NCI Pediatric Oncology Branch Clinical Trials New Targets

NCI Pediatric Oncology Branch Clinical Trials New Targets and Agents Brigitte Widemann, M. D. June 19, 2017

NIH Clinical Center Largest Research Hospital in USA Mark O. Hatfield Clinical Research Center Every patient at the NIH Clinical Center is evaluated for a clinical trial

The POB Goal: Improve the survival and quality of life of children and young adults with refractory or high risk tumors v Diverse basic, translational, clinical investigators: • Basic understanding of disease pathogenesis • Clinical Trials Program for tumors with unmet need: • Solid tumors, brain tumors, leukemias • Rare tumors, genetic tumor predisposition syndromes • Psychosocial and behavioral research v Optimal use of Intramural Research Program resources • Freedom to pursue basic and clinical questions • Research that requires long term commitment • Rare diseases, longitudinal studies, novel trial designs • Rapid clinical translation of laboratory observations
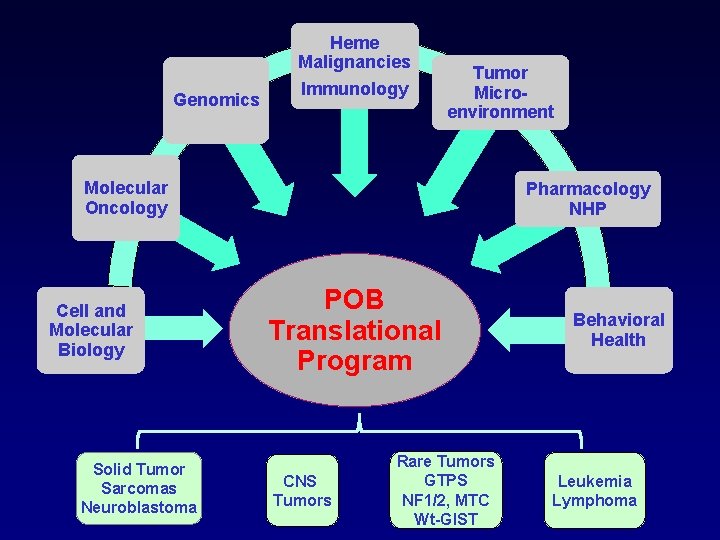
Heme Malignancies Genomics Immunology Tumor Microenvironment Molecular Oncology Cell and Molecular Biology Solid Tumor Sarcomas Neuroblastoma Pharmacology NHP POB Translational Program CNS Tumors Rare Tumors GTPS NF 1/2, MTC Wt-GIST Behavioral Health Leukemia Lymphoma

POB Trials: Genetic Tumor Predisposition Agent Mechanism Phas e Eligibility Status PI Selumetinib MEK II NF 1 PN ≥ 18 y/o NF 1 GIST ≥ 3 y/o NF 1 c. NF ≥ 18 y/o Ongoing Soon Widemann Vistusertib+ Selumetinib m. TOR MEK II MPNST ≥ 18 y/o Soon Kim Widemann Cabozantinib MET, VEGFR, KIT, RET II COG ≥ 2 y/o Ewing, RMS, non RMS STS, Wilms, rare tumors HCC, RCC, MTC… Open Akshintala Widemann Guadecitabine (SGI 110) DNA methyltransferase II ≥ 12 y/o SDH-deficient tumors: GIST, Pheo/PG, HLRCC Open Glod Longitudinal / Natural History Studies: NF 1, MTC, Rare Tumors

