NCDR Registry Requirements and EMR Capabilities Wendy Schell

NCDR Registry Requirements and EMR Capabilities Wendy Schell, MSN, RN, AGACNP-BC, APN, CCRN Amy Schwartz, BSN, RN

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship Company Grant/Research Support Consulting Fees/Honoraria Major Stock Shareholder/Equity Royalty Income Ownership/Founder Intellectual Property Rights Other Financial Benefit Company Names Company Names

Agenda • Registry Requirements Supporting NCD • Shared Decision Making • Registry – Getting Started – Resources – Data collection – Data submission • EMR Build • Q & A

Background Information • CMS released the national coverage determination (NCD) for LAA – Effective date of coverage is February 8, 2016 • All Medicare Administrative Contractors have updated their Category III code policies to include coverage of 0281 T under the LAAC NCD • CMS forwarded program instructions to the local contractors to update their claims processing systems to be in compliance with the NCD – Implementation date: October 3, 2016 – MLN article on LAAC defines billing instructions for hospitals and physicians • Registry certification occurred August 17, 2016

LAAO Registry • • • The LAAO Registry is designed to assess the prevalence, demographics, management, and outcomes of patients undergoing percutaneous and epicardial based left atrial appendage occlusion procedures to reduce the risk of stroke. The primary goals of the registry are to optimize outcomes and management of patients through the implementation of evidence-based guideline recommendations in clinical practice, facilitate efforts to improve the quality and safety for these patients, investigate novel quality improvement methods, and provide risk-adjusted assessments of patients for comparison with nationwide NCDR data. The secondary purpose of the registry is to serve as source of clinical data to support assessments of short and long term safety, comparative and cost effectiveness research and as a scalable data infrastructure for post market studies. Physicians and hospitals can use these data to support quality improvement efforts locally, monitor patterns of care, and assess procedure utilization and variability. Registry data will be used to help develop clinical guidelines leading to improved patient care and ultimately will provide a better understanding of this population and emerging treatment options.
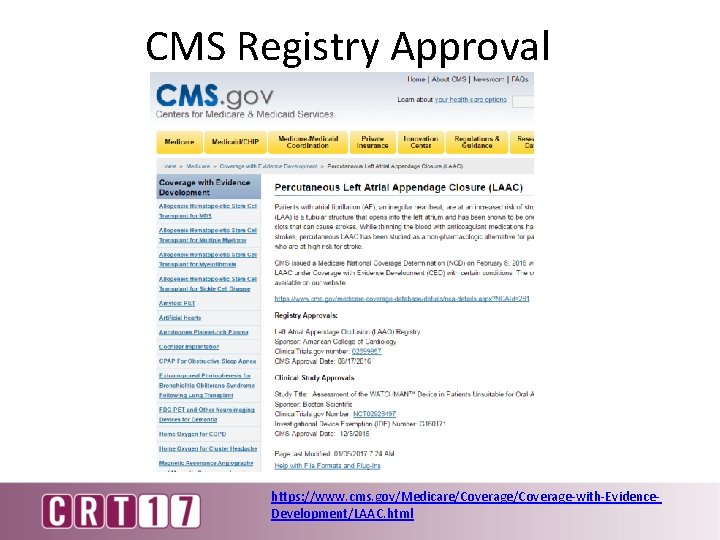
CMS Registry Approval https: //www. cms. gov/Medicare/Coverage-with-Evidence. Development/LAAC. html

NCD for LAAO • Procedure must be performed in a hospital with an established structural heart disease or EP program. • Procedure must be performed by an IC, EP, or Cardiovascular Surgeon meeting the following criteria: – Trained by the manufacturer – ≥ 25 interventional cardiac procedures involving transseptal punctures through an intact septum – Continues to perform ≥ 25 interventional cardiac procedures involving trans-septal punctures through an intact septum, with at least 12 being LAAC over a two year period • Must be enrolled in a prospective national registry

