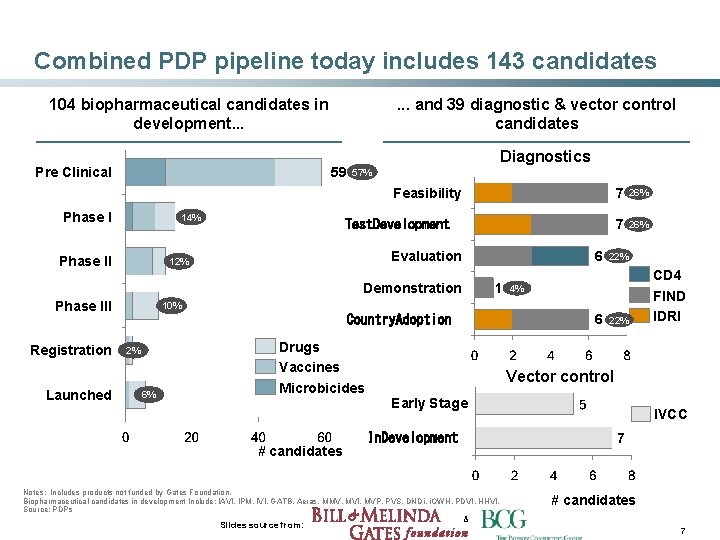
Combined PDP pipeline today includes 143 candidates 104 biopharmaceutical candidates in development. . . Pre Clinical . . . and 39 diagnostic & vector control candidates 59 Diagnostics 57% Feasibility Phase I 14% 15 Phase II Test. Development Evaluation 12% 12 6 Demonstration Phase III Registration Launched 10% 10 2% 2 6% 6 1 Drugs Vaccines Microbicides # candidates 7 26% 22% CD 4 FIND IDRI Vector control Early Stage IVCC In. Development Notes: Includes products not funded by Gates Foundation. Biopharmaceutical candidates in development Include: IAVI, IPM, IVI, GATB, Aeras, MMV, MVI, MVP, PVS, DNDi, i. OWH, PDVI, HHVI. Source: PDPs Slides source from: 26% 4% 6 Country. Adoption 7 # candidates & 7
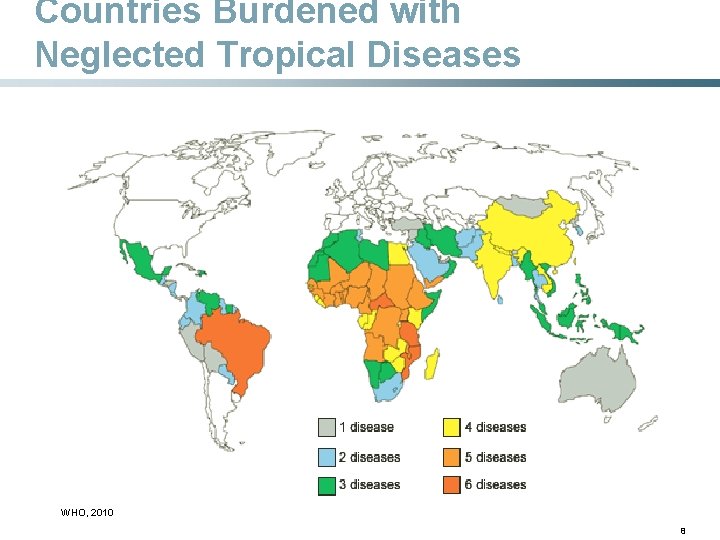
Countries Burdened with Neglected Tropical Diseases WHO, 2010 8
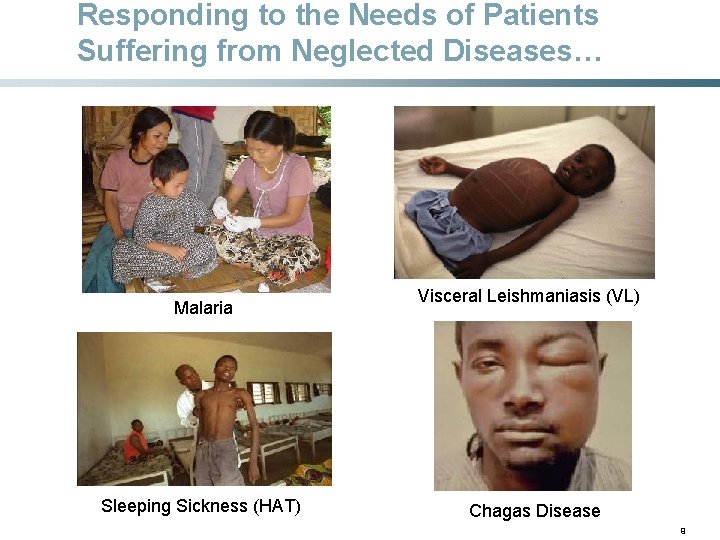
Responding to the Needs of Patients Suffering from Neglected Diseases… Malaria Sleeping Sickness (HAT) Visceral Leishmaniasis (VL) Chagas Disease 9