POB Trials: Solid Tumors-Leukemias Agent Mechanism Phase Eligibility Status PI NY-ESO II ≥ 4 y/o Synovial sarcoma Open Glod Ganitumab Dasatinib IGF 1 R YES I/II RMS Ability to swallow tablets Open Heske Atezolizumab PD-L 1 PLX 3397 CSF 1 KIT I/II Solid tumors / leukemias Open Kaplan PLX 51107+ Irinotecan Bromodomain Topo I I/II Solid tumors / leukemias Soon Shern Anti-CD 22 CAR CD 22 !/II CD 22 positive leukemia Open Fry/Shah Bispecific CAR CD 19 and CD 22 I/II CD 19 and CD 22 positive leukemia / lymphoma Soon Fry/Shah ≥ 6 y/o ASPS Chen Children’s Oncology Group Phase I Consortium: Pediatric Match
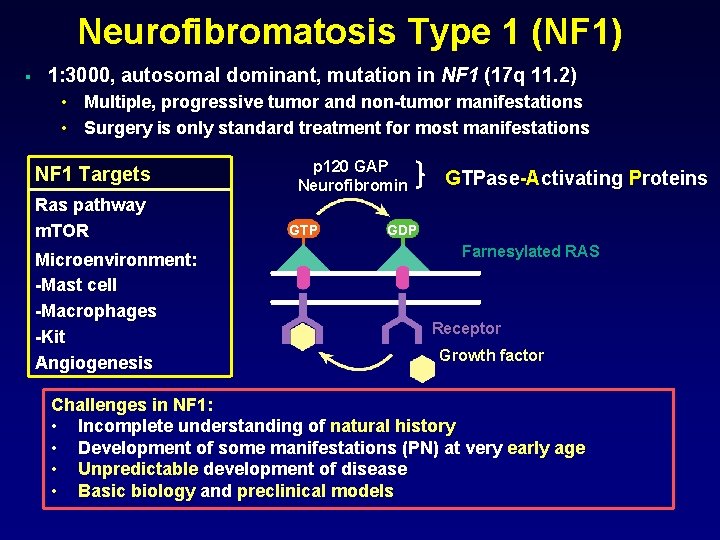
Neurofibromatosis Type 1 (NF 1) § 1: 3000, autosomal dominant, mutation in NF 1 (17 q 11. 2) • Multiple, progressive tumor and non-tumor manifestations • Surgery is only standard treatment for most manifestations NF 1 Targets Ras pathway m. TOR Microenvironment: -Mast cell -Macrophages -Kit Angiogenesis p 120 GAP Neurofibromin GTPase-Activating Proteins GDP Farnesylated RAS Receptor Growth factor Challenges in NF 1: • Incomplete understanding of natural history • Development of some manifestations (PN) at very early age • Unpredictable development of disease • Basic biology and preclinical models
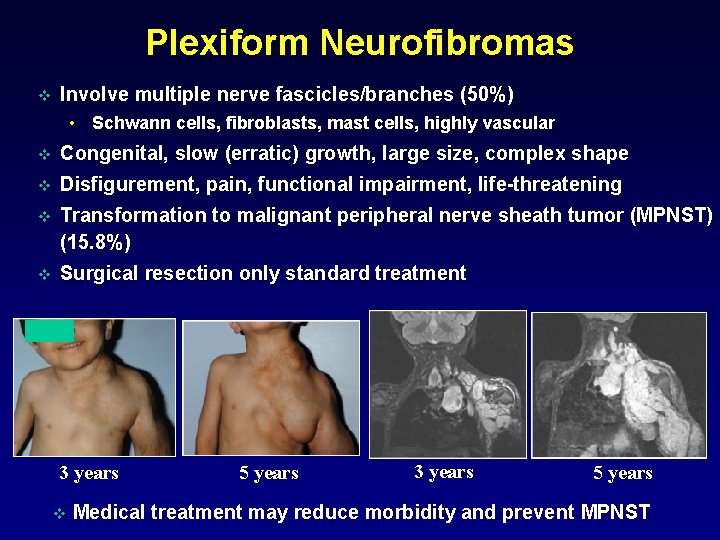
Plexiform Neurofibromas v Involve multiple nerve fascicles/branches (50%) • Schwann cells, fibroblasts, mast cells, highly vascular v Congenital, slow (erratic) growth, large size, complex shape v Disfigurement, pain, functional impairment, life-threatening v Transformation to malignant peripheral nerve sheath tumor (MPNST) (15. 8%) v Surgical resection only standard treatment 3 years v 5 years 3 years 5 years Medical treatment may reduce morbidity and prevent MPNST
Volumetric MRI analysis
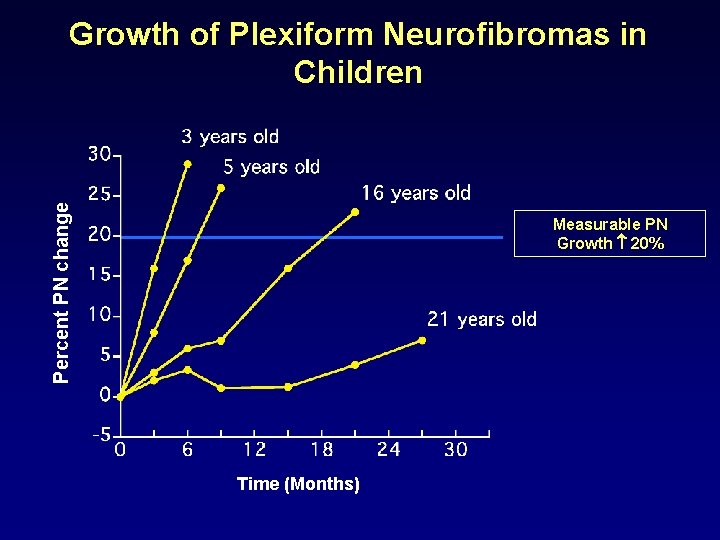
Percent PN change Growth of Plexiform Neurofibromas in Children Measurable PN Growth 20% Time (Months)
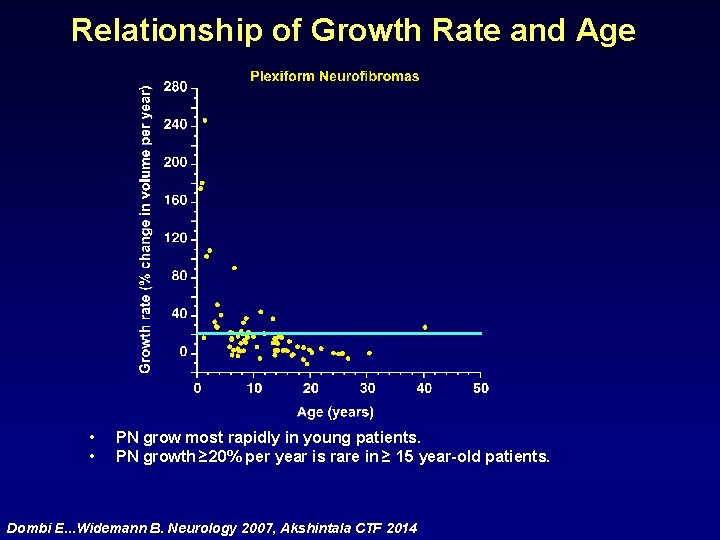
Relationship of Growth Rate and Age • • PN grow most rapidly in young patients. PN growth ≥ 20% per year is rare in ≥ 15 year-old patients. Dombi E. . . Widemann B. Neurology 2007, Akshintala CTF 2014
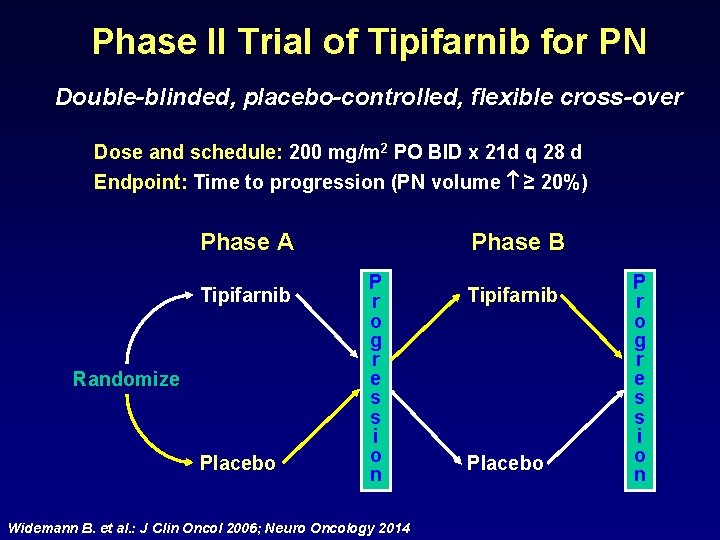
Phase II Trial of Tipifarnib for PN Double-blinded, placebo-controlled, flexible cross-over Dose and schedule: 200 mg/m 2 PO BID x 21 d q 28 d Endpoint: Time to progression (PN volume ≥ 20%) Phase A Tipifarnib Randomize Placebo Phase B P r o g r e s s i o n Widemann B. et al. : J Clin Oncol 2006; Neuro Oncology 2014 Tipifarnib Placebo P r o g r e s s i o n
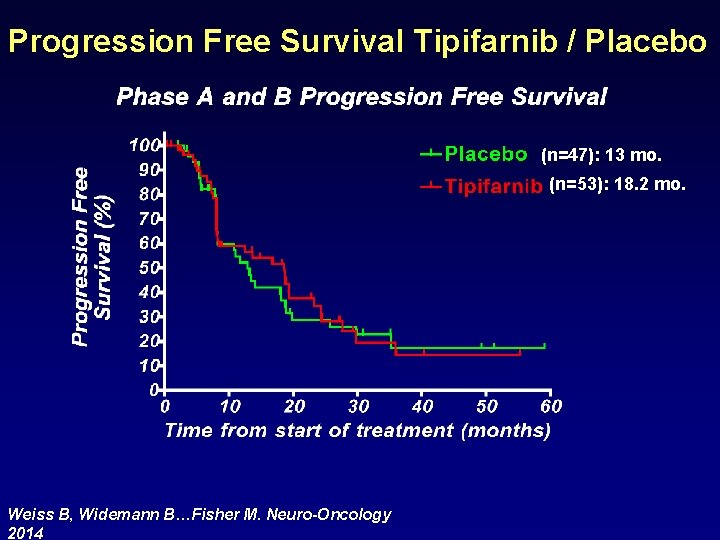
Progression Free Survival Tipifarnib / Placebo (n=47): 13 mo. (n=53): 18. 2 mo. Weiss B, Widemann B…Fisher M. Neuro-Oncology 2014
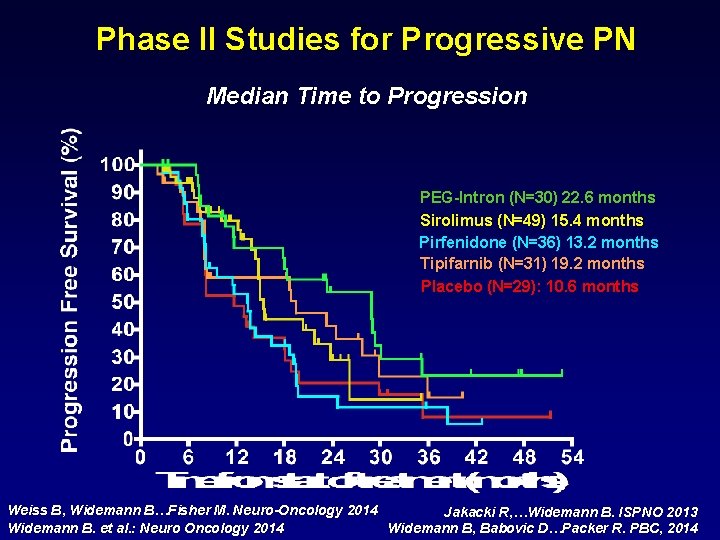
Phase II Studies for Progressive PN Median Time to Progression PEG-Intron (N=30) 22. 6 months Sirolimus (N=49) 15. 4 months Pirfenidone (N=36) 13. 2 months Tipifarnib (N=31) 19. 2 months Placebo (N=29): 10. 6 months Weiss B, Widemann B…Fisher M. Neuro-Oncology 2014 Jakacki R, …Widemann B. ISPNO 2013 Widemann B. et al. : Neuro Oncology 2014 Widemann B, Babovic D…Packer R. PBC, 2014

Phase II Trials for Progressive PN Agent N Age PN TTP PN decrease (Years) (m. L) (mo) N ≥ 20% Max % Placebo 29 8 (3 -18) 316 (40 -4896) 10. 6 0 7 Tipifarnib 31 10 (3 -22) 572 (21 -5573) 19. 2 0 11 Pirfenidone 36 9 (3 -19 349 (12 -5629) 13. 2 0 12 Pegintron 30 7 (1. 6 -18) 288 (14 -3102 22. 6 2 (7%) 27 Sirolimus 49 8 (3 -45. ) 186 (13 -4808) 15. 4 0 17

Selumetinib (AZD 6244) Phase 1 in NF 1 PN Specific MEK inhibitor Objectives: v v v MTD: Cycles 1 -3 (1 cycle = 28 days) Pharmacokinetics 3 D MRI of PN Eligibility: v Children 3 -18 y/o with NF 1 and inoperable PN Selumetinib administration: v BID (10 mg, 25 mg capsules) continuous dosing v Dose level 1: 20, DL 2: 30, DL 1. 5: 25 mg/m 2/dose Response evaluation (3 D): v Post cycle 5, 10, then every 6 cycles v PR = ≥ 20% decrease in volume Dombi E…Widemann B. . . NEJM 2016 RAS RAF MEK ERK

Selumetinib (AZD 6244) Phase 1 in NF 1 PN Primary objectives achieved v 24 patients: median age 10. 9 years (3 -18 years) v Median PN volume: 1205 m. L (29 -7, 210 m. L) v 19 patients: 1 -6 prior medical PN therapies v 11 patients: 1 -6 prior PN debulking surgeries v MTD: 25 mg/m 2/dose (60% of adult MTD) v DLTs: CK , infection, rash, LVEF , all reversible v Most frequent AEs: Acneiform rash, CK , GI Patient 1 Abdominal Plexiform Neurofibroma Baseline: 2449 ml Cycle 5: 2038 ml - 17% Cycle 22: 1477 ml - 40%

