Nanoscale Engineered Plasmonic Nanostructures for Biosensing and Bioimaging

Nanoscale Engineered Plasmonic Nanostructures for Biosensing and Bioimaging Dr Fang Xie Department of Materials Imperial College London 7 th Asia Pacific Biotech Congress 13 th July 2015, Beijing, China

Imperial College London Department of Materials
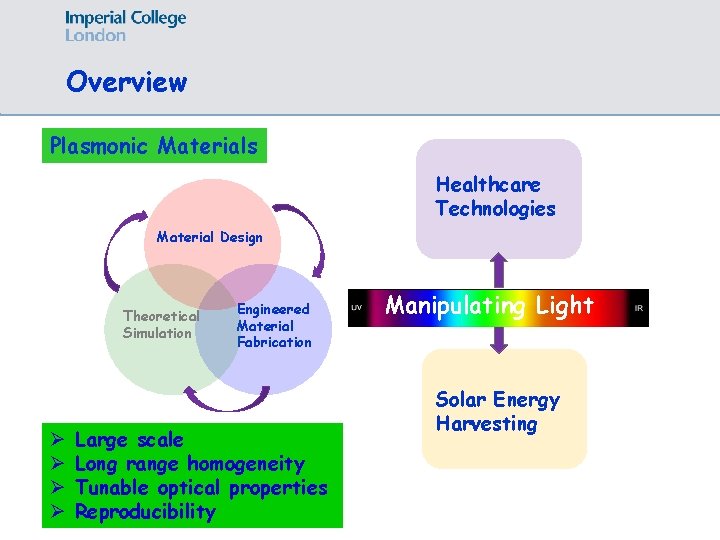
Overview Plasmonic Materials Healthcare Technologies Material Design Theoretical Simulation Ø Ø Engineered Material Fabrication Large scale Long range homogeneity Tunable optical properties Reproducibility Manipulating Light Solar Energy Harvesting
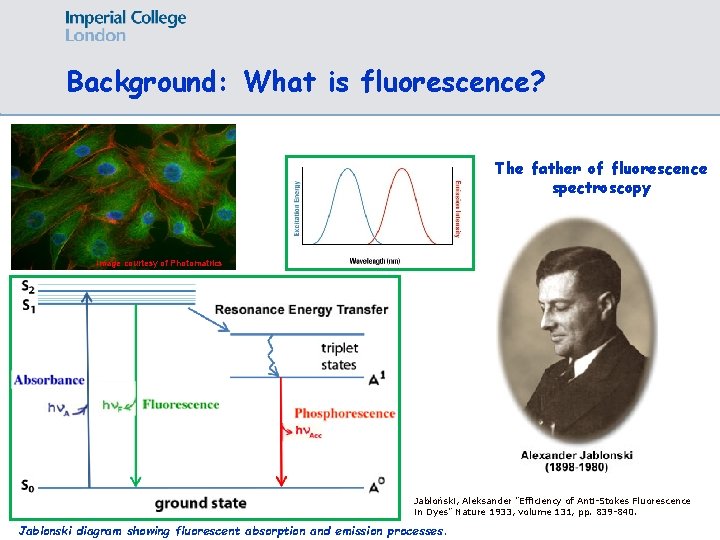
Background: What is fluorescence? The father of fluorescence spectroscopy Image courtesy of Photomatrics Jabłoński, Aleksander "Efficiency of Anti-Stokes Fluorescence in Dyes" Nature 1933, volume 131, pp. 839 -840. Jablonski diagram showing fluorescent absorption and emission processes.