Human African Trypanosomiasis (HAT) or Sleeping Sickness • 60 million at risk in sub-Saharan Africa • Transmitted by the tsetse fly • Difficult to diagnose; many patients go undiagnosed until late stage of disease • Fatal if untreated • Needs: • A safe, effective, and practical stage 2 treatment • A simple stage 1 treatment 10

Visceral Leishmaniasis (VL) • 200 million at risk worldwide (in 70 countries) • Transmitted by the sandflies • Symptoms: prolonged fever, enlarged spleen & liver, substantial weight of loss, progressive anemia • Fatal if untreated • Current drugs: antimonials, Amphotericin B, Am. Bisome®, miltefosine, paromomycin • Needs: • oral, safe, effective, low-cost and short-course treatment 11

Chagas Disease: A Silent Killer • 100 million at risk in Latin America • Kills more people in region than malaria • Patient number growing in non-endemic, developed countries • Transmitted by ‘kissing bug’, blood transfusion, organ transplantation, as congenitally or orally • Majority of patients undiagnosed until late stage • Needs: • An affordable, age-adapted, safe, and efficacious paediatric strength • A new drug for early chronic stage 12

Among the most neglected… Poorest of the poor Living in remote areas Socioeconomic burden on family and community Marginalised & voiceless patients 13

Neglected Diseases: Current Treatment Limitations • Ineffective (resistance) • Toxic • Expensive • Painful when delivered • Difficult to use • Not registered in endemic regions • Restricted by patents Melarsoprol Eflornithine We Need Safe, Effective, Easy-to-Use Drugs 14

Best Science for the Most Neglected DNDi: An innovative R&D model • Non-profit drug research & development (R&D) organization founded in 2003 • • Addressing the needs of the most neglected patients Harnessing resources from public institutions, private industry and philanthropic entities 7 worldwide offices 7 Founding Partners • Indian Council for Medical Research (ICMR) • Kenya Medical Research Institute (KEMRI) • Malaysian MOH • Oswaldo Cruz Foundation Brazil • Medecins Sans Frontieres (MSF) • Institut Pasteur France • WHO/TDR (permanent observer) Geneva Coordination Team + consultants USA Japan India DRC Malaysia Kenya Brazil

Best Science for the Most Neglected Vision A collaborative, patients’ needs-driven, virtual, non-profit drug R&D organisation to develop new treatments against the most neglected communicable diseases