Selumetinib Decreases PN Volumes v v Confirmed partial response: 17 of 24 patients (71%) Continue on study: 19 of 24 patients after median of 30 (23 -56 cycles) Subject ID Best response (cycle #) 1 2 16 14 12 13 10 15 17 3 11 18 20 22 19 24 21 23 46 46 28 22 28 28 10 22 22 10 10 5 4 9 7 5 8 6 16 22 22 22 10 16 28 16 5 5 10 5
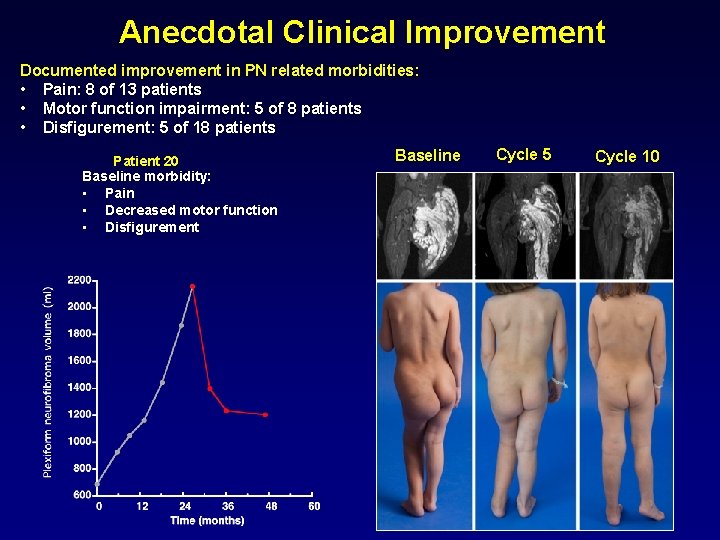
Anecdotal Clinical Improvement Documented improvement in PN related morbidities: • Pain: 8 of 13 patients • Motor function impairment: 5 of 8 patients • Disfigurement: 5 of 18 patients Patient 20 Baseline morbidity: • Pain • Decreased motor function • Disfigurement Baseline Cycle 5 Cycle 10

