Navigating the evolving therapeutic landscape in M 1



![Abiraterone vs Docetaxel (STAMPEDE post-hoc analysis) Failure-free survival [driven by PSA failure] SOC+AAP HR Abiraterone vs Docetaxel (STAMPEDE post-hoc analysis) Failure-free survival [driven by PSA failure] SOC+AAP HR](https://slidetodoc.com/presentation_image/801a6ddc4c2ec174f555b5c7c93b4b54/image-24.jpg)


- Slides: 35

Navigating the evolving therapeutic landscape in M 1 Prostate Cancer Karim Fizazi, MD, Ph. D Institut Gustave Roussy Villejuif, France

Disclosure Participation to advisory boards/honorarium for: Amgen, Astellas, Astrazeneca, Bayer, Curevac, Essa, Janssen, MSD, Orion, Sanofi

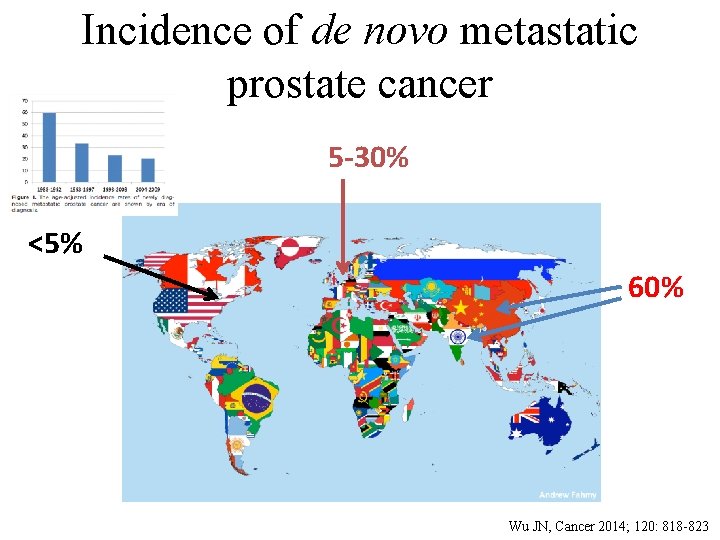
Incidence of de novo metastatic prostate cancer 5 -30% <5% 60% Wu JN, Cancer 2014; 120: 818 -823

Who Dies of Prostate Cancer? 44% of deaths Upfront localized cancer Metastases De novo metastases Death 56% of deaths Patrikidou A. Prostate Cancer Prostatic Dis. 2014; 17: 348 -352.

The « ECG » of clinical research in M 1 prostate cancer Charles Huggins 1966 Nobel Prize Medical castration PC inhibited by castrationwith LHRH agonists 1940 1980 OS improved by Abiraterone and Docetaxe 2013 2018

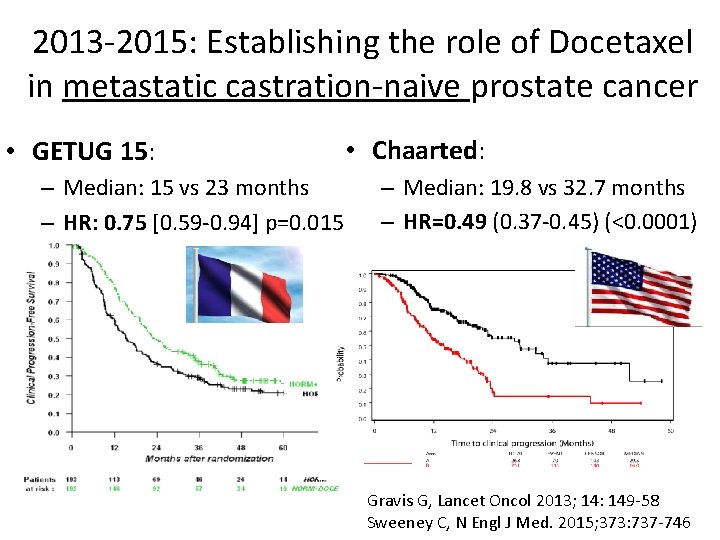
2013 -2015: Establishing the role of Docetaxel in metastatic castration-naive prostate cancer • GETUG 15: – Median: 15 vs 23 months – HR: 0. 75 [0. 59 -0. 94] p=0. 015 • Chaarted: – Median: 19. 8 vs 32. 7 months – HR=0. 49 (0. 37 -0. 45) (<0. 0001) Gravis G, Lancet Oncol 2013; 14: 149 -58 Sweeney C, N Engl J Med. 2015; 373: 737 -746

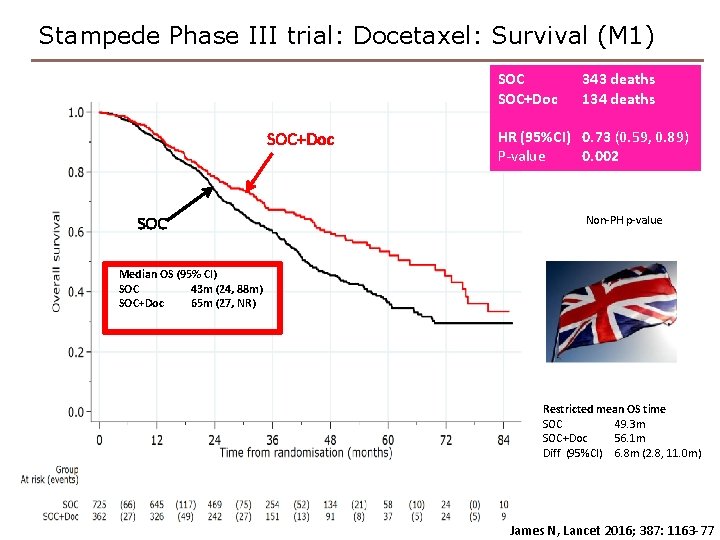
Stampede Phase III trial: Docetaxel: Survival (M 1) SOC+Doc SOC 343 deaths 134 deaths HR (95%CI) 0. 73 (0. 59, 0. 89) P-value 0. 002 Non-PH p-value Median OS (95% CI) SOC 43 m (24, 88 m) SOC+Doc 65 m (27, NR) Restricted mean OS time SOC 49. 3 m SOC+Doc 56. 1 m Diff (95%CI) 6. 8 m (2. 8, 11. 0 m) James N, Lancet 2016; 387: 1163 -77

