Native Elements Sulfides and Sulfosalts GLY 4200 Lab

Native Elements, Sulfides, and Sulfosalts GLY 4200 Lab 2 - Fall, 2019 © D. L. Warburton 2019 1

Native Elements • The native elements may be divided into metallic, non-metallic, and semi-metallic • Native elements are composed of a single element, uncombined with other elements • They are often ores, especially the metallic native elements § Gold, Silver, Copper, Platinum 2

Native Element Luster • The luster shows distinctive changes from one group to another § Metallic group – metallic to dull metallic § Semi-metal group – submetallic to dull metallic § Non-metallic – various non-metallic lusters 3

Sulfides • A sulfide is a compound between sulfur, or another sulfur group element, and a metallic element • Most of these minerals are opaque, and they often have characteristic colors, as well as distinctive streak colors • A few are transparent to translucent – examples cinnabar, realgar, and orpiment 4

Bismuth • Name officially comes from the German Weisse Masse (Eng. white mass) • Older (circa 1400’s) descriptions mention bismutum, apparently from the Ger. “wissmut” because the mineral was extracted (gemutet) in the fields (in den Weisen) • Another German word Weissmuth = white material 5

Sulfide Formulas • The general formula is Am. Xn where A represents metallic elements, X represents a sulfide group element whose oxidation state is -2, and m and n are integers • The X anions are usually in the negative two oxidation state § Exception: Minerals with the S 2 dimer, such as pyrite, marcasite, and arsenopyrite 6

Sulfide Formulas continued • X can be § § § Sulfur Selenium Tellurium Arsenic Antimony 7
Position in Periodic Table 8

Other Oxidation States • All of the sulfide group elements occur in other oxidation states, for example As 5+ forms arsenates • But in the 2 - state they belong to the sulfide group 9

Anion Size • All of the sulfide group anions are large • The large anionic size tends to favor coordination with large cations § As a result sulfide minerals are very often important economic minerals (ores) because they associate with the relatively rare larger cations 10
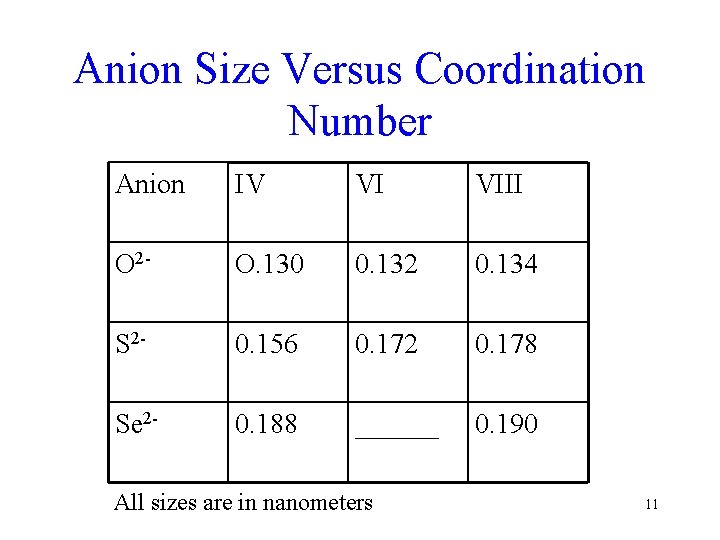
Anion Size Versus Coordination Number Anion IV VI VIII O 2 - O. 130 0. 132 0. 134 S 2 - 0. 156 0. 172 0. 178 Se 2 - 0. 188 ______ 0. 190 All sizes are in nanometers 11
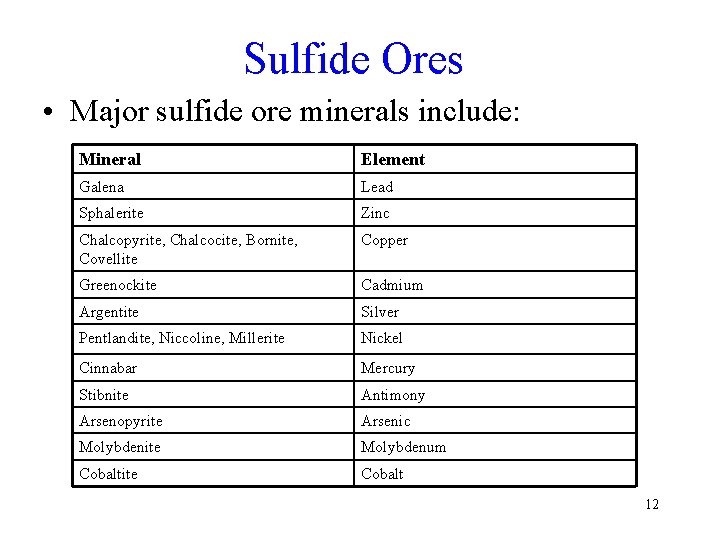
Sulfide Ores • Major sulfide ore minerals include: Mineral Element Galena Lead Sphalerite Zinc Chalcopyrite, Chalcocite, Bornite, Covellite Copper Greenockite Cadmium Argentite Silver Pentlandite, Niccoline, Millerite Nickel Cinnabar Mercury Stibnite Antimony Arsenopyrite Arsenic Molybdenite Molybdenum Cobaltite Cobalt 12

Solid Solution • The sulfides are also characterized by extensive solid solutions • Many of the cations are of similar size and charge and they can substitute extensively • Anionic substitution also occurs • The physical characteristics of solid solutions will be between their end-member minerals 13

Solid Solution Example • Galena is Pb. S, the only ore of lead • Galena is found with inclusions of other elements Ag, Zn, Cd, Sb, As & Bi • Se can substitute for S and forms a complete solid solution series 14

Sulfide Characteristics • Many sulfide minerals have resonant bonding, with a partial metallic characteristic § Results in metallic to dull metallic luster • SG is often high, because of the presence of metallic elements, and sometimes heavy anions like Se or Te 15

Sulfide Hardness • Most sulfide group minerals are soft to softmoderate hardness § Exception: Sulfides with the S 2 dimer, such as pyrite, marcasite, and arsenopyrite, are moderately hard 16

Sulfosalts • This term signifies a mineral in which a semi metallic element replaces the metals § Examples: enargite, tetrahedrite 17

Text Reference • See chapter 15 in the text for more information on native elements, sulfides, and sulfosalts, pp. 331 -367 18
- Slides: 18