PN Phase II Trials of Selumetinib Eligibility Pediatric Registration Study Adult Study Age (years) 2 -18 >18 Primary objective Confirmed response rate by volumetric MRI Secondary objectives PRO: Pain, QOL, function All patients Disfigurement All patients with visible PN: Photography / video Function All patients based on PN location: Orbit, airway, motor, bowel, bladder, other PK, PD: Cytokine, PBMC All patients pre selumetinib and on treatment Study specific Target response rate / pts. -Long term safety -Bone mineral density -Optic glioma 36%, 50 patients (stratum 1) Paired biopsy: PN, DNF p. ERK, p. AKT, p. MEK, kinome, transcriptome, PDX, immune infiltrate 45% , max 35 patients (two-stage design) Pediatric study has completed enrollment in stratum 1 within less than 1 year
Whole Body MRI of NF 1 PN Examples of Tumor Burden
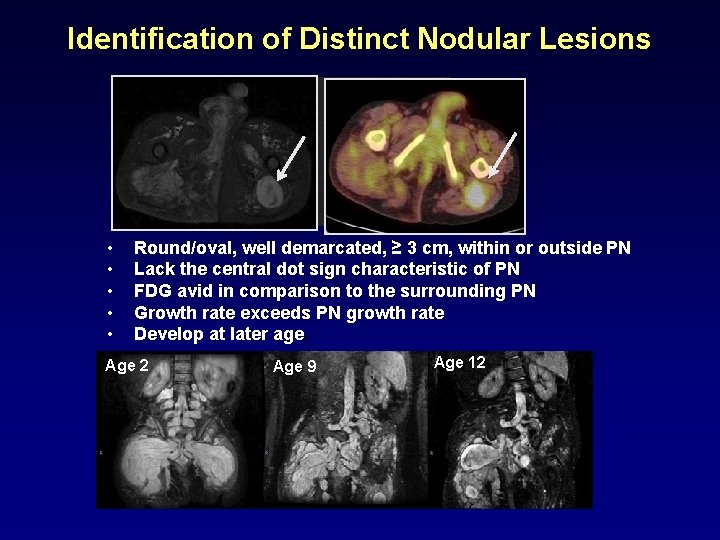
Identification of Distinct Nodular Lesions • • • Round/oval, well demarcated, ≥ 3 cm, within or outside PN Lack the central dot sign characteristic of PN FDG avid in comparison to the surrounding PN Growth rate exceeds PN growth rate Develop at later age Age 2 Age 9 Age 12
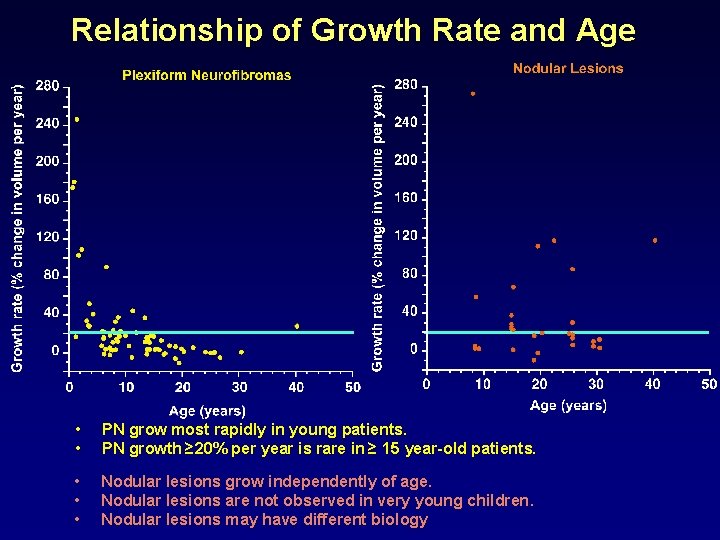
Relationship of Growth Rate and Age • • PN grow most rapidly in young patients. PN growth ≥ 20% per year is rare in ≥ 15 year-old patients. • • • Nodular lesions grow independently of age. Nodular lesions are not observed in very young children. Nodular lesions may have different biology
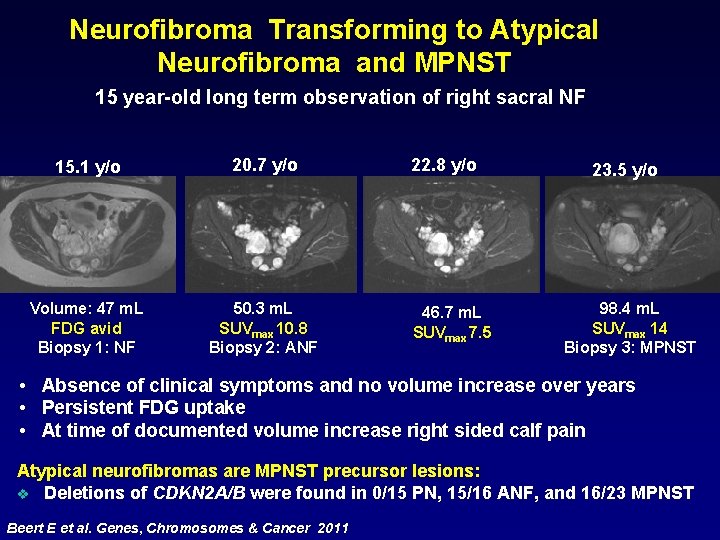
Neurofibroma Transforming to Atypical Neurofibroma and MPNST 15 year-old long term observation of right sacral NF • • • 15. 1 y/o 20. 7 y/o Volume: 47 m. L FDG avid Biopsy 1: NF 50. 3 m. L SUVmax 10. 8 Biopsy 2: ANF 22. 8 y/o 46. 7 m. L SUVmax 7. 5 23. 5 y/o 98. 4 m. L SUVmax 14 Biopsy 3: MPNST Absence of clinical symptoms and no volume increase over years Persistent FDG uptake At time of documented volume increase right sided calf pain Atypical neurofibromas are MPNST precursor lesions: v Deletions of CDKN 2 A/B were found in 0/15 PN, 15/16 ANF, and 16/23 MPNST Beert E et al. Genes, Chromosomes & Cancer 2011
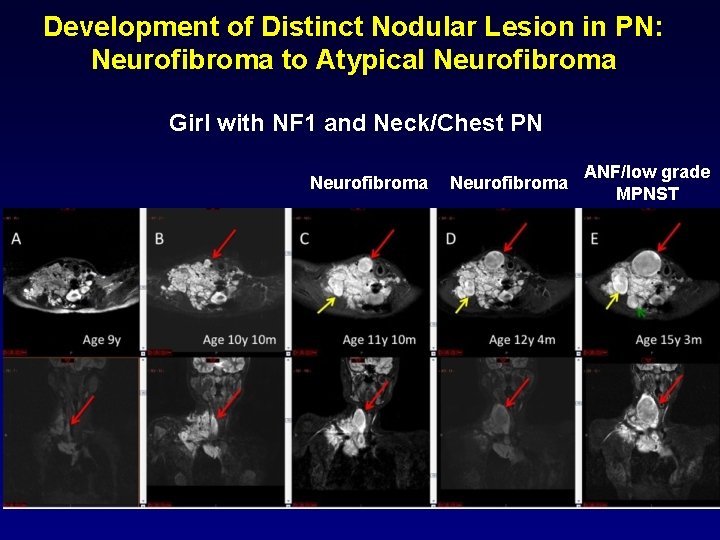
Development of Distinct Nodular Lesion in PN: Neurofibroma to Atypical Neurofibroma Girl with NF 1 and Neck/Chest PN Neurofibroma ANF/low grade MPNST
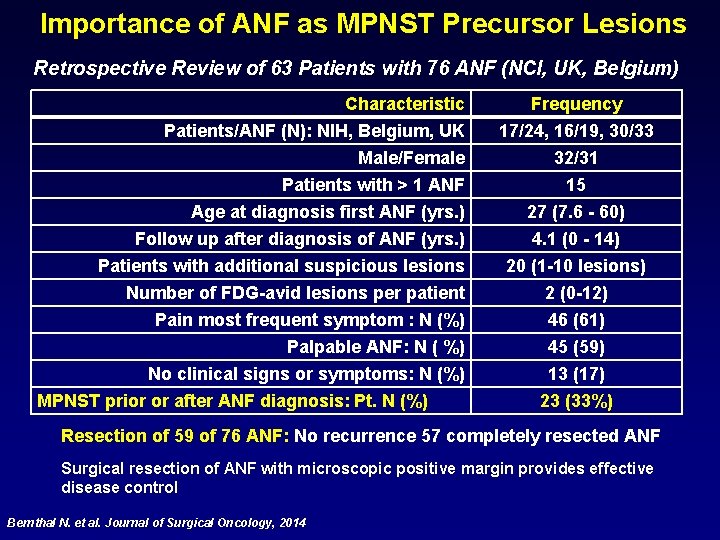
Importance of ANF as MPNST Precursor Lesions Retrospective Review of 63 Patients with 76 ANF (NCI, UK, Belgium) Characteristic Patients/ANF (N): NIH, Belgium, UK Male/Female Patients with > 1 ANF Age at diagnosis first ANF (yrs. ) Follow up after diagnosis of ANF (yrs. ) Patients with additional suspicious lesions Number of FDG-avid lesions per patient Pain most frequent symptom : N (%) Palpable ANF: N ( %) No clinical signs or symptoms: N (%) MPNST prior or after ANF diagnosis: Pt. N (%) Frequency 17/24, 16/19, 30/33 32/31 15 27 (7. 6 - 60) 4. 1 (0 - 14) 20 (1 -10 lesions) 2 (0 -12) 46 (61) 45 (59) 13 (17) 23 (33%) Resection of 59 of 76 ANF: No recurrence 57 completely resected ANF Surgical resection of ANF with microscopic positive margin provides effective disease control Bernthal N. et al. Journal of Surgical Oncology, 2014
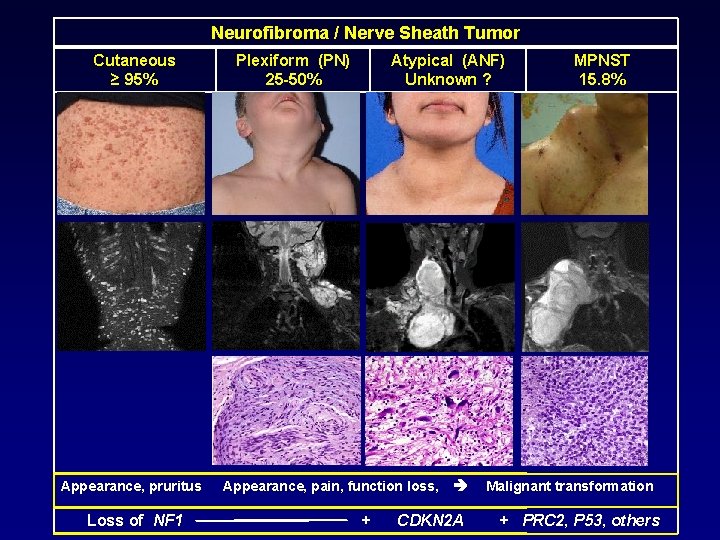
Neurofibroma / Nerve Sheath Tumor Cutaneous ≥ 95% Appearance, pruritus Loss of NF 1 Plexiform (PN) 25 -50% Atypical (ANF) Unknown ? Appearance, pain, function loss, + CDKN 2 A MPNST 15. 8% Malignant transformation + PRC 2, P 53, others
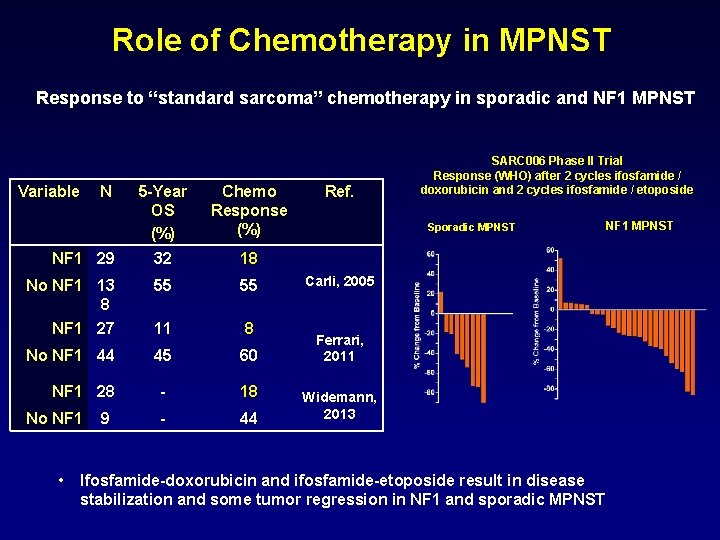
Role of Chemotherapy in MPNST Response to “standard sarcoma” chemotherapy in sporadic and NF 1 MPNST Variable N 5 -Year OS (%) 32 Chemo Response (%) No NF 1 13 8 NF 1 27 55 55 11 8 No NF 1 44 45 60 - 18 - 44 NF 1 29 NF 1 28 No NF 1 • 9 Ref. SARC 006 Phase II Trial Response (WHO) after 2 cycles ifosfamide / doxorubicin and 2 cycles ifosfamide / etoposide Sporadic MPNST 18 Carli, 2005 Ferrari, 2011 Widemann, 2013 Ifosfamide-doxorubicin and ifosfamide-etoposide result in disease stabilization and some tumor regression in NF 1 and sporadic MPNST NF 1 MPNST
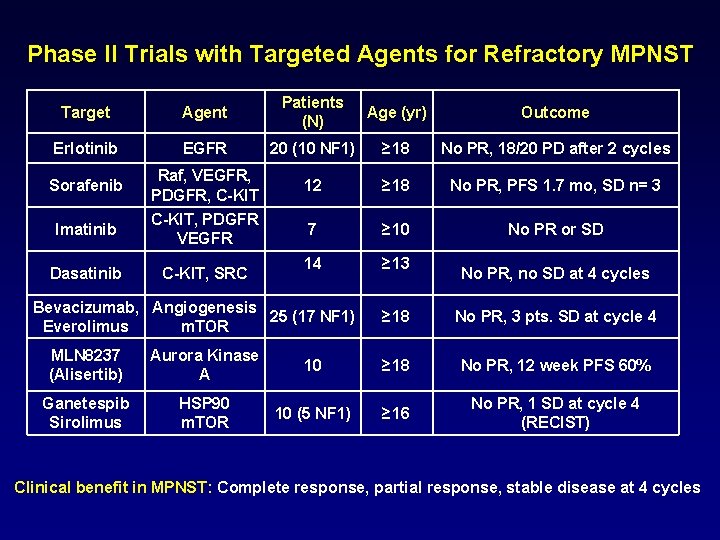
Phase II Trials with Targeted Agents for Refractory MPNST Target Agent Patients (N) Age (yr) Outcome Erlotinib EGFR 20 (10 NF 1) ≥ 18 No PR, 18/20 PD after 2 cycles Sorafenib Raf, VEGFR, PDGFR, C-KIT 12 ≥ 18 No PR, PFS 1. 7 mo, SD n= 3 Imatinib C-KIT, PDGFR VEGFR 7 ≥ 10 No PR or SD Dasatinib C-KIT, SRC 14 ≥ 13 Bevacizumab, Angiogenesis 25 (17 NF 1) Everolimus m. TOR No PR, no SD at 4 cycles ≥ 18 No PR, 3 pts. SD at cycle 4 MLN 8237 (Alisertib) Aurora Kinase A 10 ≥ 18 No PR, 12 week PFS 60% Ganetespib Sirolimus HSP 90 m. TOR 10 (5 NF 1) ≥ 16 No PR, 1 SD at cycle 4 (RECIST) Clinical benefit in MPNST: Complete response, partial response, stable disease at 4 cycles
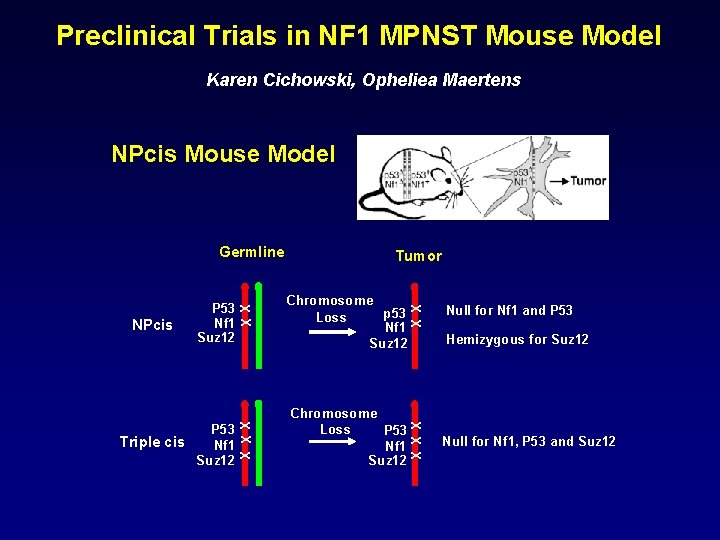
Preclinical Trials in NF 1 MPNST Mouse Model Karen Cichowski, Opheliea Maertens NPcis Mouse Model Germline Chromosome p 53 Loss Nf 1 Suz 12 X X Chromosome Loss P 53 Nf 1 Suz 12 X X X P 53 Nf 1 Suz 12 XX X Triple cis XX NPcis P 53 Nf 1 Suz 12 Tumor Null for Nf 1 and P 53 Hemizygous for Suz 12 Null for Nf 1, P 53 and Suz 12

