NEEP 541 Interatomic Potentials Fall 2003 Jake Blanchard

NEEP 541 – Interatomic Potentials Fall 2003 Jake Blanchard

Outline n Interatomic Potentials n n n Impact Parameter Potentials Closest Approach

Binary Collisions n Energetic particle strikes stationary particle V 1 f 1 M 1 V 10 M 2 V 2 f 2

CM System M 1 u 1 F 1 u 10 M 1 2 u 20 u 2 F M 2
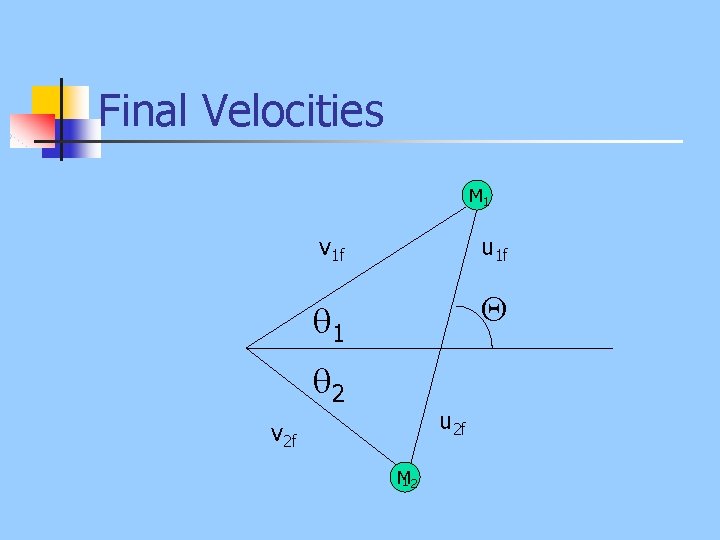
Final Velocities M 1 v 1 f u 1 f 1 2 u 2 f v 2 f M 12

Results
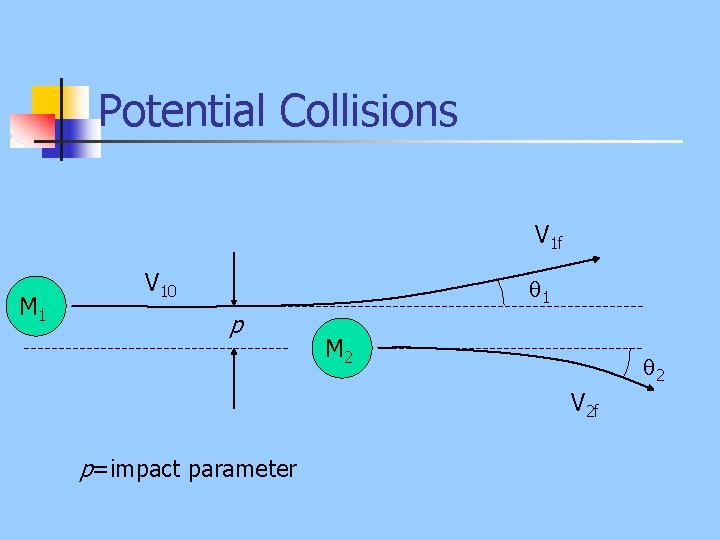
Potential Collisions V 1 f M 1 V 10 1 p M 2 2 V 2 f p=impact parameter
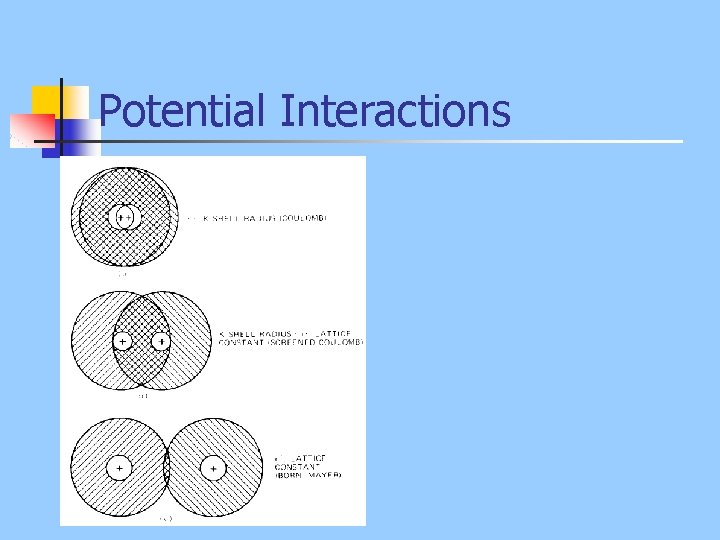
Potential Interactions

