Navigating e IRB and e IRB Submission Types

Navigating e. IRB and e. IRB Submission Types Katie Daniels IRB Coordinator Committee C 864 -455 -4984 Kdaniels@ghs. org Amanda Goode IRB Coordinator Committees A & B 864 -455 -4360 Agoode@ghs. org

Objectives • • • Learn what is e. IRB Understand how GHS uses e. IRB Know the functions of e. IRB Understand the different types of submissions Observe a demonstration of how to submit in the e. IRB

e. IRB Overview • electronic Institutional Review Board • HSSC- Health Sciences South Carolina – Made up of 8 Institutions: 1. 2. 3. 4. 5. 6. 7. 8. GHS USC MUSC An. Med Health Palmetto Health Self-Regional Spartanburg Regional Clemson (only HSSC institution that does not use e. IRB)

How to get started in e. IRB? • Who needs an e. IRB account? – Any person participating in research activities… • • Principal Investigators Co-Investigator(s) Study Coordinator(s) Other Study Team Member(s) – Regulatory staff, project assistants, students, etc. • Guest List – Read-only access – Studies collaborating with Clemson- add Clemson IRB as guest

How to get started in e. IRB? (Cont. ) • e. IRB user account – HSSC affiliates visit: https: //eirb. healthsciencessc. org – Select affiliated institution – Complete registration fields, click ‘Register’ – 3 -5 business days to activate account

Uses for e. IRB • • Submit research proposals for IRB approval Submit Continuing Reviews Submit study Amendments/Changes Submit Reportable Events

Submitting Studies. Research vs. Quality Improvement • “Is this project a systematic investigation, including research development, testing, and evaluation, designed to develop or contribute to generalizable knowledge? ” • If Yes, Research! – i. e. , Intended to publish in journals, present results at conference; anything that leaves the institution

Submitting Studies. Human Subjects vs. Non-Human Subjects • “Does this project involve the investigator obtaining data about living individuals through 1) intervention or interaction with the individual; or 2) identifiable information? ” • If Yes, Human Subjects Research! – i. e. , prospective studies, retrospective chart reviews (with access to PHI), studies involving collection of specimens, etc.

Submitting Studies. Full Board vs. Expedited vs. Exempt • Full Board: Studies involving more than minimal risk – Committee A: Non-Pediatric/Non-Oncology – Committee B: Pediatrics, Adult Studies (not adult oncology) – Committee C: Adult Oncology • Expedited: Studies involving no more than minimal risk – IRB Chairs review • Exempt: IRB oversight not required – IRB Chairs review

Submitting Studies. In Conclusion • Submit Everything! • Best to let IRB determine if oversight is required • IRB notice of review is required for most publications and/or conference presentations, even if research is non-human subjects, exempt, etc

Submitting Studies. What will I Need? • New Study Submittal Checklist – Investigator Signature Page – Consent Forms – Protocol – Data collection forms, surveys, questionnaires, etc. – CV of PI – CITI Training • For full list, visit e. IRB website: https: //hsc. ghs. org/eirb/forms/
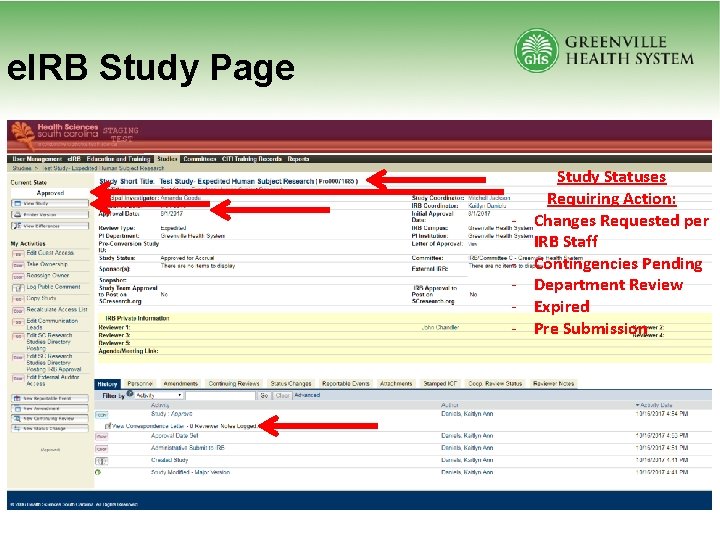
e. IRB Study Page - Study Statuses Requiring Action: Changes Requested per IRB Staff Contingencies Pending Department Review Expired Pre Submission
Continuing Reviews