SARC 031: A Phase II trial of selumetinib in combination with the dual m. TOR inhibitor vistusertib for recurrent or refractory MPNST Principal Investigator: Ae. Rang Kim, MD, Ph. D, Children’s National Medical Center Co-Principal Investigator: Brigitte Widemann, MD, National Cancer Institute SARC Sponsored Do. D Funded Astra. Zeneca: Selumetinib and vistusertib (AZD 2014)
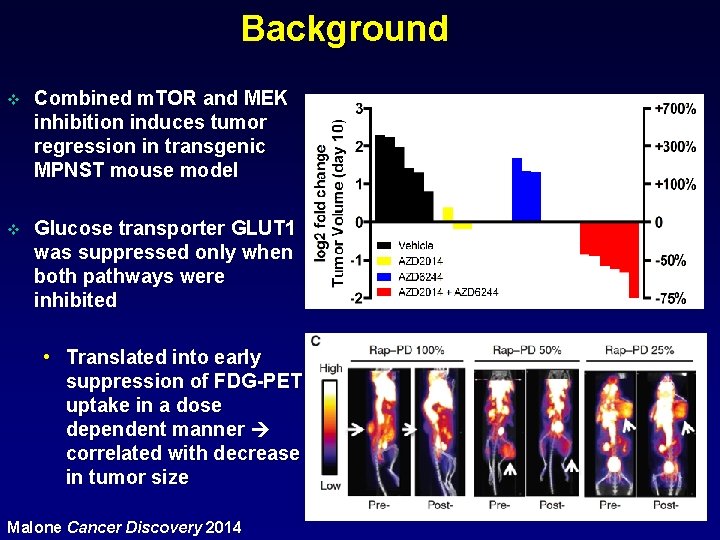
Background v Combined m. TOR and MEK inhibition induces tumor regression in transgenic MPNST mouse model v Glucose transporter GLUT 1 was suppressed only when both pathways were inhibited • Translated into early suppression of FDG-PET uptake in a dose dependent manner correlated with decrease in tumor size Malone Cancer Discovery 2014

Study Overview v v v Primary objective: • Objective response rate Secondary objectives: • Explore early FDG-PET changes • Changes in PD markers: p. ERK, p. S 6, GLUT 1 in blood and tumor Germline and tumor genomic alterations • Assess impact on pain and quality of life Eligibility: • Measurable refractory, unresectable, or metastatic histologically confirmed NF 1 associated or sporadic MPNST • Age ≥ 18 years old • Normal organ function Treatment plan: One cycle = 28 days • Selumetinib 75 mg BID (1 cycle =28 days) • Vistusertib 35 mg PO BID 2 days on, 5 days off Design: • Two-stage • Primary endpoint clinical benefit using RECIST (CR/PR/SD ≥ 4 cycles) • Target response 30% • 1 st stage 7 patients, • If ≥ 1 response, expand enrollment in 2 nd stage to 22 patients

Conclusions Plexiform neurofibromas: • Congenital, most rapid growth in young children • Responsive to MEK inhibition Distinct nodular lesions: • Develop after early childhood • Distinct nodular appearance, growth rate independent of age, FDGavid • May be less responsive to MEK inhibition Atypical neurofibromas: • CDKN 2 A/B deletion • Growth of DNL, FDG-avidity, and pain should raise concern for ANF and evaluation • Diagnosis of MPNST is more frequent in patients with ANF • ANF are MPNST precursors • Surgical removal of ANF may reduce risk for malignant transformation MPNST: • Additional genetic changes: PRC 2, EED, SUZ 12, p 53 • Early diagnosis is critical to improve outcome • Novel medical treatment strategies are needed

Hereditary Medullary Thyroid Carcinoma (MTC) v v Rare calcitonin producing tumor (hereditary 25 -30%) Mutation in RET proto-oncogene Age of onset MEN 2 B: Children Most common cause of death in individuals with MEN Vandetanib: v Multi-targeted oral RTK inhibitor: • RET, VEGFR 2, EGFR, PDGFR, C-KIT v Activity in adults with advanced hereditary MTC: • Partial response 20%, stable disease ≥ 24 weeks 30% • FDA approval for advanced MTC in adults (300 mg/dose once daily) Pediatric phase I/II trial of vandetanib: v Objectives: Safety, tolerability, activity (RECIST) , phase II dose v Results: v Recommended dose: 100 mg/m 2/dose v Partial response rate in MEN 2 B 47% Wells S, et al. , J Clin Oncol 2010 Fox E, Widemann B…Balis F, CCR 2013
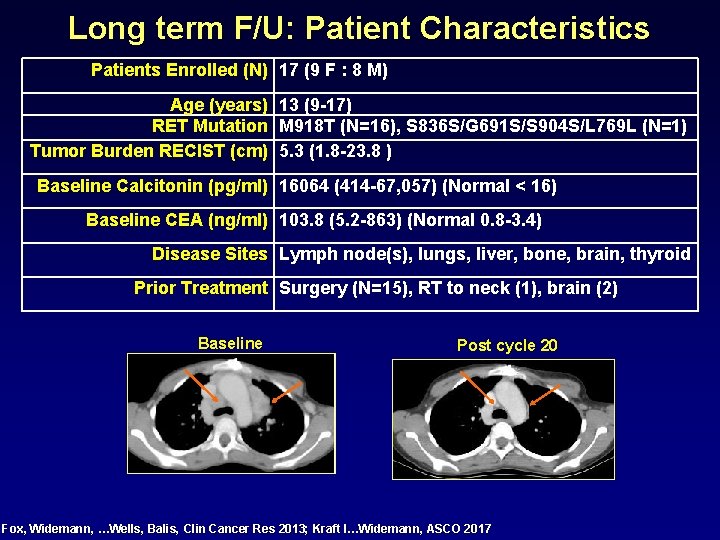
Long term F/U: Patient Characteristics Patients Enrolled (N) 17 (9 F : 8 M) Age (years) 13 (9 -17) RET Mutation M 918 T (N=16), S 836 S/G 691 S/S 904 S/L 769 L (N=1) Tumor Burden RECIST (cm) 5. 3 (1. 8 -23. 8 ) Baseline Calcitonin (pg/ml) 16064 (414 -67, 057) (Normal < 16) Baseline CEA (ng/ml) 103. 8 (5. 2 -863) (Normal 0. 8 -3. 4) Disease Sites Lymph node(s), lungs, liver, bone, brain, thyroid Prior Treatment Surgery (N=15), RT to neck (1), brain (2) Baseline Post cycle 20 Fox, Widemann, …Wells, Balis, Clin Cancer Res 2013; Kraft I…Widemann, ASCO 2017
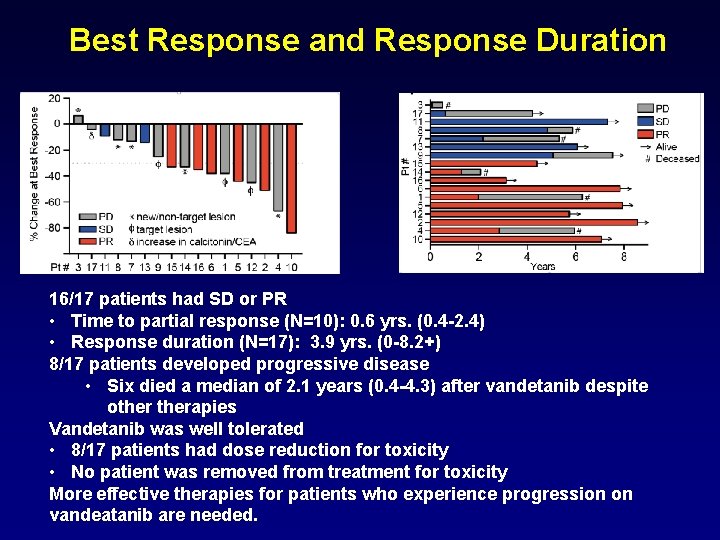
Best Response and Response Duration 16/17 patients had SD or PR • Time to partial response (N=10): 0. 6 yrs. (0. 4 -2. 4) • Response duration (N=17): 3. 9 yrs. (0 -8. 2+) 8/17 patients developed progressive disease • Six died a median of 2. 1 years (0. 4 -4. 3) after vandetanib despite otherapies Vandetanib was well tolerated • 8/17 patients had dose reduction for toxicity • No patient was removed from treatment for toxicity More effective therapies for patients who experience progression on vandeatanib are needed.