LAAO Registry Inclusion Population • Consecutive left atrial appendage occlusion procedures on patients >= 18 years • Devices implanted via a percutaneous/catheter-based approach • Device is non-investigational and FDA approved • Inclusive of off label device use where device is being used for another purpose other than the described indication given to the device as part of FDA approval • Open surgical approaches not included • Sampling not permitted • Patient entered location in which procedure is intended to be performed
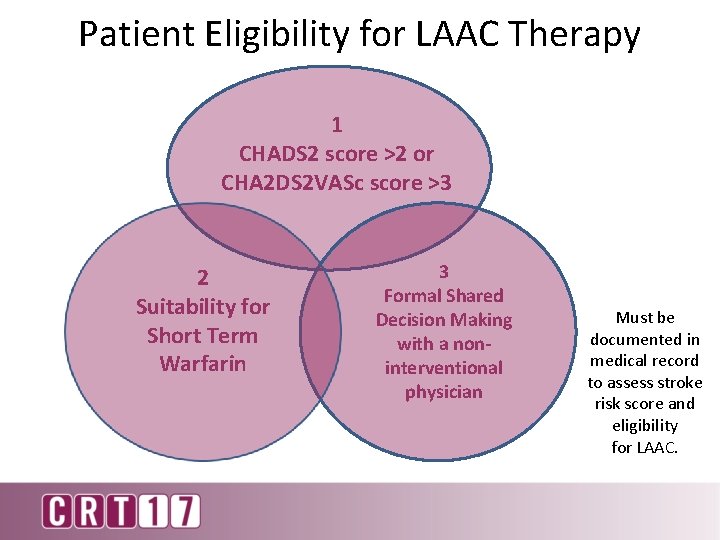
Patient Eligibility for LAAC Therapy 1 CHADS 2 score >2 or CHA 2 DS 2 VASc score >3 2 Suitability for Short Term Warfarin 3 Formal Shared Decision Making with a noninterventional physician Must be documented in medical record to assess stroke risk score and eligibility for LAAC.

NCD for LAAO Eligible patients must have: • CHADS 2 score ≥ 2 or CHA 2 DS 2 -VASc score ≥ 3. • Suitable for short-term warfarin • Deemed unable to take long-term oral anticoagulation. Documented Evidence of a formal shared decision interaction between the patient and an independent, non interventional physician using an evidence-based decision tool on oral anticoagulation in patients with NVAF prior to LAAC. • (LAAC is only covered as second line therapy to OAC) Sign Up for Cardio. Smart on ACC Website

CMS Shared Decision Shared decision making has the patient at the center and is much more structured, evidencebased and importantly interactive with bilateral exchange of information compared to informed consent. Patients differ in their risk for ischemic stroke and risk of major bleeding, making generic information inadequate. The NICE patient decision aid does not mention LAAC. While CMS recognizes that no evidence based shared decision making tool on LAAC has been published, we encourage the development of such a tool given the complex considerations for anticoagulation in NVAF and invasive interventions. We have required the use of an evidence-based tool on anticoagulation in NVAF since consideration of anticoagulation is inseparable from the decision on LAAC. In the meantime, the shared decision making interaction will be augmented with the addition of non-interventional providers that either have a relationship with the patient, such as the primary care provider, or those with experience caring for stroke patients, to the patient’s care team.