Best Science for the Most Neglected DNDi’s Main Objectives • Deliver 6 - 8 new treatments by 2014 for sleeping sickness, Chagas disease, leishmaniasis and malaria • Establish a robust pipeline for future needs • Use and strengthen existing capacity in diseaseendemic countries • Raise awareness and advocate for increased public responsibility
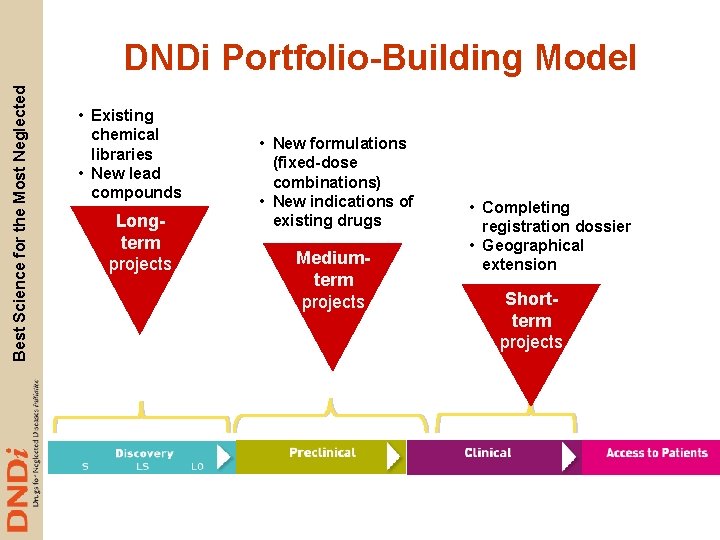
Best Science for the Most Neglected DNDi Portfolio-Building Model • Existing chemical libraries • New lead compounds Longterm projects • New formulations (fixed-dose combinations) • New indications of existing drugs Mediumterm projects • Completing registration dossier • Geographical extension Shortterm projects
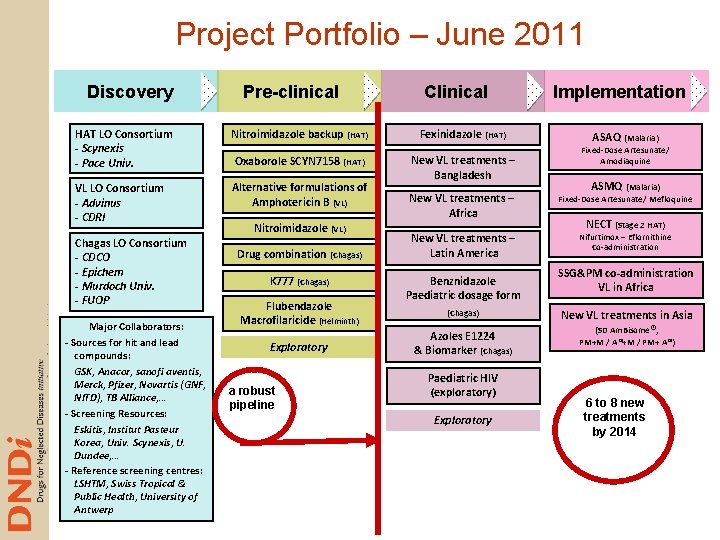
Project Portfolio – June 2011 Discovery Pre-clinical Clinical HAT LO Consortium - Scynexis - Pace Univ. Nitroimidazole backup (HAT) Fexinidazole (HAT) Oxaborole SCYN 7158 (HAT) VL LO Consortium - Advinus - CDRI Alternative formulations of Amphotericin B (VL) New VL treatments – Bangladesh Chagas LO Consortium - CDCO - Epichem - Murdoch Univ. - FUOP Major Collaborators: - Sources for hit and lead compounds: GSK, Anacor, sanofi aventis, Merck, Pfizer, Novartis (GNF, NITD), TB Alliance, … - Screening Resources: Eskitis, Institut Pasteur Korea, Univ. Scynexis, U. Dundee, … - Reference screening centres: LSHTM, Swiss Tropical & Public Health, University of Antwerp Nitroimidazole (VL) Drug combination (Chagas) K 777 (Chagas) Flubendazole Macrofilaricide (Helminth) Exploratory a robust pipeline New VL treatments – Africa New VL treatments – Latin America Benznidazole Paediatric dosage form (Chagas) Azoles E 1224 & Biomarker (Chagas) Paediatric HIV (exploratory) Exploratory Implementation ASAQ (Malaria) Fixed-Dose Artesunate/ Amodiaquine ASMQ (Malaria) Fixed-Dose Artesunate/ Mefloquine NECT (Stage 2 HAT) Nifurtimox – Eflornithine Co-administration SSG&PM co-administration VL in Africa New VL treatments in Asia (SD Am. Bisome®, PM+M / A®+M / PM+ A®) 6 to 8 new treatments by 2014