Continuing Reviews • All studies require IRB review no less than once per year (i. e. , 364 days or 6 months) – Current Study Status – Number of Subjects – Subjects’ Demographic Information – Interim Findings – Redacted Last Signed Consent Form – For pediatric studies: updated pediatric risk grading form – Updated COI information
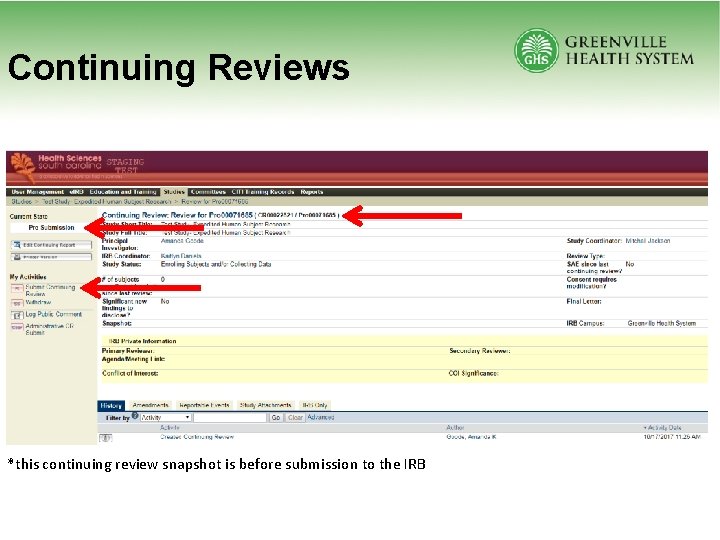
Continuing Reviews *this continuing review snapshot is before submission to the IRB
Amendments

Amendments • Submit Amendments whenever there are changes to: – – – – – Study Personnel Advertisements/Study Recruitment material Informed Consent Documents Protocol Documents Investigator’s Brochures Editorial/Administrative Changes Additional Sites for Treatment/Follow-up Modification in Subject Enrollment Goals Location of Program Activities Questionnaires & Assessment Tools Risk Change(s) Subject Confidentiality/Anonymity Subject Population HIPPA Authorization Study Funding Source/ Sponsorship Conflict of Interest (COI) Other Changes, etc.