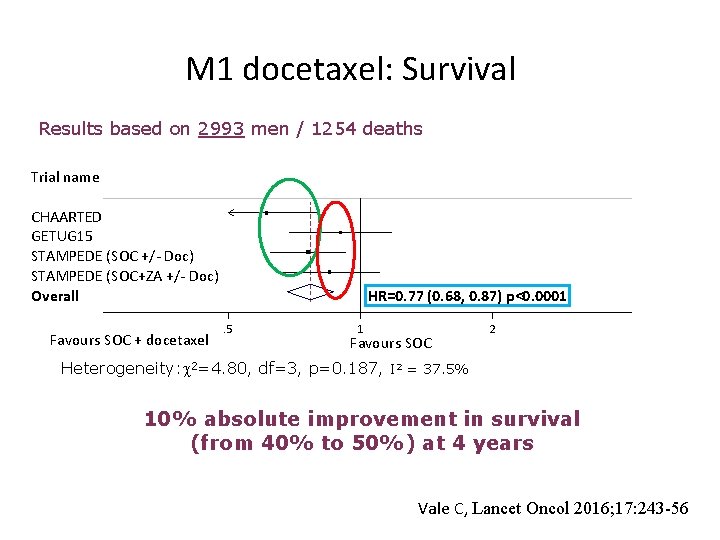
M 1 docetaxel: Survival Results based on 2993 men / 1254 deaths Trial name CHAARTED GETUG 15 STAMPEDE (SOC +/- Doc) STAMPEDE (SOC+ZA +/- Doc) Overall Favours SOC + docetaxel HR=0. 77 (0. 68, 0. 87) p<0. 0001. 5 1 Favours SOC 2 Heterogeneity: 2=4. 80, df=3, p=0. 187, I 2 = 37. 5% 10% absolute improvement in survival (from 40% to 50%) at 4 years Vale C, Lancet Oncol 2016; 17: 243 -56

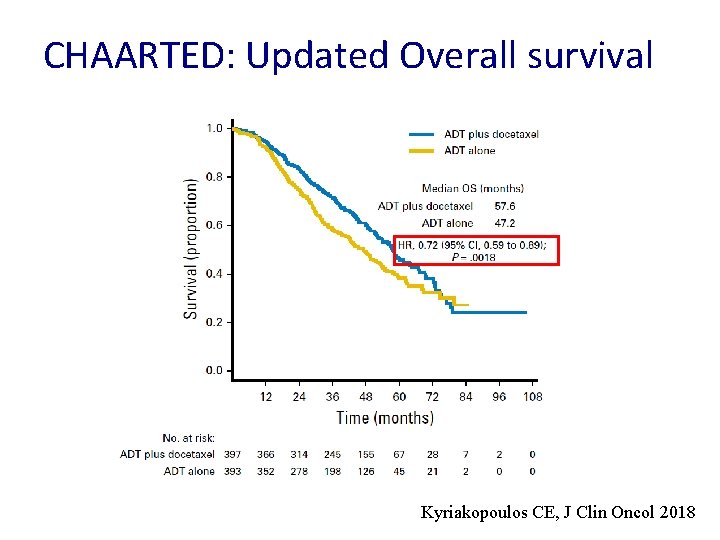
CHAARTED: Updated Overall survival Kyriakopoulos CE, J Clin Oncol 2018

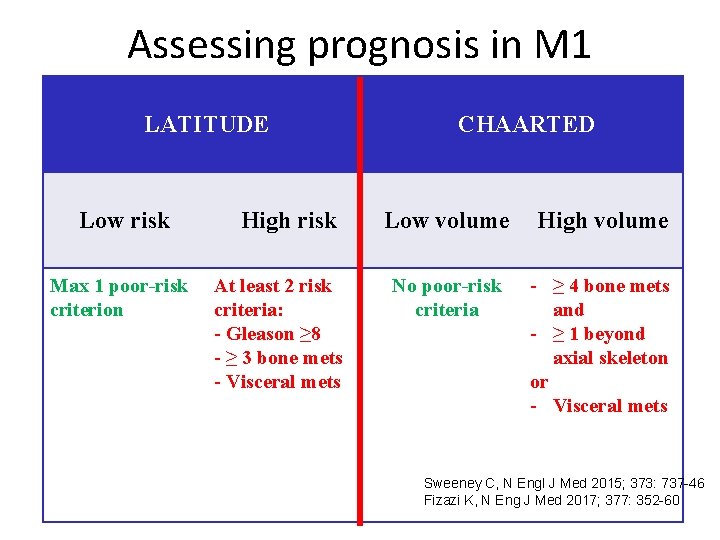
Assessing prognosis in M 1 LATITUDE Low risk Max 1 poor-risk criterion High risk At least 2 risk criteria: - Gleason ≥ 8 - ≥ 3 bone mets - Visceral mets CHAARTED Low volume High volume No poor-risk criteria - ≥ 4 bone mets and - ≥ 1 beyond axial skeleton or - Visceral mets Sweeney C, N Engl J Med 2015; 373: 737 -46 Fizazi K, N Eng J Med 2017; 377: 352 -60

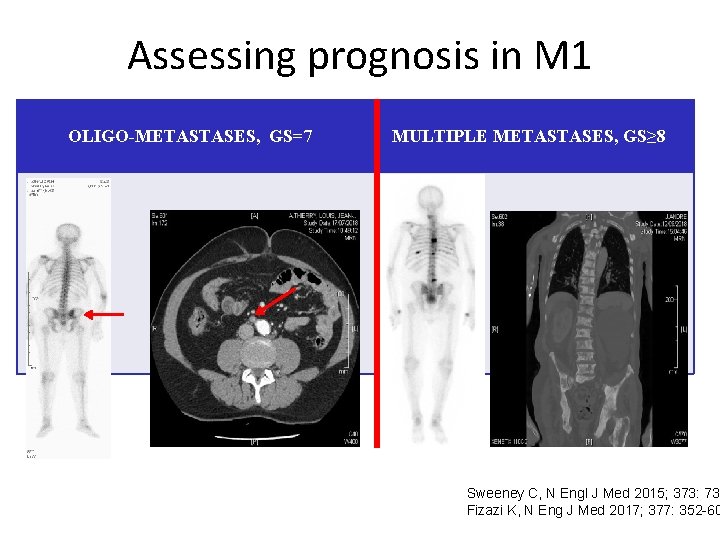
Assessing prognosis in M 1 OLIGO-METASTASES, GS=7 MULTIPLE METASTASES, GS≥ 8 Sweeney C, N Engl J Med 2015; 373: 737 Fizazi K, N Eng J Med 2017; 377: 352 -60