Documentation Criteria Referring/Consult note should reflect: – Outline the decision for LAAO based on past history – List current and past medication use (antiplatelets , NSAIDs, NOACs, Warfarin) & contraindication to anticoagulation – SDM Tool utilized – CHA 2 DS 2 -VASc score and Yearly Risk of Stroke % – (Encourage HAS-BLED) Specialist Consults Involved: – GI – Neuro – Hematologist

Supporting the Shared Decision • Education Packets • Tools available – ACC – NICE – SPARC
Patient Welcome & Education

Shared Decision Making Tools: q ACC Shared Decision Tool q NICE Tool q Sparc Tool

d e r a h l S o o C T C A n o i s i c e D

l o o T C y C p A o C d r a H https: //www. cardiosmart. org/hea lthwise/te 81/26/te 8126#te 8129

l o o T E NIC

E C d I i N n. A n o o i i t s i a c t De men u c Do

L O O T C R A SP
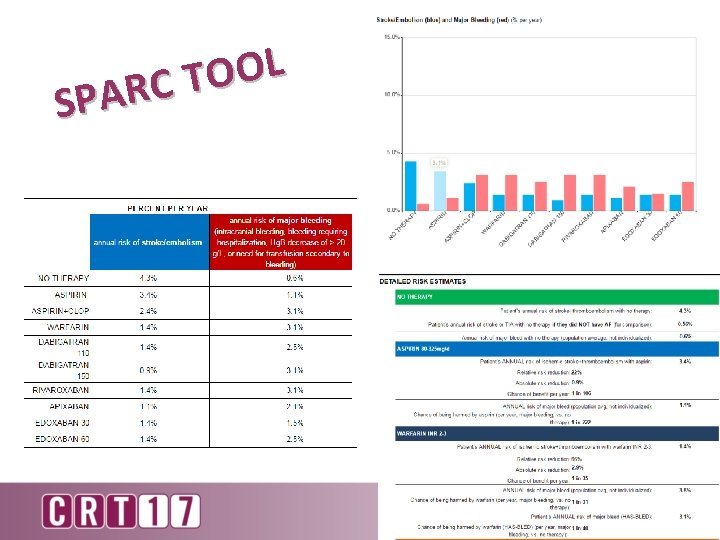
L O O T C R SPA

r o P f C P orm n o F i l s a eci r r e D f e R ared Sh


T L U S N O C N N O I T AP A T N E M U C S E DO L P SAM

CHA 2 DS 2 VASc Score 5 (CHF, HTN, Age, DM) HASBLED Score 4 Assessment/Plan: 78 year old gentleman with PMH chronic diastolic heart failure with mild systolic failure, chronic persistent afib on warfarin who presents with hematochezia and shortness of breath now evaluated for potential Watchman therapy. Mr. X. has a history of non valvular AF. He is at increased risk for stroke and systemic embolism based on a CHA 2 DS 2 -VASc 1 Score of 5 and is recommended for anticoagulation therapy. He is being evaluated for a Watchman Device as an alternative to warfarin and Mr. X has been found to be suitable for short term Warfarin however is unable to take long term Warfarin for the following reasons: recurrent episodes of GI bleeding and gastritis found on recent EGD with a HASBLED Score of 4 The patient’s current medical management regimen includes: ASA and Warfarin Side effects suffered from these medications include: Recurrent GI Bleeding requiring hospitalization and temporary discontinuation of Warfarin. He was deemed to be not a candidate for ablation per electrophysiology The rational for this procedure over pharmacologic therapy includes: Recurrent GI bleeding affecting Mr X's quality of life taking into account the safety and effectiveness of the device compared to warfarin. A formal shared decision was reached with Mr, X Through the use of the ACC Shared decision making tool (Scanned to media section of Epic) and He has made the decision to have this device implanted with hopes of eliminating continued necessity for the long-term use of warfarin. I feel that Mr. X will benefit greatly from this procedure. His quality of life and well-being is greatly impacted by atrial fibrillation and other health conditions including recurrent Diverticulosis , Gastritis, GI bleeding, dizziness with recent fall, and Chronic congestive heart failure. - TEE for Assessment of LAA Case and Plan discussed with Dr. Sabir Wendy Schell APN-C Cardiology – Structural Heart Team University Cardiology Service Cooper University Hospital