Phase I Trial of Cabozantinib (XL 184) in Refractory Solid Tumors (ADVL 1211) • Oral small molecule inhibitor of , multiple kinases including VEGF, MET, RET, CKIT • Phase I trial ADVL 1211: • 41 patients, median age 13 yrs (4 -18 yrs) • Recommended phase II dose: 40 mg/m 2/dose once daily continuous dosing • Cycle 1 DLTs: fatigue, headache, proteinuria, hypertension, RPLS, PPES, oral mucositis, ALT/lipase/bilirubin increase • 4 PRs: MTC (2), Wilms tumor (1) clear cell sarcoma (1) • 7 patients had SD for ≥ 6 cycles: MTC (2 pts) paraganglioma, Ewing sarcoma, synovial sarcoma, ependymoma, and alveolar soft part sarcoma, all N=1 • PK was similar for the 3 dose levels evaluated

Phase II Trial of Cabozantinib (XL 184) in Solid Tumors (ADVL 1622) Primary objective: • Objective response rate (ORR) per RECIST v 1. 1 in the following disease strata: • Ewing sarcoma • Rhabdomyosarcoma • Non-rhabdomyosarcoma soft tissue sarcoma • Wilms tumor (non-statistical cohorts) • Rare tumors (non-statistical cohorts) • Rare tumor strata: Hepatoblastoma, hepatocellular carcinoma, adrenocortical carcinoma, renal cell carcinoma, medullary thyroid carcinoma, tumors with known molecular alterations in targets of cabozantinib Eligibility: • Age: 2 -30 y/o (exception MTC, HCC, RCC 18 y/o) • Ability to swallow tablets Trial status: Ongoing

NIH Pediatric & Wild-type GIST Clinic A model to study rare tumors through intramural extramural collaboration GIST: Connective tissue tumor in the GI tract; KIT or PDGFRA mutations “Wild-type” GIST: Lack KIT or PDGFRA mutations, • No effective medical therapies Pediatric and Wild-type GIST Clinic: • Patients, clinicians (multidisciplinary expertise) • Clinical and basic researchers • Patient support groups • Clinical history and evaluations • Expert clinical advice • Biospecimens for molecular analysis Accomplishments include: • • • Genomic characterization of wt-GIST: Ø SDH mutation, Ø Promotor hypermethylation Increased clinical understanding Ø Changes in clinical management Clinical trials with targeted agents Janeway…Helman, Stratakis, PNAS , 2011 Miettinen…Helman, Meltzer, Am J Surg Pathol 2013 Killian…Helman, Meltzer. Cancer Dicovery, 2013 Killian…Helman, Meltzer, Sci Transl Med, 2014 40 Boikos…Helman. JAMA Oncol, 2016

Phase II trial of the DNA methyl transferase inhibitor, Guadecitabine (SGI-110), in succinate dehydrogenase (SDH) and fumarate hydratase (FH) deficient tumors v v Background: • Increased Krebscycle metabolites lead to inhibition of TET DNA hydroxylases • Hypermethylation in • Wt-GIST Paraganglioma /Pheochromocytoma, Hereditary leiomyomatosis and renal cell cancer (HLRCC) SGI-110: DNMT inhibitor • Derivative of decitabine, but resistant to inactivation by cytidine deaminase • Administered Sub. Q and gradually converted to decitabine resulting in prolonged drug exposure (Several fold increase in apparent T 1/2) • Dosing for this trial 45 mg/m 2/day on days 1 -5 (1 cycle =28 days) 41
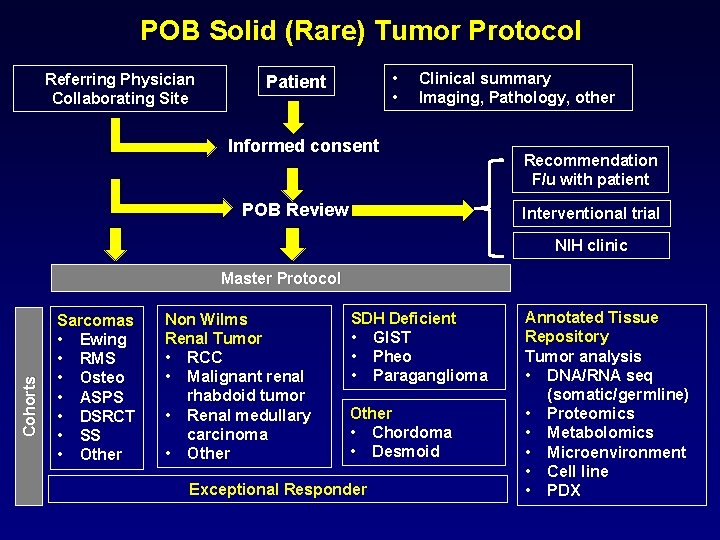
POB Solid (Rare) Tumor Protocol Referring Physician Collaborating Site • • Patient Clinical summary Imaging, Pathology, other Informed consent POB Review Recommendation F/u with patient Interventional trial NIH clinic Cohorts Master Protocol Sarcomas • Ewing • RMS • Osteo • ASPS • DSRCT • SS • Other Non Wilms Renal Tumor • RCC • Malignant renal rhabdoid tumor • Renal medullary carcinoma • Other SDH Deficient • GIST • Pheo • Paraganglioma Other • Chordoma • Desmoid Exceptional Responder Annotated Tissue Repository Tumor analysis • DNA/RNA seq (somatic/germline) • Proteomics • Metabolomics • Microenvironment • Cell line • PDX

Phase I/II Trial of Insulin-Like Growth Factor 1 Receptor (IGF-1 R) Antibody AMG 479 (Ganitumab) in Combination with the Src Family Kinase (SFK) Inhibitor Dasatinib in Patients with Embryonal and Alveolar Rhabdomyosarcoma Christine Heske, MD NCI POB Rhabdomyosarcoma: • Most common soft-tissue sarcoma of childhood • Patients with recurrent alveolar (ARMS) and embryonal (ERMS) • or metastatic disease have dismal survival rates

Study Background / Rationale IGF 1 -R: v Highly expressed in ERMS and ARMS v Early phase studies of IGF-1 R antibodies activity in refractory RMS • ~14% PR or short-lived response, ~8% SD, rapid onset of resistance YES v A non-receptor tyrosine kinase highly expressed in ERMS and ARMS v In vitro blockade of IGF-1 R in RMS cells resulted in YES activation v In mouse tumors YES activation was associated with resistance to IGF-1 R blockade Combination treatment blocking both IGF-1 R and YES results in enhanced growth inhibition of RMS in vitro and in vivo Wan X, et al, Neoplasia, 2015.

Study Overview v v v Primary objective: • Phase I: Determine safety and phase II dose • Phase II: Objective response rate, progression free survival Secondary objectives: • Expolremechanisms of response and resistance: Archival and biopsy tissue: IGF-1 R, IGF-2 and YES Eligibility: • Relapsed or refractory ARMS or ERMS • Ability to swallow tablets • Normal organ function Treatment plan: One cycle = 28 days • Ganitumab (human monoclonal antibody against IGF-1 R): • 18 mg/kg IV every 2 weeks (days 0 and 14) • Dasatinib (multi-targeted kinase inhibitor including SR and YES) • Start at 60 mg/m 2 po QD, escalate to 60 mg/m 2 po BID Single stage design: • Primary endpoint objective response rate (PR + CR) • Target response 30%; ≥ 3 responses consistent with activity • Additional endpoint: Estimate fraction of patients without progression at 4 mo.