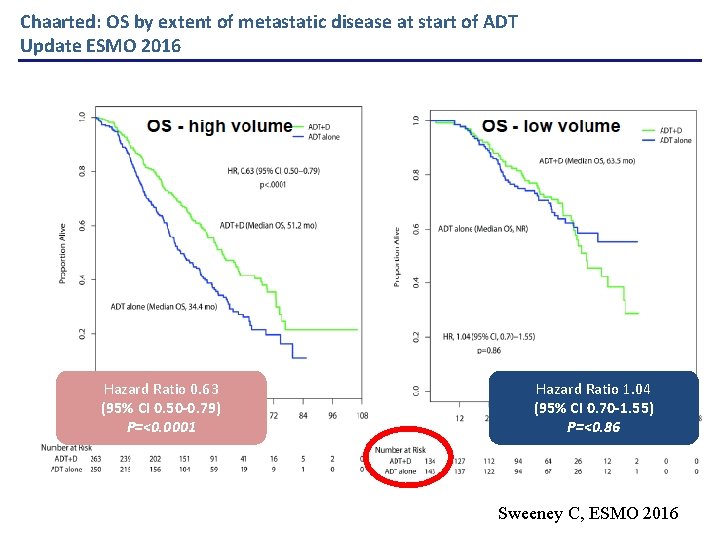
Chaarted: OS by extent of metastatic disease at start of ADT Update ESMO 2016 Hazard Ratio 0. 63 (95% CI 0. 50 -0. 79) P=<0. 0001 Hazard Ratio 1. 04 (95% CI 0. 70 -1. 55) P=<0. 86 Sweeney C, ESMO 2016

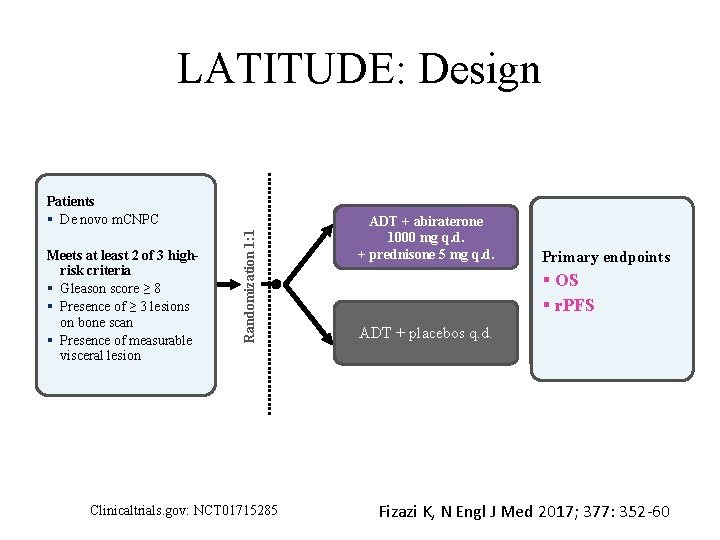
LATITUDE: Design Meets at least 2 of 3 highrisk criteria § Gleason score ≥ 8 § Presence of ≥ 3 lesions on bone scan § Presence of measurable visceral lesion Randomization 1: 1 Patients § De novo m. CNPC Clinicaltrials. gov: NCT 01715285 ADT + abiraterone Abiraterone 1000 mg QD + Prednisone mg QD 1000 mg 5 q. d. + ADT + prednisone 5 mg q. d. Placebo 1000 mg QD + Placebo 5 mg QDq. d. ADT + placebos + ADT Efficacy endpoints Primary: endpoints • Primary OS • r. PFS Secondary: • Time to next skeletal-related event • Time to PSA progression § OS § r. PFS Fizazi K, N Engl J Med 2017; 377: 352 -60

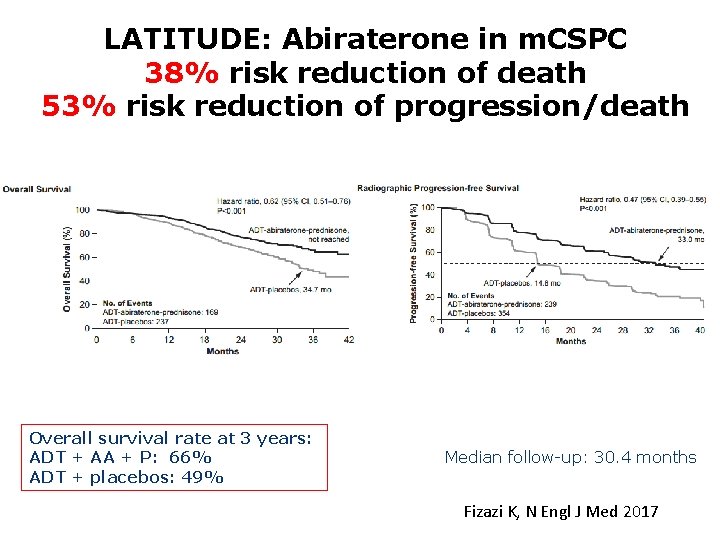
LATITUDE: Abiraterone in m. CSPC 38% risk reduction of death 53% risk reduction of progression/death Overall survival rate at 3 years: ADT + AA + P: 66% ADT + placebos: 49% Median follow-up: 30. 4 months Fizazi K, N Engl J Med 2017 19