Best Science for the Most Neglected Five Products Making a Difference 2007 ASAQ (Malaria) Fixed-Dose Artesunate/ Amodiaquine Main Partners sanofi-aventis (France) 2008 ASMQ (Malaria) Fixed-Dose Artesunate/ Mefloquine Farmanguinhos (Brazil) Cipla (India) 2009 NECT Nifurtimox Eflornithine Co-Administration (HAT) 2010 Paramomycin + SSG (VL) National Control Programs MSF / Epicentre Bayer / sanofi-aventis WHO • Affordable • Field-Adapted • Non-Patented LEAP National Programs i. OWH 2011 New VL treatments in Asia (SD Am. Bisome®, PM+M / A®+M / PM+ A®) • Easy to Use National Programs ICMR, i. OWH, TDR
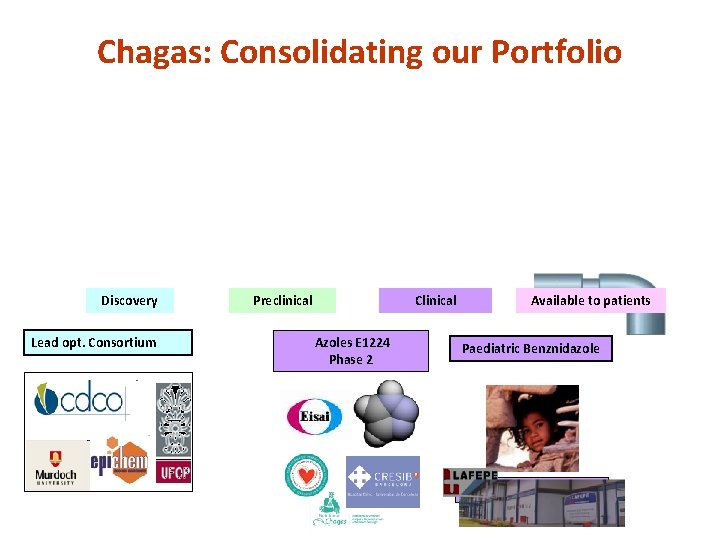
Best Science for the Most Neglected Chagas: Consolidating our Portfolio Discovery Lead opt. Consortium Preclinical Clinical Azoles E 1224 Phase 2 Available to patients Paediatric Benznidazole Lafepe

Best Science for the Most Neglected Chagas Azoles E 1224 • License signed with the Japanese pharma Eisai for clinical development of Ravuconazole for treatment of Chagas disease funded by DNDi (September 29, 2009) – E 1224, pro-drug of ravuconazole, an anti-fungal drug discovered by Eisai – Implementation of Phase II clinical trial in adult patients with chronic indeterminate Chagas disease (Bolivia, 2 sites in 2011) • Study initiation in June 2011

Best Science for the Most Neglected Chagas Biomarkers in Chagas • PCR study: optimization of sampling procedures in chronic Chagas Disease (n=220) – Collaboration with MSF Spain, Bolivia Mission (MSFOCBA) and UMSS – Study initiation – April 13 th (102 patients recruited) • RT-PCR lab optimization and validation for clinical studies – Collaboration with UMSS, CEADES, CONICET • NHEPACHA network for long term evaluation of candidate biomarkers • Coordination of activities with different partners – Work towards the integration of data on candidate markers – Participation in PAHO/TDR PCR meeting in B. Aires – Collaboration with Univ. of Georgia and TBRI– nonhuman primate study

Chagas Pediatric Benznidazole Best Science for the Most Neglected A pediatric formulation available in 2011 Benznidazole registered by Roche in 1971, licensed to Brazilian government in 2003 Objective: An affordable, age-adapted, easy to use, pediatric formulation for Chagas disease Current ways to administer in children • 100 mg tablet fractionated or macerated for administration • High risk of delivering improper dosages • 40 -160% of Target BZ content Target: 12. 5 mg tablets for <20 kg children DNDi-Lafepe agreement in 2008 for development of pediatric formulation

Best Science for the Most Neglected Chagas Clinical Research Platform Objectives: • Facilitate effective and efficient trials to deliver improved treatment for Chagas disease • Strengthen institutional research capacity • Support an environment conducive to quality research • Develop a critical mass of expertise • Define priority areas for clinical evaluation of new treatments in Chagas disease • Conduct periodic review and update of Target Product Profile in Chagas Disease • Articulate with other initiatives