Phase II Study of Anti-PD-L 1 Antibody Atezolizumab for Alveolar soft part sarcoma PI: Alice Chen Background: v v v v ASPS rare, slowly progressive, indolent sarcoma Associated with t(X; 17)(p 11; q 25) translocation resulting in a ASPL-TFE 3 fusion protein which activates Mi. T (microphthalmia transcription factor) resulting in over-expression of MET Frquent presentation with metastatic disease Diagnosis in children and adults No effective medical therapies Activity of antiangiogenic agents: • Cediranib, bevacizumab, sunitinib, cabozantinib Immunohistochemical analysis of the metastatic lesions from ASPS demonstrated PD-1 expressing CD 8 T cells and PD-L 1 expressing sarcoma cells 46

Phase II Study of Anti-PD-L 1 Antibody Atezolizumab for Alveolar soft part sarcoma Trial objective: Objective response rate (RECIST), progression free survival v Tumor biopsies (NCI) v Circulating tumor cells Eligibility: v Patients > 6 years of age with histologically or cytologically confirmed ASPS that is not v curable by surgery at the NCI clinical center (≥ 18 years at other participating sites) Drug administration: v IV on day 1 only of every 21 day-cycle (1200 mg in adults, 15 mg/kg [capped at 1200 mg] in children) Design: v Two-stage, expansion if ≥ 1 response in 9 patients v 47

Pilot Study of Genetically Engineered NY-ESO-1 c 259 T in HLA-A 2+ Patients with Synovial Sarcoma Crystal Mackall, John Glod, Sandra D’Angelo Background: • Synovial sarcoma represents ~20% soft tissue sarcomas • The NY-ESO-1 cancer/testis antigen is expressed in 80% of patients with synovial cell sarcoma • Metastatic disease has poor prognosis • Attractive target for immune-based therapies Pilot trial NCI Surgery Branch: • Patients with NY-ESO 1 positive synovial sarcoma treated with autologous TCR-transduced T cells to recognize NY-ESO plus 720, 000 IU/kg of interleukin-2 to tolerance after preparative chemotherapy • Objective responses in 4/6 patients with synovial sarcoma (one response +18 months) Robbins S, Rosenberg SA, JCO 2 o 11

Pilot Study of Genetically Engineered NY-ESO-1 c 259 T in HLA-A 2+ Patients with Synovial Sarcoma Crystal Mackall, John Glod, Sandra D’Angelo Trial: A T-cell receptor recognizing NY-ESO-1 in the context of HLA-A*02: 01 was cloned from a patient with cancer and modified for higher affinity Eligibility: • HLA-A*02: 01, HLA-A*02: 05, HLA-A*02: 06 • NY-ESO-1 expression by IHC • High (2+ or 3+ in ≥ 50% cells) • Low ( ≥ 1+ in ≥ 1% to ≤ 50% cells) Treatment: • Lymphodepletion with fludarabine and cyclophosphamide • No Interleukin 2 administration

Study Design /Response 3 -4 week treatment washout T-cell expansion Harvesting, bead removal & formulation Cell manufacturing 4 -5 weeks T-cell infusion Activation & Transduction of CD 3+ T-cells with NY-ESO-1 TCR Lymphodepletion HLA/antigen screening Eligibility confirmation Leukapheresis Enrichment selection of CD 3+ T -cells Post infusion follow up Disease assessment: Weeks 4, 8, 12 and every 3 months Long term follow up Day -7 to -2 0 Optional hospitalization Results: NY-ESO well tolerated Partial response rate: • 40 -50% in patients receiving fludarabine and cyclophosphamide • 20% in patients receiving cyclophosphamide only

Phase I study of Anti-CD 22 Chimeric Receptor T Cells in Children and Young Adults with Recurrent or Refractory CD 22 Expressing Malignancies Nirali Shah, M. D. , Terry Fry, M. D. , Haying Qin
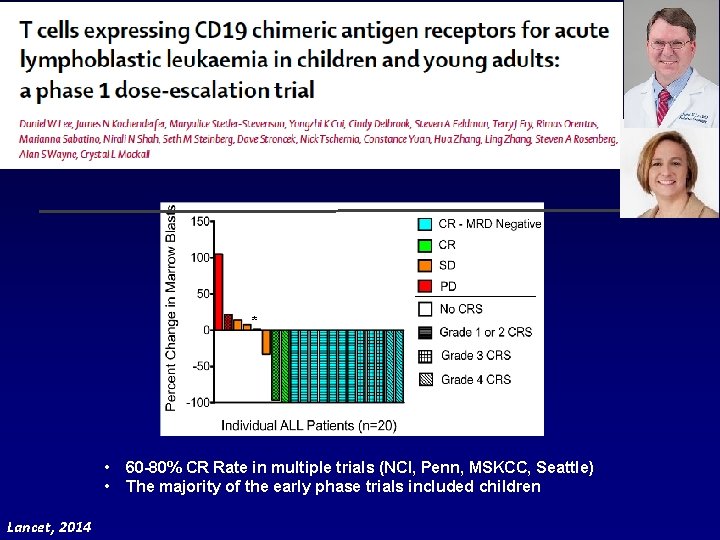
* • • Lancet, 2014 60 -80% CR Rate in multiple trials (NCI, Penn, MSKCC, Seattle) The majority of the early phase trials included children

Construction of Anti-CD 22 CAR § Anti-CD 19 CAR highly effective in pre-B ALL § Loss of CD 19 may limit therapeutic benefit § CD 22, a B-lineage restricted antigen, is a promising target § Second generation CAR § Utilizes m 971 anti-CD 22 sc. Fv § 4 -1 BB/CD 3 -zeta signaling Haso et al, Blood 2013