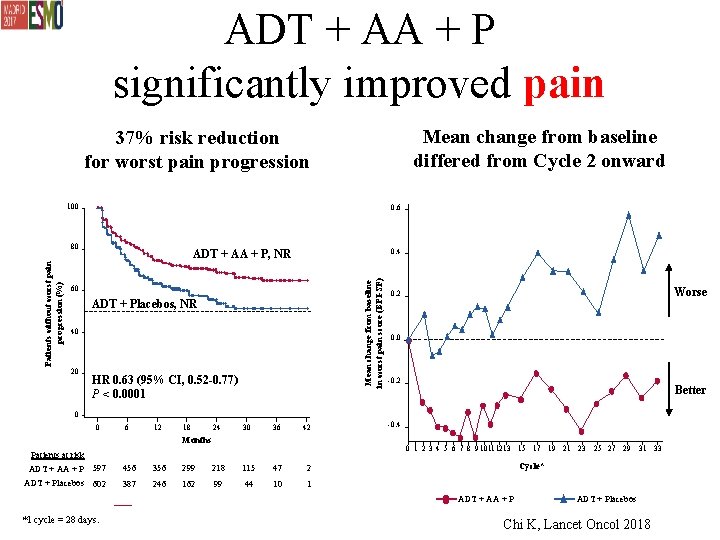
ADT + AA + P significantly improved pain Mean change from baseline differed from Cycle 2 onward 37% risk reduction for worst pain progression 100 0. 6 0. 4 ADT + AA + P, NR Mean change from baseline in worst pain score (BPI-SF) Patients without worst pain progression (%) 80 60 ADT + Placebos, NR 40 20 HR 0. 63 (95% CI, 0. 52 -0. 77) P < 0. 0001 Worse 0. 2 0. 0 -0. 2 Better 0 0 6 12 18 24 30 36 42 Months -0. 4 0 1 2 3 4 5 6 7 8 9 10 11 1213 15 17 19 21 23 25 27 29 31 33 Patients at risk ADT + AA + P 597 456 356 299 218 115 47 2 ADT + Placebos 602 387 246 162 99 44 10 1 Cycle* ADT + AA + P *1 cycle = 28 days. ADT + Placebos Chi K, Lancet Oncol 2018

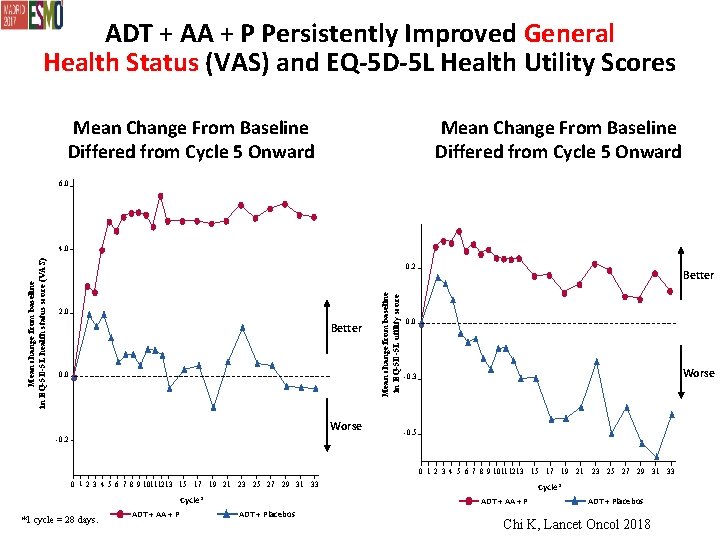
ADT + AA + P Persistently Improved General Health Status (VAS) and EQ-5 D-5 L Health Utility Scores Mean Change From Baseline Differed from Cycle 5 Onward 6. 0 0. 2 2. 0 Better 0. 0 Worse -0. 2 Mean change from baseline in EQ-5 D-5 L utility score Mean change from baseline in EQ-5 D-5 L health status score (VAS) 4. 0 Better 0. 0 Worse -0. 3 -0. 5 0 1 2 3 4 5 6 7 8 9 1011 12 13 15 17 19 21 23 25 27 29 31 33 0 1 2 3 4 5 6 7 8 9 1011 1213 15 17 19 21 23 25 27 29 31 33 Cycle* *1 cycle = 28 days. ADT + AA + P Cycle* ADT + AA + P ADT + Placebos Chi K, Lancet Oncol 2018

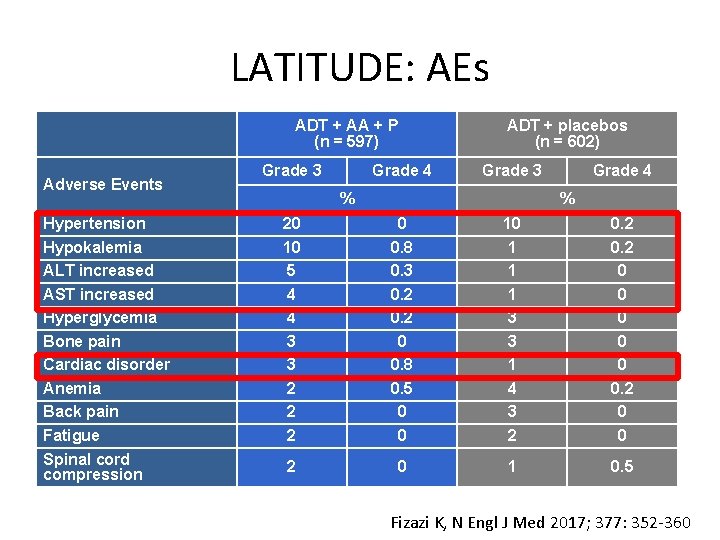
LATITUDE: AEs ADT + AA + P (n = 597) Adverse Events Hypertension Hypokalemia ALT increased AST increased Hyperglycemia Bone pain Cardiac disorder Anemia Back pain Fatigue Spinal cord compression Grade 3 Grade 4 ADT + placebos (n = 602) Grade 3 % Grade 4 % 20 10 5 4 4 3 3 2 2 2 0 0. 8 0. 3 0. 2 0 0. 8 0. 5 0 0 10 1 1 1 3 3 1 4 3 2 0 0 2 0 1 0. 5 Fizazi K, N Engl J Med 2017; 377: 352 -360

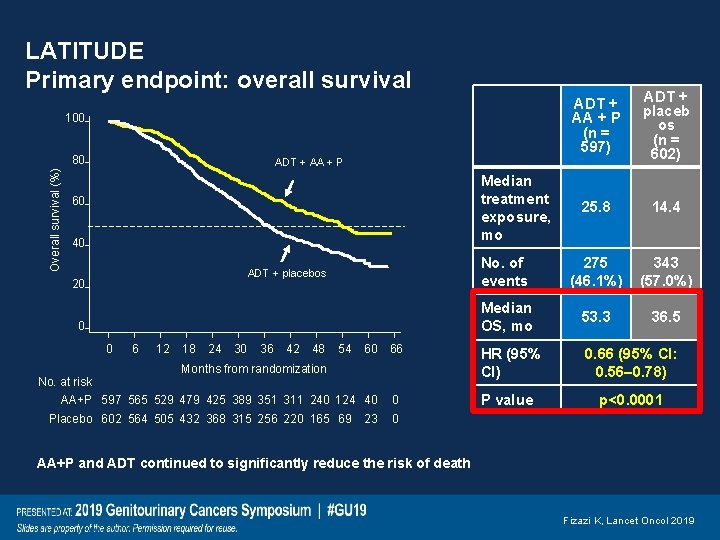
LATITUDE Primary endpoint: overall survival ADT + AA + P (n = 597) ADT + placeb os (n = 602) 25. 8 14. 4 No. of events 275 (46. 1%) 343 (57. 0%) Median OS, mo 53. 3 36. 5 66 HR (95% CI) 0. 66 (95% CI: 0. 56 0. 78) 0 P value 100 Overall survival (%) 80 ADT + AA + P Median treatment exposure, mo 60 40 ADT + placebos 20 0 0 No. at risk 6 12 18 24 30 36 42 48 54 60 Months from randomization AA+P 597 565 529 479 425 389 351 311 240 124 40 Placebo 602 564 505 432 368 315 256 220 165 69 23 p<0. 0001 0 AA+P and ADT continued to significantly reduce the risk of death Fizazi K, Lancet Oncol 2019