7 -Year Results • • 2 new malaria treatments 1 new sleeping sickness combination 1 new visceral leishmaniasis combination for Africa 1 new visceral leishmaniasis combination for Asia Largest pipeline ever for the kinetoplastid diseases Clinical research platforms in Africa and Latin America On track to deliver new treatments per business plan
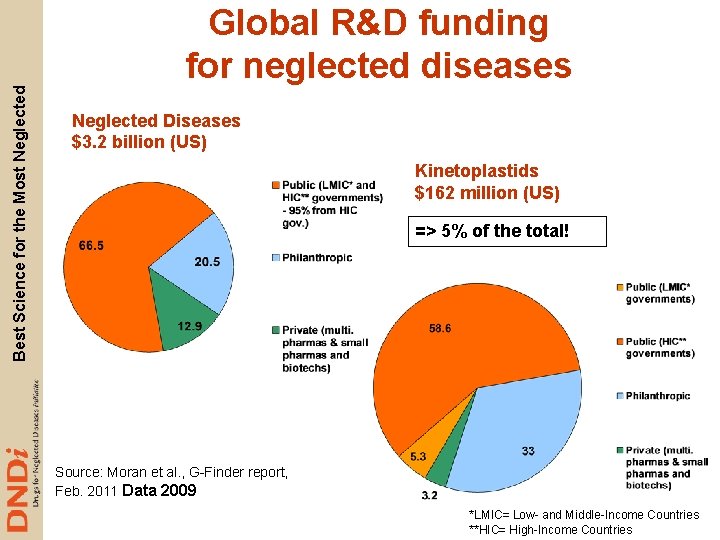
Best Science for the Most Neglected Global R&D funding for neglected diseases Neglected Diseases $3. 2 billion (US) Kinetoplastids $162 million (US) => 5% of the total! Source: Moran et al. , G-Finder report, Feb. 2011 Data 2009 *LMIC= Low- and Middle-Income Countries **HIC= High-Income Countries
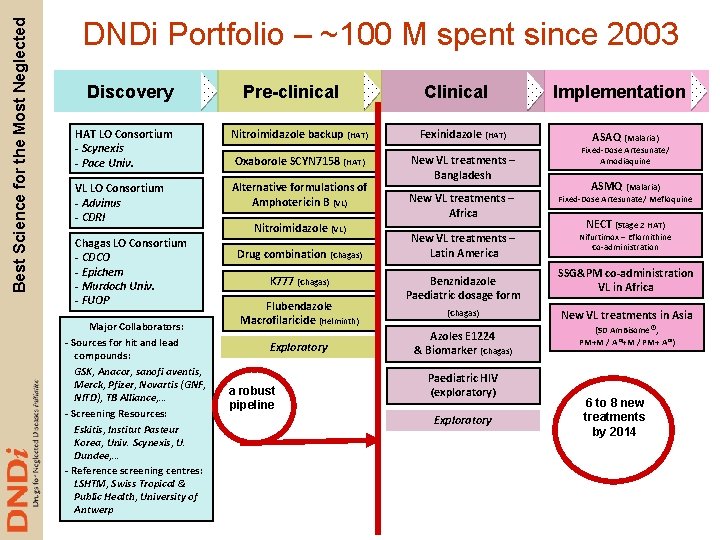
Best Science for the Most Neglected DNDi Portfolio – ~100 M spent since 2003 Discovery Pre-clinical Clinical HAT LO Consortium - Scynexis - Pace Univ. Nitroimidazole backup (HAT) Fexinidazole (HAT) Oxaborole SCYN 7158 (HAT) VL LO Consortium - Advinus - CDRI Alternative formulations of Amphotericin B (VL) New VL treatments – Bangladesh Chagas LO Consortium - CDCO - Epichem - Murdoch Univ. - FUOP Major Collaborators: - Sources for hit and lead compounds: GSK, Anacor, sanofi aventis, Merck, Pfizer, Novartis (GNF, NITD), TB Alliance, … - Screening Resources: Eskitis, Institut Pasteur Korea, Univ. Scynexis, U. Dundee, … - Reference screening centres: LSHTM, Swiss Tropical & Public Health, University of Antwerp Nitroimidazole (VL) Drug combination (Chagas) K 777 (Chagas) Flubendazole Macrofilaricide (Helminth) Exploratory a robust pipeline New VL treatments – Africa New VL treatments – Latin America Benznidazole Paediatric dosage form (Chagas) Azoles E 1224 & Biomarker (Chagas) Paediatric HIV (exploratory) Exploratory Implementation ASAQ (Malaria) Fixed-Dose Artesunate/ Amodiaquine ASMQ (Malaria) Fixed-Dose Artesunate/ Mefloquine NECT (Stage 2 HAT) Nifurtimox – Eflornithine Co-administration SSG&PM co-administration VL in Africa New VL treatments in Asia (SD Am. Bisome®, PM+M / A®+M / PM+ A®) 6 to 8 new treatments by 2014
Best Science for the Most Neglected PDP costs will likely increase in the next years Risks in research and development of pharmaceutical drugs (Preziosi, P. 2004, Nature Reviews in Drug Discovery 3, 521 -526)

Best Science for the Most Neglected Access • Need to facilitate equitable access to new treatments • Unmet medical needs == often non-existing procurement and distribution mechanisms and lack of traditional commercial partners • Important to full transition, in long term, new treatments to natural implementers, i. e. , Ministry of Health, National Control Programmes, WHO, NGOs Access strategies: pragmatic and focused on most pressing “actionable” access barriers

