NATURAL RUBBER Natural Polymer Monomer Natural Rubber Polyisoprene

NATURAL RUBBER Natural Polymer Monomer Natural Rubber (Polyisoprene) Isoprene Starch Glucose Cellulose Glucose Protein Amino acid Faridah Hamat Saser 2010 1

Natural Rubber Monomer : Isoprene or 2 -methylbuta-1, 3 -diene Structure Formula: Faridah Hamat Saser 2010 2

Coagulation process Rubber molecule - - - Rubber particle - Negatively charged protein membrane The repulsion between the negative-charged particles prevents the rubber particles from coming close to each other. Hence, latex could not coagulate Faridah Hamat Saser 2010 3

When acid is added; • The hydrogen ions, H+ neutralize the negativecharges on the protein membrane • The rubber particles can now close together • This enables them to collide with one another • Resulting in the breakage of the protein membrane • The rubber molecules combine with one another and entangle • Causing the latex to coagulate Faridah Hamat Saser 2010 4

Latex will coagulate if left after 2 or 3 hours (without acid is added) because; • Activities (growth) of Bacteria on the protein membrane produce lactic acid • Lactic acid contains hydrogen ions, H+ • Hydrogen ion neutralize the negative charges on the protein membrane • Allow the coagulation to occur Faridah Hamat Saser 2010 5

Prevent Coagulation Process When Alkali (NH 3 (aq) ) is added; • Ammonia aqueous contains hydroxide ion, OH • Hydroxide ion can neutralize the acid produced by the bacteria • Hence, the rubber particles remain negatively charged and the coagulation is prevented Faridah Hamat Saser 2010 6

Properties of natural rubber • Elastic Stretched released • Unstable to heat • When heated above 50 o. C, its softens and becomes sticky Faridah Hamat Saser 2010 7

Unstable to oxidation – Because the presence of double bonds between carbon atoms – Oxygen from air with UV radiation breaks up the polymers chains Insoluble in water - Cannot dissolve in water - Soluble in organic solvents Faridah Hamat Saser 2010 8

Vulcanisation of Rubber 1 – 3 % by weight of sulphur is added to raw rubber and the mixture is carefully heated In Laboratory; Strip of rubber sheet is dipped into a solution of disulphur dichloride for about 5 minutes Faridah Hamat Saser 2010 9
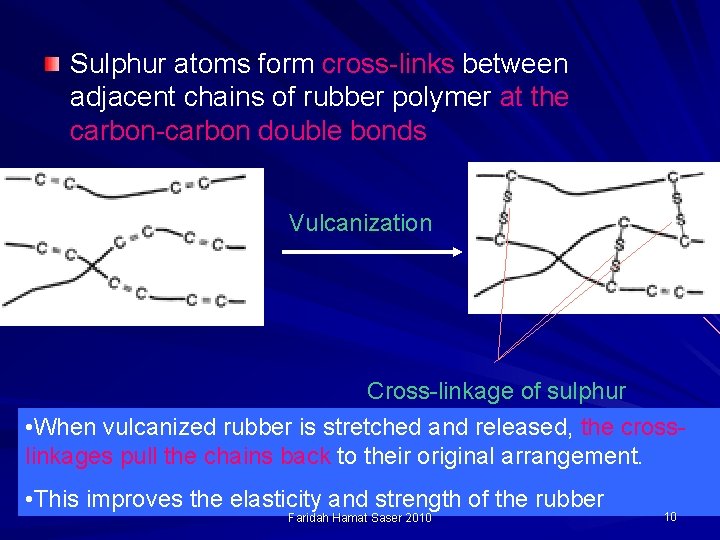
Sulphur atoms form cross-links between adjacent chains of rubber polymer at the carbon-carbon double bonds Vulcanization Cross-linkage of sulphur • When vulcanized rubber is stretched and released, the crosslinkages pull the chains back to their original arrangement. • This improves the elasticity and strength of the rubber Faridah Hamat Saser 2010 10
- Slides: 10