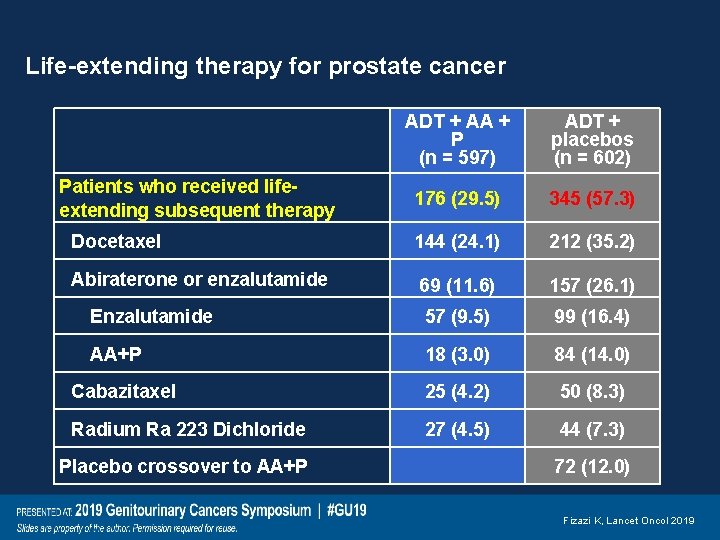
Life-extending therapy for prostate cancer ADT + AA + P (n = 597) ADT + placebos (n = 602) 176 (29. 5) 345 (57. 3) Docetaxel 144 (24. 1) 212 (35. 2) Abiraterone or enzalutamide 69 (11. 6) 157 (26. 1) Enzalutamide 57 (9. 5) 99 (16. 4) AA+P 18 (3. 0) 84 (14. 0) Cabazitaxel 25 (4. 2) 50 (8. 3) Radium Ra 223 Dichloride 27 (4. 5) 44 (7. 3) Placebo crossover to AA+P 72 (12. 0) Patients who received lifeextending subsequent therapy Fizazi K, Lancet Oncol 2019

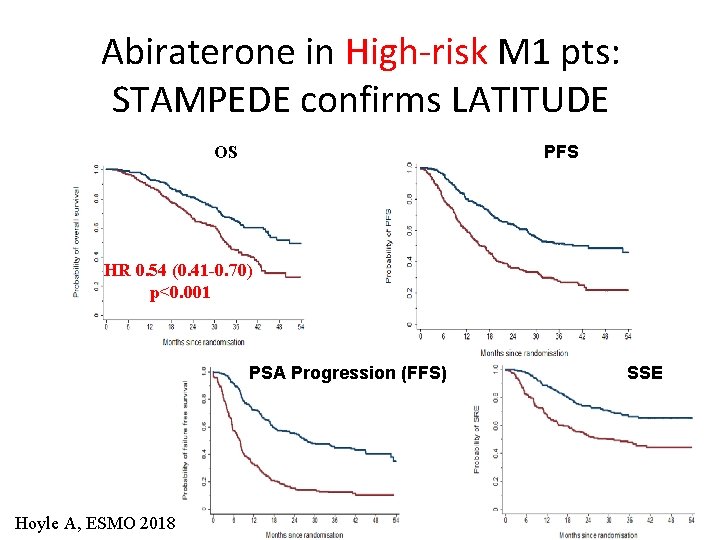
Abiraterone in High-risk M 1 pts: STAMPEDE confirms LATITUDE PFS OS HR 0. 54 (0. 41 -0. 70) p<0. 001 PSA Progression (FFS) Hoyle A, ESMO 2018 SSE

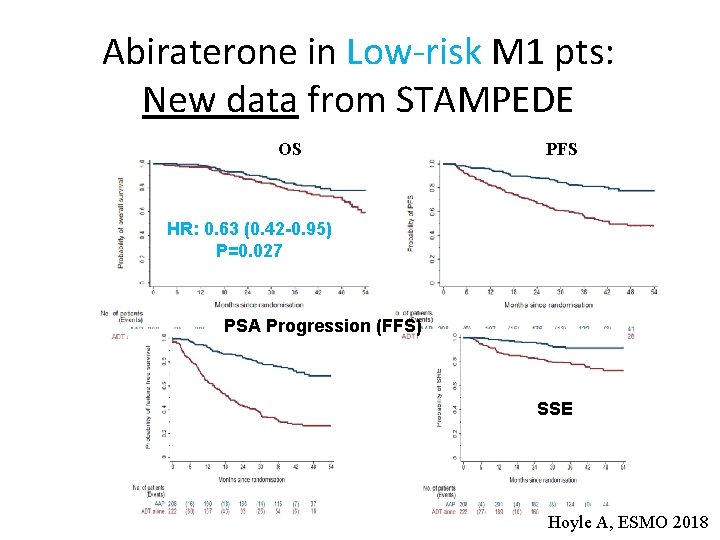
Abiraterone in Low-risk M 1 pts: New data from STAMPEDE OS PFS HR: 0. 63 (0. 42 -0. 95) P=0. 027 PSA Progression (FFS) SSE Hoyle A, ESMO 2018