Towards a global public health & equitable access framework for R&D WHO Global Strategy on Public Health, Innovation and Intellectual Property Incentives and financing mechanisms tailored to particular stages of R&D, types of diseases and health technologies are necessary to address existing gaps and the unmet needs of neglected patients

Best Science for the Most Neglected Increasing resources for neglected disease R&D • Investigate innovative sustainable funding for product development and access • Pilot milestone prizes to stimulate discovery of new clinical candidates Reducing the costs of R&D for neglected diseases • IP management, open innovation and sharing of knowledge • Accessing compounds and annotated data • Negotiating freedom to operate, paving the way for access • Pushing for transparency and sharing of knowledge • Innovative regulatory pathways to expedite access in endemic regions and strengthening local regulatory capacity

Best Science for the Most Neglected Regulatory • Historically, most new drugs for neglected diseases have been submitted to regulatory authorities such as the U. S. Food and Drug Administration, the European Medicines Agency, or Swiss. Medic, either for routine regulatory review or under specific pathways such as orphan drug legislation or expedited approval mechanisms This regulatory process has drawbacks: • Potential for delays in access, if there is a two-step process with agencies waiting for regulatory decisions in the North before commencing action • It puts neglected disease product decisions in the hands of regulators who have less experience in tropical disease products, presentations, and epidemiology, and who are not accountable for the needs and safety of target patients

Best Science for the Most Neglected Current Regulatory Mechanisms New Drugs for Neglected Diseases Standard regulatory review by stringent authorities • Routine regulatory review • Orphan drug designation and review process • Expedited review Neglected disease-specific review by stringent authorities • European Union: Article 58 • US FDA: “Tentative approval” • WHO drug prequalification

Best Science for the Most Neglected Regulatory Review by Non-Endemic Countries • Decades of regulatory experience to bear on assessment of the neglected disease product but • Delayed access for target patients • Inability to provide clear guidance on the clinical trial design and the data required for marketing approval • Lack of sufficient safety and efficacy data requirements for wider use • Inappropriate data requirements • Inappropriate risk-benefit assessment for wider use

Best Science for the Most Neglected “Registering New Drugs: The African Context” DNDi-commissioned report, by the George Institute for International Health. Experts involved in this study expressed that African regulators have a crucial role to play in assessing health tools being used to respond to specific patient needs in their countries. Key recommendations to strengthen regulatory authorisation processes in Africa for new drugs against neglected diseases: • Ensure closer collaboration between developing and developed countries by involving regulators of endemic countries in regulatory assessments of new drugs for neglected diseases; • Extend WHO’s role in the prequalification process of new tools against neglected tropical diseases, in addition to HIV/ AIDS, malaria, and tuberculosis; and • Strengthen regulatory capacity in Africa through the creation of Regional Centres of Excellence in each of Africa’s main sub-regions.

Best Science for the Most Neglected Regulatory Context – Latin America - Unique opportunity of the PARF - Pan American initiative for harmonisation of regulatory requirements - Identification of Reference National Authorities - Prioritization of diseases with public health impact and affecting neglected populations - Key examples in the region where not for profit development and access on neglected diseases could be facilitated by innovative review process

Best Science for the Most Neglected Benznidazole - Product developed by Roche and registered in Brazil and Argentina in the 70’s - Licensed to LAFEPE, second largest public laboratory in Brazil - Following technology transfer, product registered in Brazil in 2006 - Current status: - 17 of 21 countries: Bz is listed among first line treatment for Chagas disease - 6 of 21 countries include it in the Essential Medicines List - 6 of 21 countries product is currently registered - Well-recognised bottleneck in product availability in the countries: difficulties in importation process and distribution issues - Adult and new pediatric presentation would benefit from concerted review and action from regulatory authorities in the Latin American region

Best Science for the Most Neglected R&D of new candidate drugs for Chagas - Challenges of the assessment of therapeutic response in Chagas disease has hampered drug development for many years - No available early test of cure - Several meetings indicate that current marker of choice for assessment of therapeutic failures in Chagas: PCR - Regional mechanisms for consultation and agreement with regulatory authorities on R&D development strategy and definition on necessary steps for registration of new products

By working together in a creative way, PDPs, large and small pharma, and the public sector can bring innovation to neglected patients! Thank you! www. dndi. org
- Slides: 40