CD 22 Study Overview v v First in human first in children study Primary objective: • Safety and feasibility • Activity, persistence of CAR T-cells Eligibility: • Relapsed / refractory CD 22 + hematologic malignancy Treatment plan: • Lymphodepletion with • Fludarabine: 25 mg/m 2/day x 3 days • Cyclophosphamide: 900 mg/m 2/day x 1 day Dose level 1 3 x 10 e 5 transduced CAR T cells/kg Dose level 2 1 x 10 e 6 transduced CAR T cells/kg Dose level 3 3 x 10 e 6 transduced CAR T cells/kg
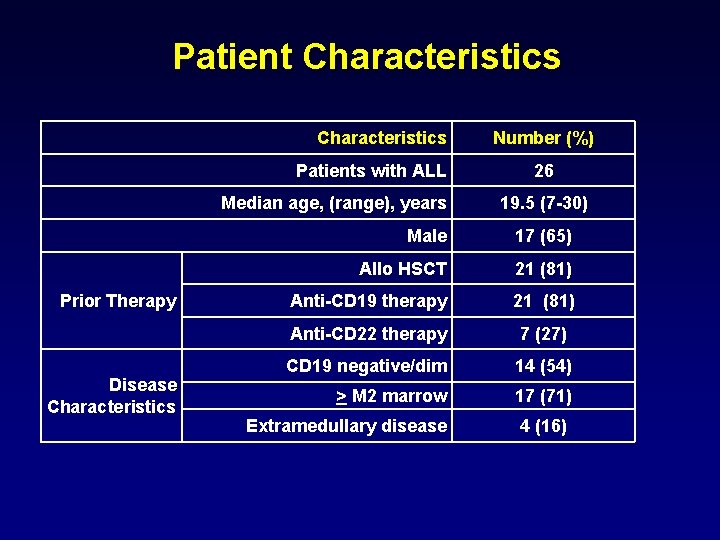
Patient Characteristics Patients with ALL Median age, (range), years Prior Therapy Disease Characteristics Number (%) 26 19. 5 (7 -30) Male 17 (65) Allo HSCT 21 (81) Anti-CD 19 therapy 21 (81) Anti-CD 22 therapy 7 (27) CD 19 negative/dim 14 (54) > M 2 marrow 17 (71) Extramedullary disease 4 (16)
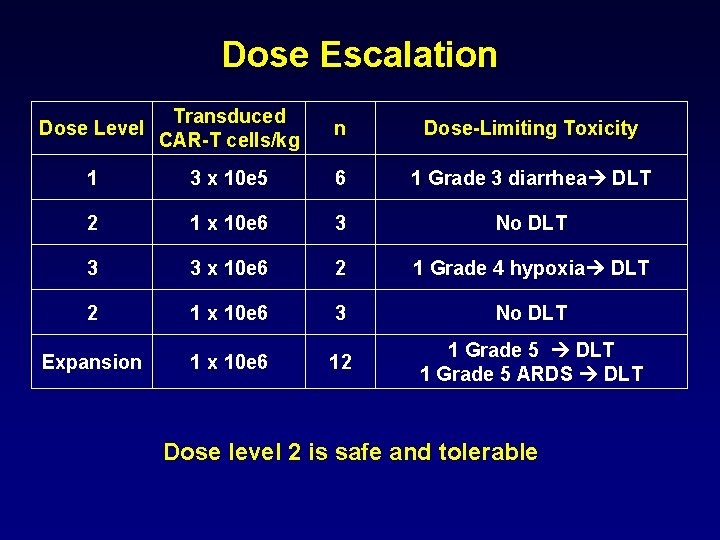
Dose Escalation Dose Level Transduced CAR-T cells/kg n Dose-Limiting Toxicity 1 3 x 10 e 5 6 1 Grade 3 diarrhea DLT 2 1 x 10 e 6 3 No DLT 3 3 x 10 e 6 2 1 Grade 4 hypoxia DLT 2 1 x 10 e 6 3 No DLT Expansion 1 x 10 e 6 12 1 Grade 5 DLT 1 Grade 5 ARDS DLT Dose level 2 is safe and tolerable
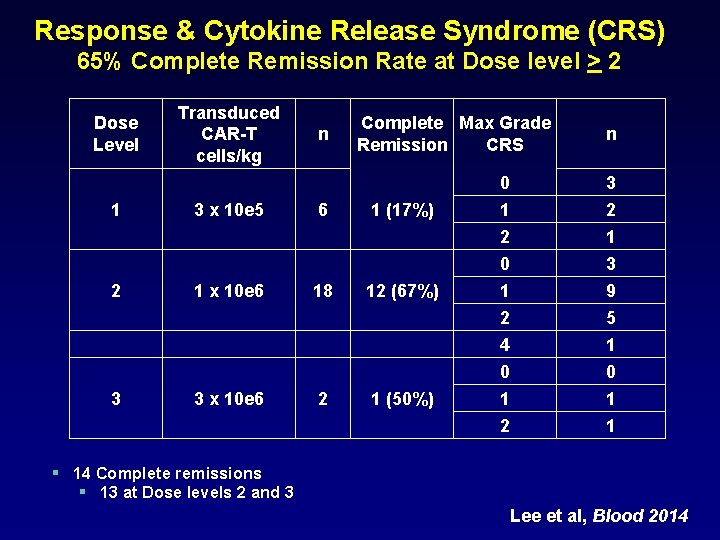
Response & Cytokine Release Syndrome (CRS) 65% Complete Remission Rate at Dose level > 2 Dose Level 1 2 3 Transduced CAR-T cells/kg 3 x 10 e 5 1 x 10 e 6 3 x 10 e 6 n 6 18 2 Complete Max Grade Remission CRS 1 (17%) 12 (67%) 1 (50%) n 0 1 2 3 2 1 0 1 2 4 3 9 5 1 0 1 2 0 1 1 § 14 Complete remissions § 13 at Dose levels 2 and 3 Lee et al, Blood 2014
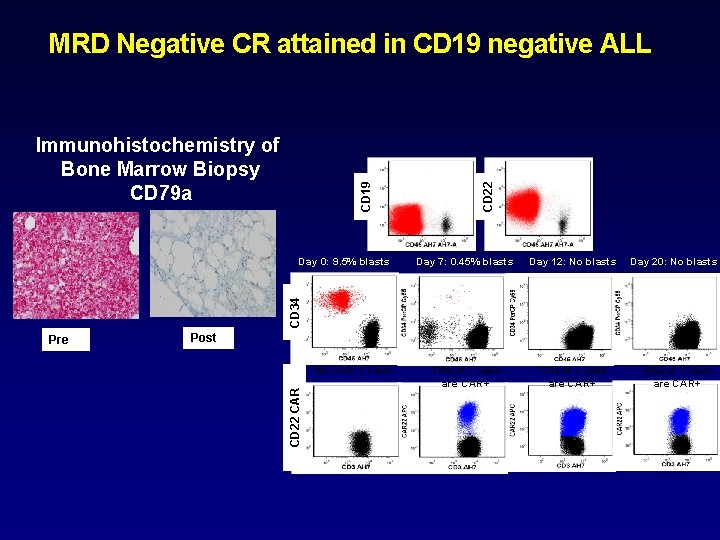
CD 19 Immunohistochemistry of Bone Marrow Biopsy CD 79 a Day 7: 0. 45% blasts Day 12: No blasts 11% of T cells are CAR+ 77% of T cells are CAR+ Day 20: No blasts CD 34 Day 0: 9. 5% blasts CD 22 MRD Negative CR attained in CD 19 negative ALL Post No CAR-T Cells CD 22 CAR Pre 45% of T cells are CAR+
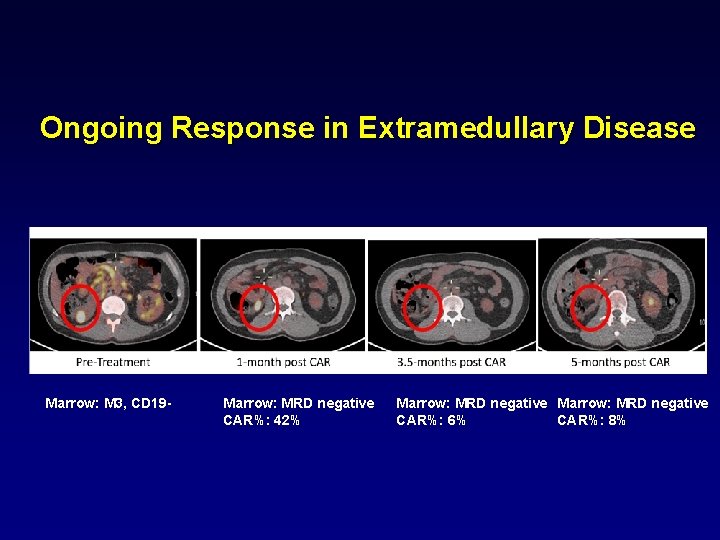
Ongoing Response in Extramedullary Disease Marrow: M 3, CD 19 - Marrow: MRD negative CAR%: 42% Marrow: MRD negative CAR%: 6% CAR%: 8%

CD 22 Antigen Expression at Relapse § 10 § § patients with relapse 1—CAR cell loss (dose level 1) 7—with changes in CD 22 § 1 with relapse at 11 months § 3 with relapse at 6 months § 1 with relapse at 3 months § 2 with relapse at 2 months* § Changes in CD 22 § Decrease in Site Density (n=6) § Antigen loss (n=1) § Both (n=1) *Received prior anti-CD 22 targeted therapy

Summary from Initial Experience First successful salvage CAR therapy for CD 19 negative BALL v Preliminary experience suggests comparable potency to anti-CD 19 CAR v Response correlated with dose level v Mostly Grade 1 or 2 CRS v Relapses associated with changes in CD 22 expression level Challenges: v Durability of Remissions • Optimizing CAR T cell Product • Understanding leukemic resistance v Incorporation into standard treatment paradigms v Other targets? v

What is Pediatric MATCH? 62

NCI-COG Pediatric Molecular Analysis for Therapy Choice (MATCH) APEC 1621 Study A phase 2 precision medicine cancer trial Co-developed by the Children’s Oncology Group and the National Cancer Institute Nita Seibel, M. D. CTEP June 4, 2017

Hypothesis By identifying genetic changes affecting pathways of interest in refractory and recurrent pediatric cancers, we will be able to deliver targeted anticancer therapy that produces a clinically meaningful objective response rate. 64

NCI-COG Pediatric MATCH BRAF inhibitor PI 3 K-m. TOR inhibitor MEK inhibitor FGFR inhibitor 1. 2. 3. 4. 5. TRKinhibitor EZH 2 inhibitor ALK inhibitor PARP inhibitor Single stage 20 patients per arm Non-histology driven Estimate 300 patients/year ~8 agents to start NO ACTIONABLE MUTATION DETECTED (90%) 65

Levels of Evidence for Drugs in NCI-COG Pediatric MATCH v Level 1: FDA approved for any indication for that target v Level 2: Agent met a clinical endpoint (objective response, PFS, or OS) with evidence of target inhibition v Level 3: Agent demonstrated evidence of clinical activity with evidence of target inhibition at some level 66

NCI-COG Pediatric MATCH Design Features v Test many children and adolescents to find widely distributed genetic alterations v Requirement for biopsy: must obtain tissue postrelapse for study eligibility except for brain stem glioma patients • Rationale: Tumor genomes evolve. To identify potential targets for therapy a “current” relapsed sample is needed v Most patients screened will be biomarker negative and will not match to a treatment arm v Inclusion of agents with adult RP 2 D 67

NCI-COG Pediatric MATCH Design Features v Response rate (tumor regression) will be primary efficacy measure v Possibility of assignment of patients with non-target-bearing tumors to selected agents that have demonstrated activity in targetbearing tumors v Evaluation of germline DNA Parsons DW et al. JAMA Oncol, 2015 68

THANK YOU
- Slides: 69