Potentials n n Overlap of electrons leads to repulsion Mechanisms: n n Electrostatic repulsion due to charge imbalance Closed shell repulsion resulting from Pauli exclusion principle
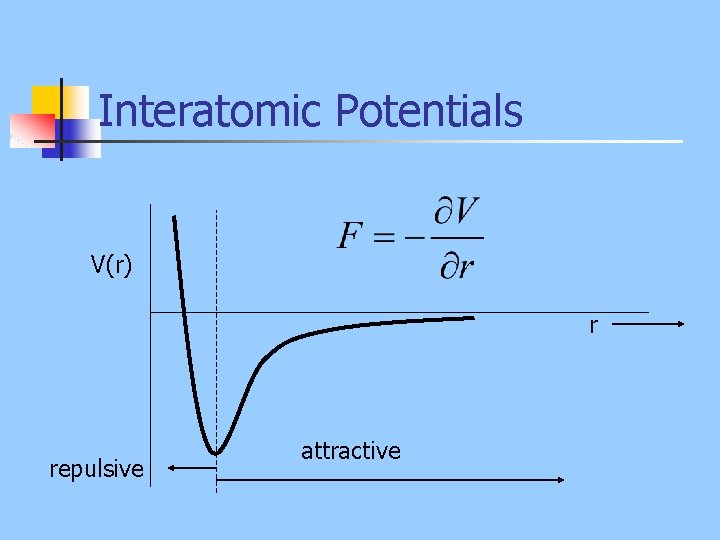
Interatomic Potentials V(r) r repulsive attractive

Functions n Large separation: Born-Mayer n Very close: Coulomb n Medium range: modified Coulomb

Screening Radius

Hard Sphere “billiard ball”

Screened Coulomb Ignores closed shell repulsion

Power Law

Thomas-Fermi Moliere

Using Potentials n n n n Most scattering occurs near closest approach Hence, one can use single potential Distance of closest approach determines “best” potential Low E: Born-Mayer Medium E: Inverse square High E: Coulomb Best overall is Thomas-Fermi
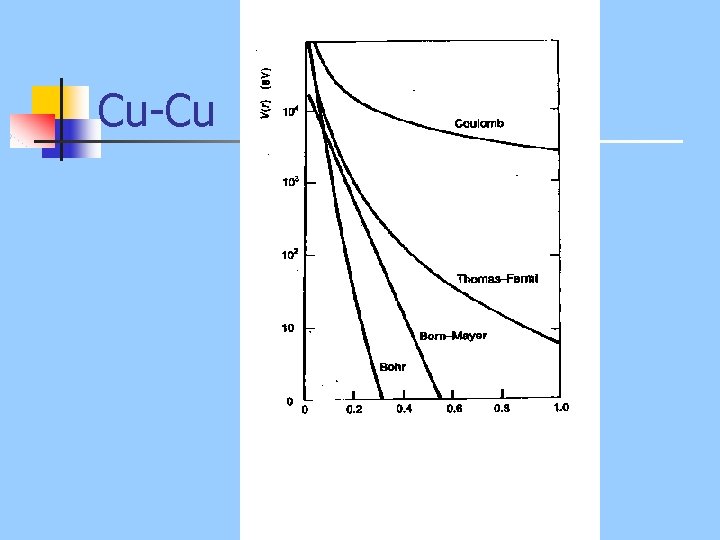
Cu-Cu

Collisions n Distance of Closest Approach M 1 V 10 M 2 V 20

Distance of Closest Approach

Distance of Closest Approach • At closest approach, Er=0 (g=0) • = distance of closest approach • 0 = distance of closest approach for p=0

Example – Coulomb
- Slides: 22