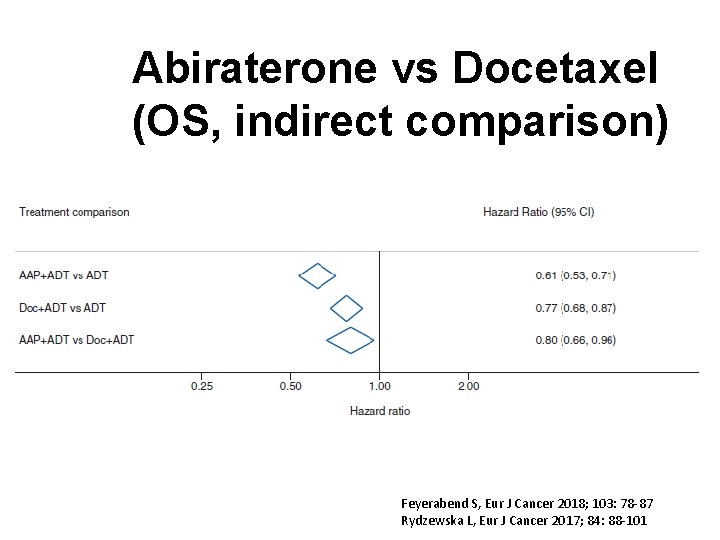
Abiraterone vs Docetaxel (OS, indirect comparison) Feyerabend S, Eur J Cancer 2018; 103: 78 -87 Rydzewska L, Eur J Cancer 2017; 84: 88 -101

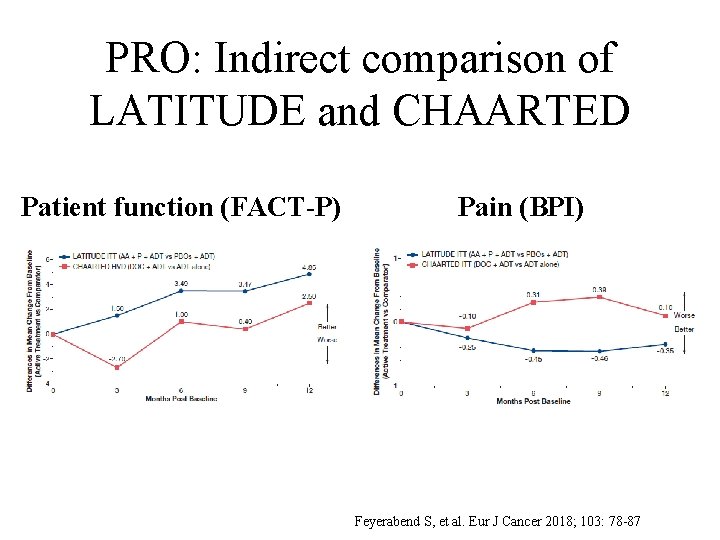
PRO: Indirect comparison of LATITUDE and CHAARTED Patient function (FACT-P) Pain (BPI) Feyerabend S, et al. Eur J Cancer 2018; 103: 78 -87
![Abiraterone vs Docetaxel STAMPEDE posthoc analysis Failurefree survival driven by PSA failure SOCAAP HR Abiraterone vs Docetaxel (STAMPEDE post-hoc analysis) Failure-free survival [driven by PSA failure] SOC+AAP HR](https://slidetodoc.com/presentation_image/801a6ddc4c2ec174f555b5c7c93b4b54/image-24.jpg)
Abiraterone vs Docetaxel (STAMPEDE post-hoc analysis) Failure-free survival [driven by PSA failure] SOC+AAP HR (95%CI) P-val SOC+Doc. P Interactn test 0. 17 M 1 0. 56 (0. 42 to 0. 75) <0. 001 Key: HR<1 favours SOC+AAP HR>1 favours SOC+Doc. P Interactn = test for interaction (heterogeneity of treatment effect) Sydes M, Ann Oncol 2018; 29: 1235 -48

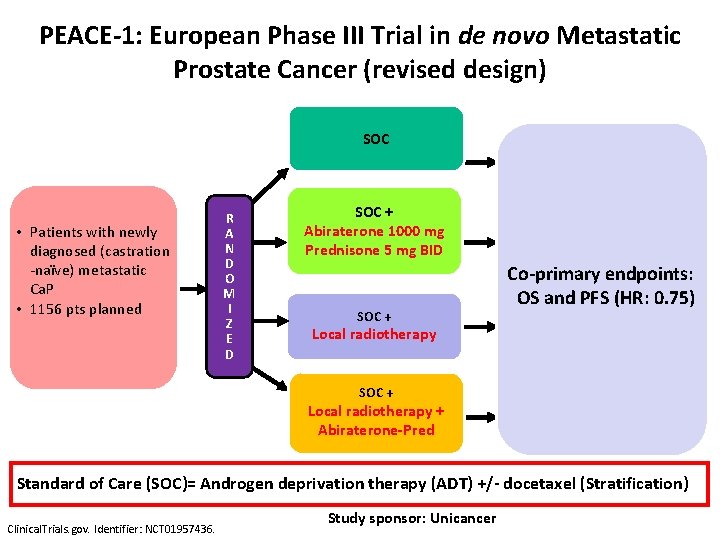
PEACE-1: European Phase III Trial in de novo Metastatic Prostate Cancer (revised design) SOC • Patients with newly diagnosed (castration -naïve) metastatic Ca. P • 1156 pts planned R A N D O M I Z E D SOC + Abiraterone 1000 mg Prednisone 5 mg BID SOC + Co-primary endpoints: OS and PFS (HR: 0. 75) Local radiotherapy SOC + Local radiotherapy + Abiraterone-Pred Standard of Care (SOC)= Androgen deprivation therapy (ADT) +/- docetaxel (Stratification) Clinical. Trials. gov. Identifier: NCT 01957436. Study sponsor: Unicancer