I, Sajjad A Sabir, MD, have seen and examined the patient on 3/24/2016. I agree with the resident's assessment and plan as written, with exceptions as noted. TEE images reviewed personally and 3 D reconstruction performed. Complex left atrial appendage (LAA) with multiple lobes. Two small lobes are just distal to the LAA os. Would have to deploy the device just above that to ensure no lobe is left uncovered. There is a small chance of not being able to do that in which case the procedure would have to be aborted. I discussed this and the rest of the details of the procedure including risks and benefits with patient in detail. The risks include but are not limited to bleeding, groin hematoma, pseudoaneurysm, AV fistula, infection, pericardial effusion, tamponade, arrhythmias, stroke, device embolization, emergency surgery, contrast induced nephropathy, and death. Patient would also need to be "cleared" by GI to be able to take the coumadin for at least 6 weeks post-implant. At 6 weeks, we repeat a TEE. If no thrombus on the device or no significant leak around the device, coumadin will be stopped and replaced by Plavix up until he is 6 months out from the procedure. Pt would like to proceed "as soon as possible". He was hoping to have it done during this admission but the next available date is Wednesday April 20. Will tentatively add pt on for that day. Called Dr. Nussbaum's cell and left a message on her cell. Team can call me at my cell at 267 -716 -7757 if any questions. Pt also has our Structural Heart direct cell phone number he can call us on if he has any additional questions. ------------1 January CT, Wann LS, Alpert JS, et. al. , 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society, Circulation, 2014; 130: e 199 -e 267.




N A L N P O I C T A A A T L N E M U C O D


LAAO Registry: Getting Started • Go to NCDR website: • http: //cvquality. acc. org/NCDR-Home/Registries/Hospital. Registries. aspx • Click on “Join a Registry” and fill out required information and choose LAAO Registry™ under Hospital Registries. • Fee: $15, 000 • 1. Download LAAO Registry Packet • 2. Complete enrollment materials • 3. Mail completed materials along with payment to NCDR • Contact: 800 -257 -4737 or ncdr@acc. org

National Audited Registry • Consecutively enrolls LAAO patients • Tracks the following annual outcomes for each patient for a period of at least two years from the time of the LAAC: • • • Operator-specific complications Device-specific complications including device thrombosis Stroke/ TIA, adjudicated, by type Systemic embolism Death Major bleeding, by site and severity
Education Home Page

Monthly Site Manager & User Calls

RSM and New User Call Schedule

Monthly Case Scenarios

Monthly Case Scenario Education
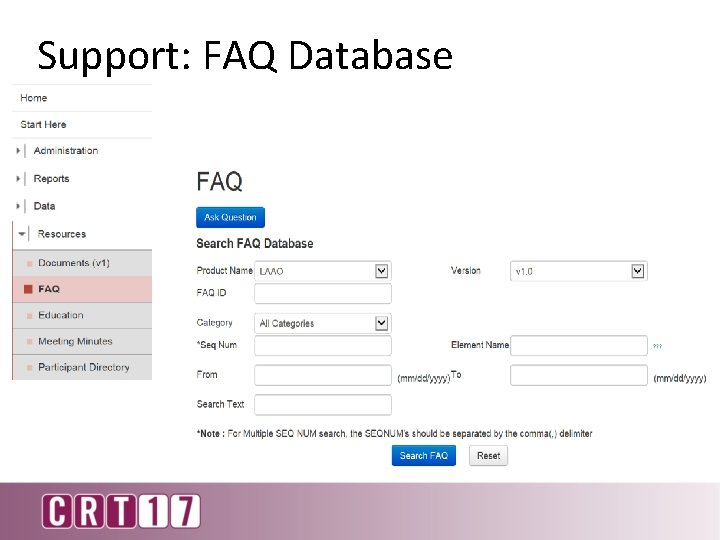
Support: FAQ Database