Amendments • Amendments are reviewed based on: – Risk – The initial study submission review type • Amendment Requirements – Risk change assessment – Description of what is changing – Update changes in Smart. Form
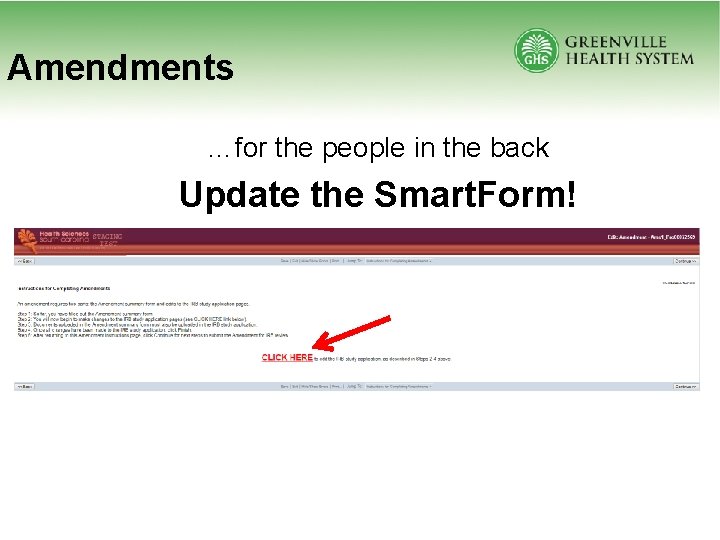
Amendments …for the people in the back Update the Smart. Form!
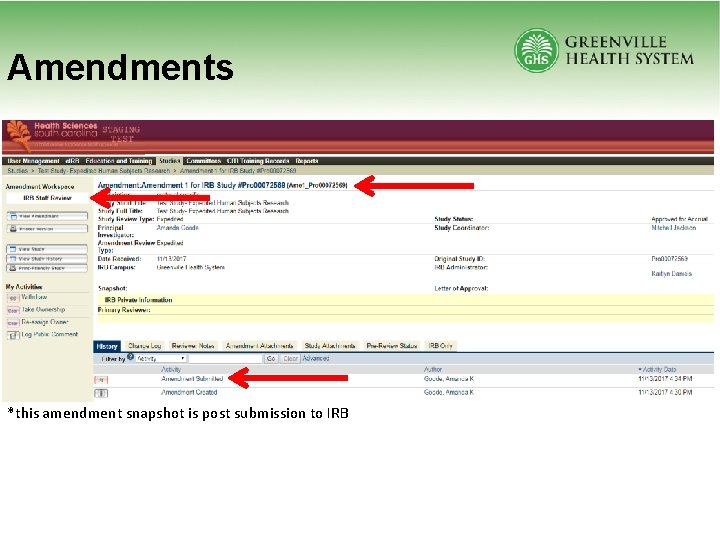
Amendments *this amendment snapshot is post submission to IRB
Reportable Events

Reportable Events • Unanticipated Problem – Unexpected – Related or possibly related – Change in known or recognized risk • Adverse Event – undesirable/unintended , although typically not unexpected, result of therapy or intervention • Protocol Deviations • Other Reports/Events

Submitting Reportable Events • Type of reportable event • Multi-study reportable event • Internal vs. External event – If only local study, always Internal – External AEs should only be reported to local IRB when event requires changes to study protocol, IB, or Informed Consent • Specific event questions
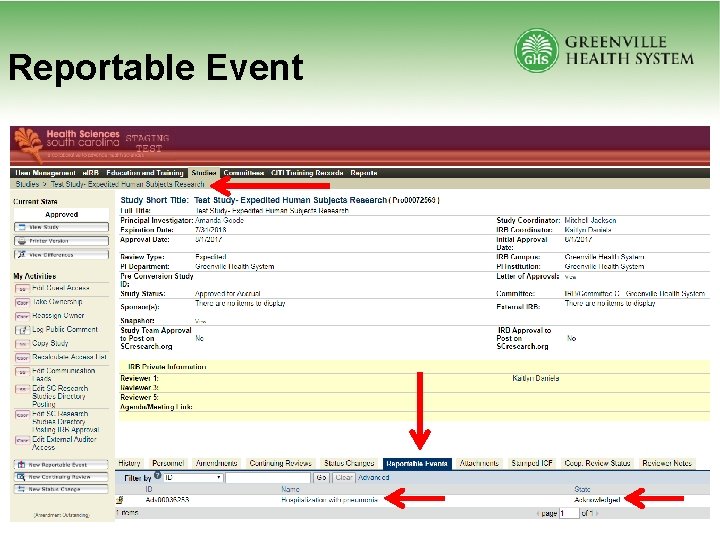
Reportable Event

e. IRB Demo http: //eirb. healthsciencessc. org

Contact Us Office of Human Research Protection (OHRP) 701 Grove Road- Employee Services Center (ESC) Greenville, SC 29605 864 -455 -8997 Matt Hudson, Ph. D OHRP Director MHudson 2@ghs. org Chris Wright, MD OHRP Medical Director CWright 2@ghs. org Katie Daniels IRB Coordinator Committee C 864 -455 -4984 Kdaniels@ghs. org Amanda Goode IRB Coordinator Committees A & B 864 -455 -4360 Agoode@ghs. org

- Slides: 27