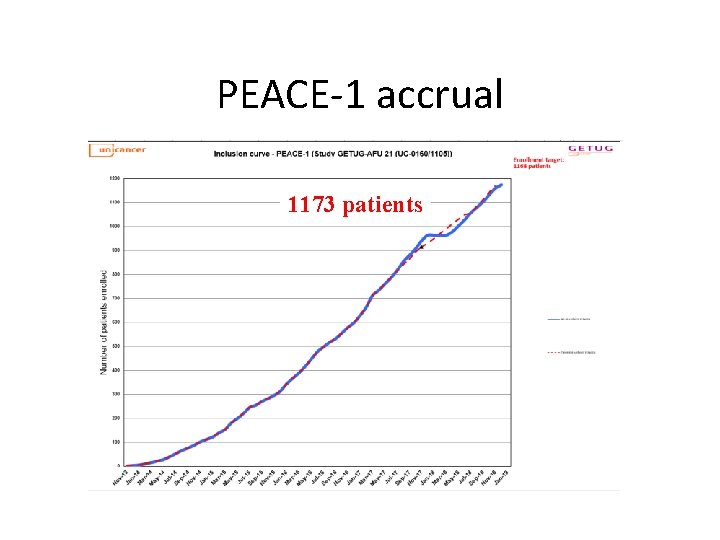
PEACE-1 accrual 1173 patients

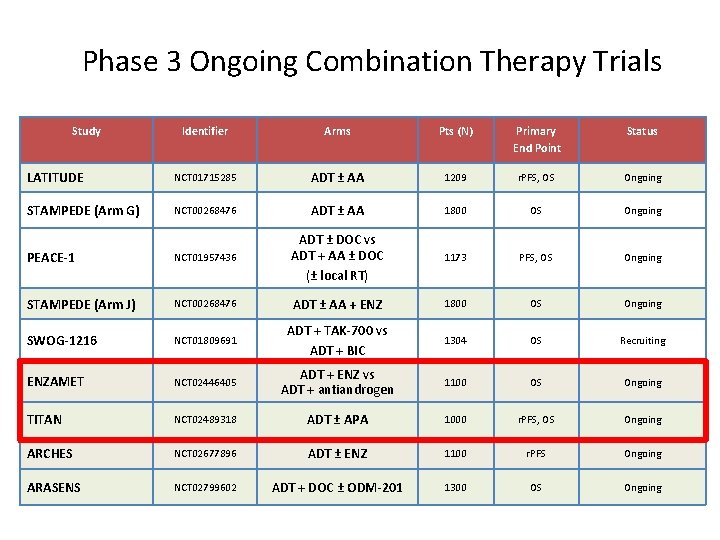
Phase 3 Ongoing Combination Therapy Trials Study Identifier Arms Pts (N) Primary End Point Status LATITUDE NCT 01715285 ADT ± AA 1209 r. PFS, OS Ongoing STAMPEDE (Arm G) NCT 00268476 ADT ± AA 1800 OS Ongoing PEACE-1 NCT 01957436 ADT ± DOC vs ADT + AA ± DOC (± local RT) 1173 PFS, OS Ongoing STAMPEDE (Arm J) NCT 00268476 ADT ± AA + ENZ 1800 OS Ongoing SWOG-1216 NCT 01809691 ADT + TAK-700 vs ADT + BIC 1304 OS Recruiting ENZAMET NCT 02446405 ADT + ENZ vs ADT + antiandrogen 1100 OS Ongoing TITAN NCT 02489318 ADT ± APA 1000 r. PFS, OS Ongoing ARCHES NCT 02677896 ADT ± ENZ 1100 r. PFS Ongoing ARASENS NCT 02799602 ADT + DOC ± ODM-201 1300 OS Ongoing 36

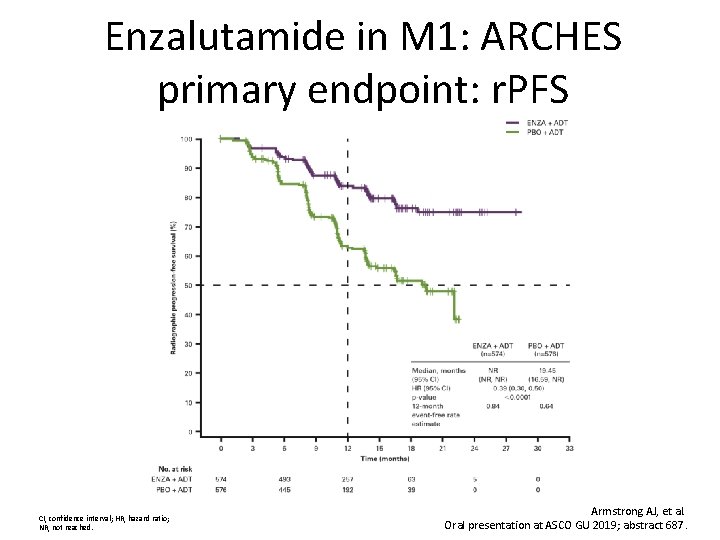
Enzalutamide in M 1: ARCHES primary endpoint: r. PFS CI, confidence interval; HR, hazard ratio; NR, not reached. Armstrong AJ, et al. Oral presentation at ASCO GU 2019; abstract 687.

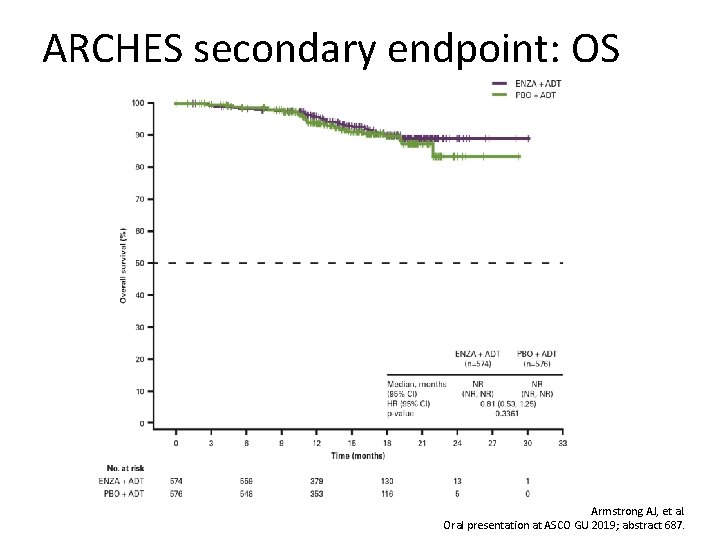
ARCHES secondary endpoint: OS Armstrong AJ, et al. Oral presentation at ASCO GU 2019; abstract 687.