Are the Modified Rankin Scale and Barthel Index Evaluation required to be administered for each follow-up? • Participants should provide responses for all of the data elements, including follow-up. • Individual clinicians or facilities may have their own protocols based on device selection and directions for use as to what is evaluated at each follow-up • All sequences in the follow-up require a response, if there is no documentation of the Modified Rankin Scale and the Barthel Index Evaluation performed… • sequence #13081 Modified Rankin Scale “Not Administered” • sequence #13085 “No”, Barthel Index Evaluation Performed

Data Dictionary Coding Instructions Target Values Valid Ranges
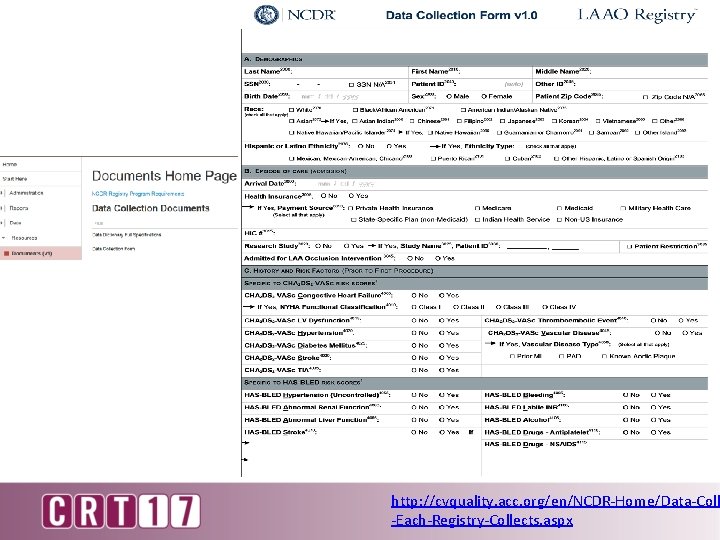
http: //cvquality. acc. org/en/NCDR-Home/Data-Coll -Each-Registry-Collects. aspx
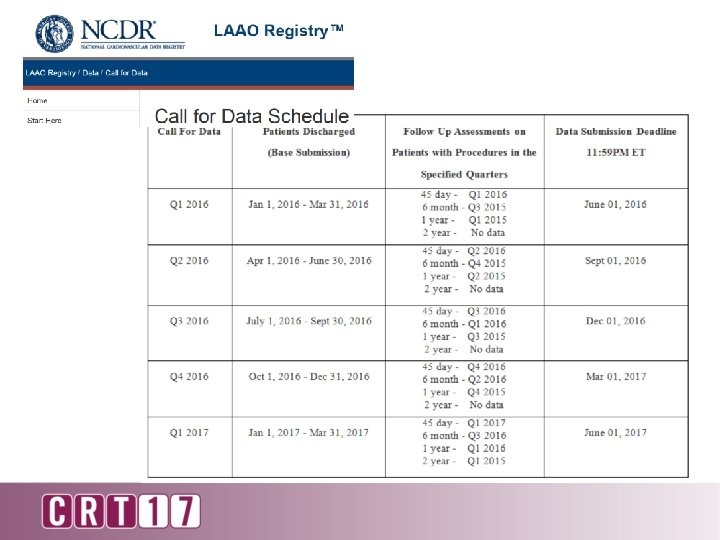
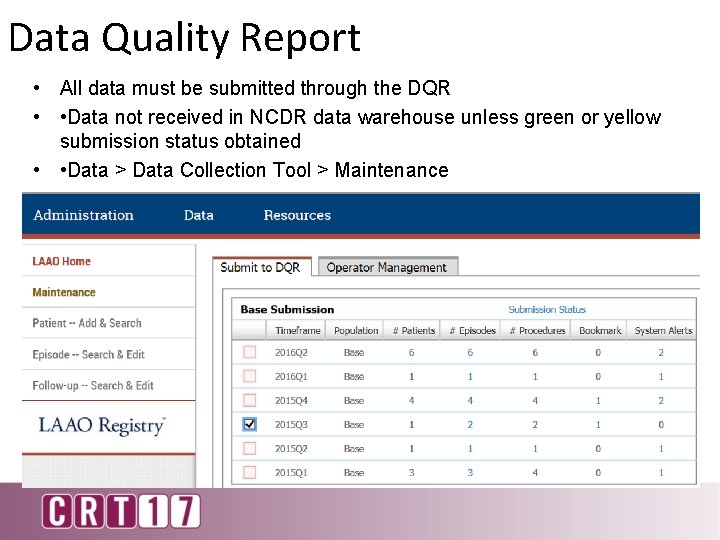
Data Quality Report • All data must be submitted through the DQR • • Data not received in NCDR data warehouse unless green or yellow submission status obtained • • Data > Data Collection Tool > Maintenance

DQR Benchmark Inclusion Status Falling Data Assessment • Data NOT received in data warehouse • Remains only on the hospital platform in DCT • Will not be in the Outcomes Report Falling Completeness Assessment • • Data received in the data warehouse NOT included in metrics, rolling 4 quarters, or national aggregates Displayed in detail section quarterly statistics Passing the DQR • • • Data received in the data warehouse included in metrics, rolling 4 quarters, and national aggregates
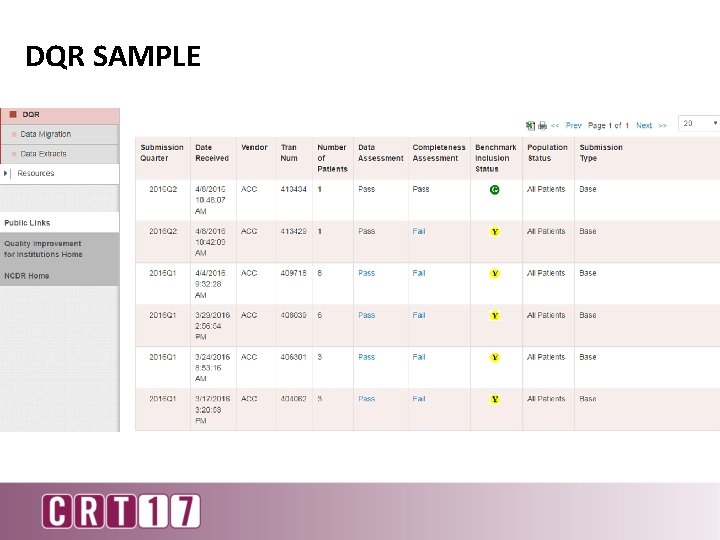
DQR SAMPLE

Follow Up Data • Base submissions must be complete and pass data submission prior to submitting follow up data • Create only 1 follow up for each of the 4 time frames • Submit all follow ups regardless of when they occur

Follow Up Details (No metrics yet established for follow-ups) • Each procedure requires follow up at these intervals: (these have changed) • 45 day (+/- 14 days) • 6 month (+60 days/- 30 days) • 1 year (+/- 60 days) • 2 year (+/- 60 days) • Methods to determine follow up status may vary: • Office Visit • Medical Records • Letter from Medical Provider • Phone Call • Social Security Death Master File • Hospitalized (this has to be >24 hours or overnight otherwise check office visit)

In-Hospital Metrics


Adjudication of adverse events occurring in hospital as well as follow up period: A. Neurologic 1. 2. 3. 4. 5. B. Hemorrhagic Stroke Ischemic Stroke Undetermined Stroke TIA Intracranial Hemorrhage Bleeding 1. 2. 3. 4. 5. Access Site Bleeding Hematoma Vascular Complications GI Bleeding Retroperitoneal Bleeding Future adjudication - Systemic Thromboembolism

EMR Build

EMR Build

EMR Build

R M n E o i s i c e D n d o e i r t Sha menta u c o D

R EM SD
CQI = Improved Patient Care

Questions?
- Slides: 58