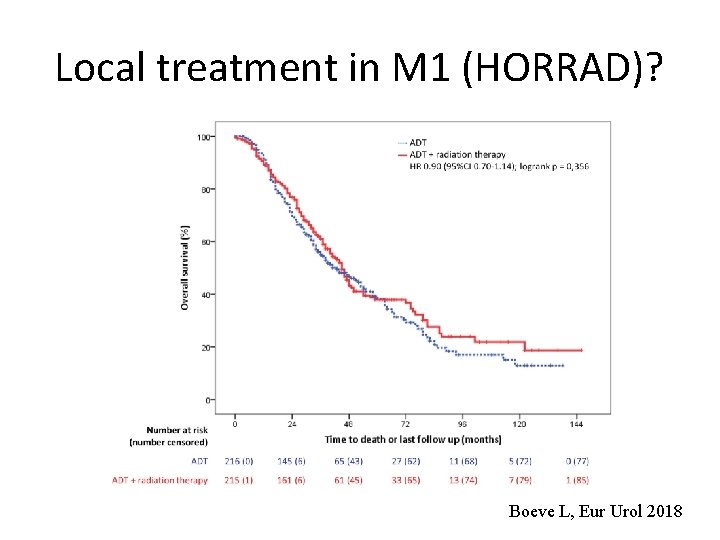
Local treatment in M 1 (HORRAD)? Boeve L, Eur Urol 2018

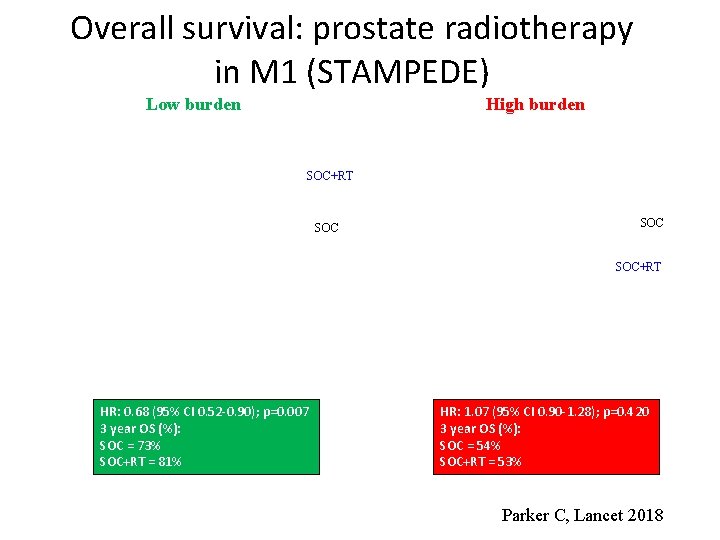
Overall survival: prostate radiotherapy in M 1 (STAMPEDE) Low burden High burden SOC+RT SOC SOC+RT HR: 0. 68 (95% CI 0. 52 -0. 90); p=0. 007 3 year OS (%): SOC = 73% SOC+RT = 81% HR: 1. 07 (95% CI 0. 90 -1. 28); p=0. 420 3 year OS (%): SOC = 54% SOC+RT = 53% Parker C, Lancet 2018

Standard of care for M 1 prostate cancer (assuming patients are fit) Standard of care before ESMO 2018 Standard of care after ESMO 2018 High risk, de novo ADT+Abiraterone ADT+Docetaxel Low risk, de novo (oligo. M 1) Debated: ADT+ Abiraterone ADT alone RXT of the prostate or ADT+Docetaxel ? Previous local Tx, low risk ? ? Previous local Tx, high risk ? ? Metastases detected on PET ? ?

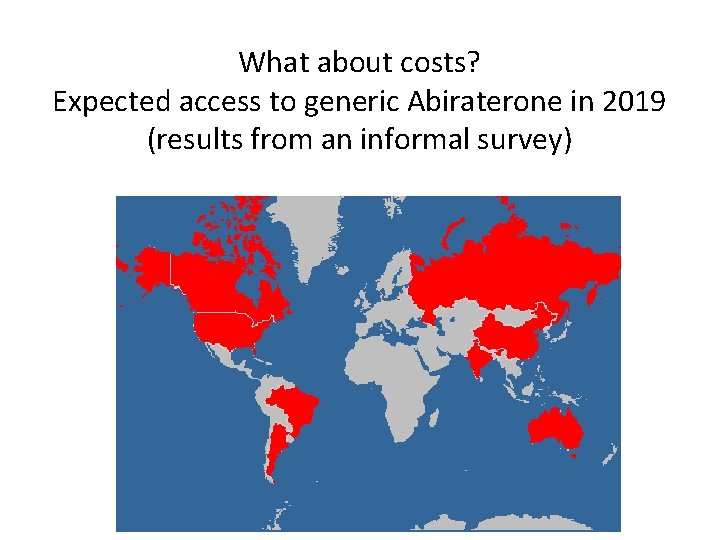
What about costs? Expected access to generic Abiraterone in 2019 (results from an informal survey)

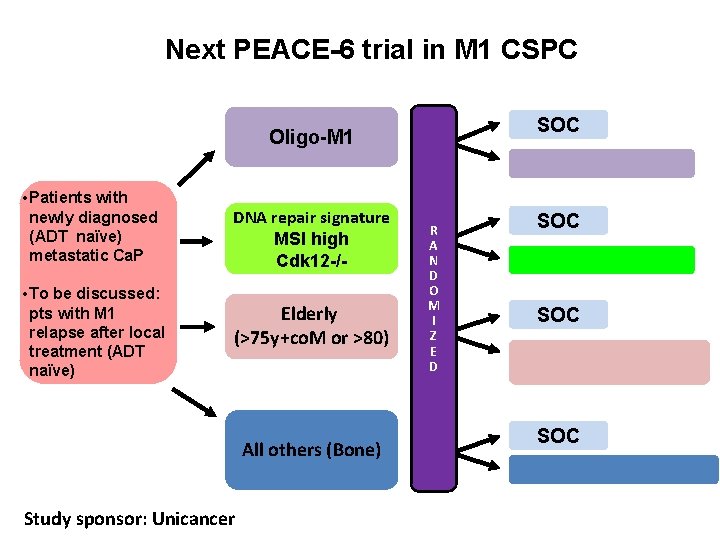
Next PEACE-6 trial in M 1 CSPC SOC Oligo-M 1 • Patients with newly diagnosed (ADT naïve) metastatic Ca. P • To be discussed: pts with M 1 relapse after local treatment (ADT naïve) DNA repair signature MSI high Cdk 12 -/- Elderly (>75 y+co. M or >80) All others (Bone) Study sponsor: Unicancer R A N D O M I Z E D SOC SOC

Conclusion • Current systemic treatment for M 1 patients: – ADT + Abiraterone – ADT + Docetaxel – ADT alone for frail/very elderly patients? • RXT to prostate for oligo-M 1 • Triplet? (ADT+Abiraterone+Docetaxel): PEACE-1 • Next-generation trial stratified on clinical/molecular parameters: PEACE-6