Background: What is plasmonic materials? The optical properties of Nanoparticles—localized surface plasmon resonances (LSPRs): Ø Oscillations of the conduction electrons coupled to the E -field Ø The frequency and intensity of the oscillations are sensitive to the geometry and surrounding media Lycurgus Cup Reflection Adv. Mater. 2007, 19, 3771– 3782 Transmission The artwork was crafted from glass stained with colloidal noble metal particles in the 4 th Century; their strong interaction with visible light due to the excitation of LSPRs, gives rise to the vibrant green and red colours.
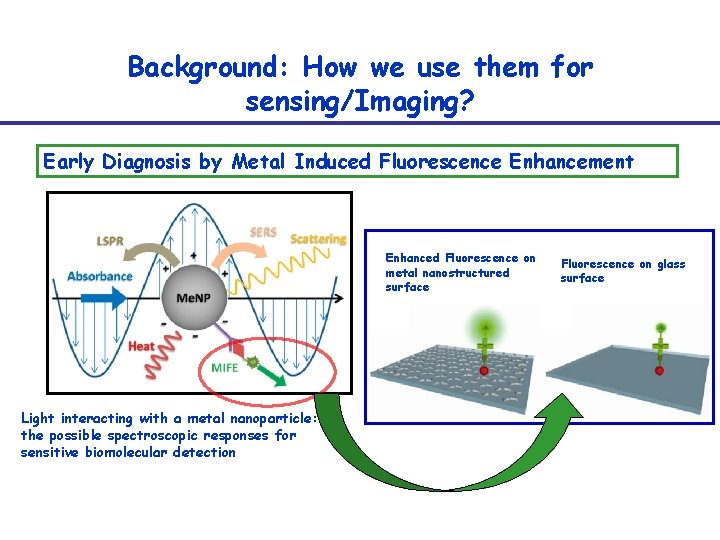
Background: How we use them for sensing/Imaging? Early Diagnosis by Metal Induced Fluorescence Enhancement Enhanced Fluorescence on metal nanostructured surface Light interacting with a metal nanoparticle: the possible spectroscopic responses for sensitive biomolecular detection Fluorescence on glass surface
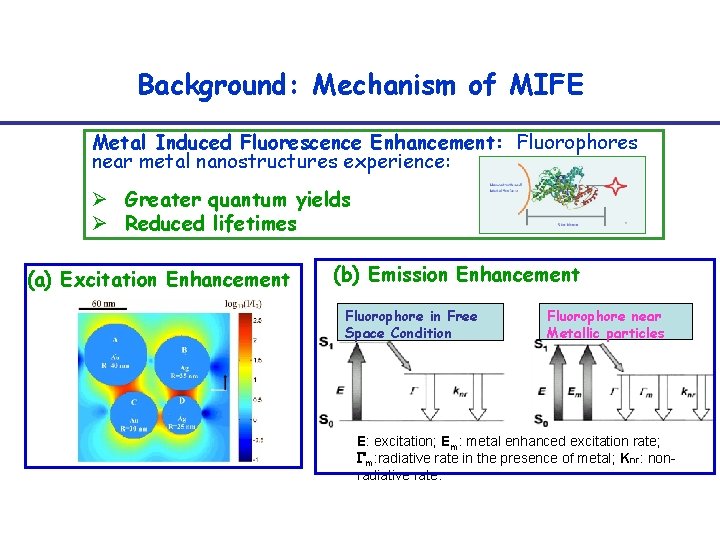
Background: Mechanism of MIFE Metal Induced Fluorescence Enhancement: Fluorophores near metal nanostructures experience: Ø Greater quantum yields Ø Reduced lifetimes (a) Excitation Enhancement (b) Emission Enhancement Fluorophore in Free Space Condition Fluorophore near Metallic particles E: excitation; Em: metal enhanced excitation rate; m: radiative rate in the presence of metal; Knr: nonradiative rate.
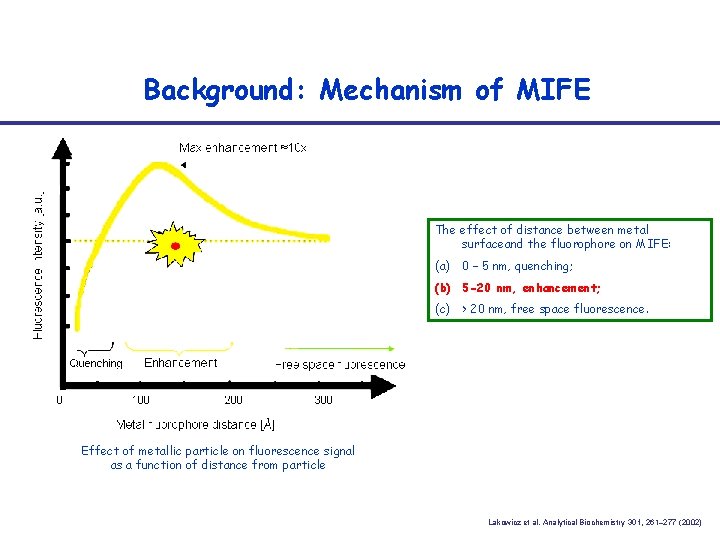
Background: Mechanism of MIFE The effect of distance between metal surfaceand the fluorophore on MIFE: (a) 0 – 5 nm, quenching; (b) 5 -20 nm, enhancement; (c) > 20 nm, free space fluorescence. Effect of metallic particle on fluorescence signal as a function of distance from particle Lakowicz et al. Analytical Biochemistry 301, 261– 277 (2002)

Nanostructures/Nanoparticles Fabrication/Synthesis

Nanostructures by Nanosphere Lithography Substrate Polystyrene Monolayer Metal Deposition Physicochem. Eng. Aspects 219 (2003) 1 /6 Polystyrene Removed
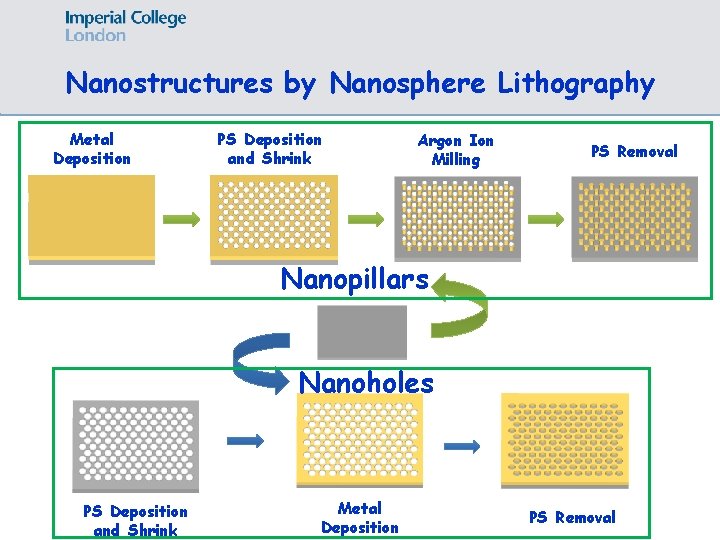
Nanostructures by Nanosphere Lithography Metal Deposition PS Deposition and Shrink Argon Ion Milling PS Removal Nanopillars Nanoholes PS Deposition and Shrink Metal Deposition PS Removal

Nanostructures by Nanosphere Lithography

Nanostructures by Synthesis

Plasmonic Materials for Fluorescence Enhancement Investigation
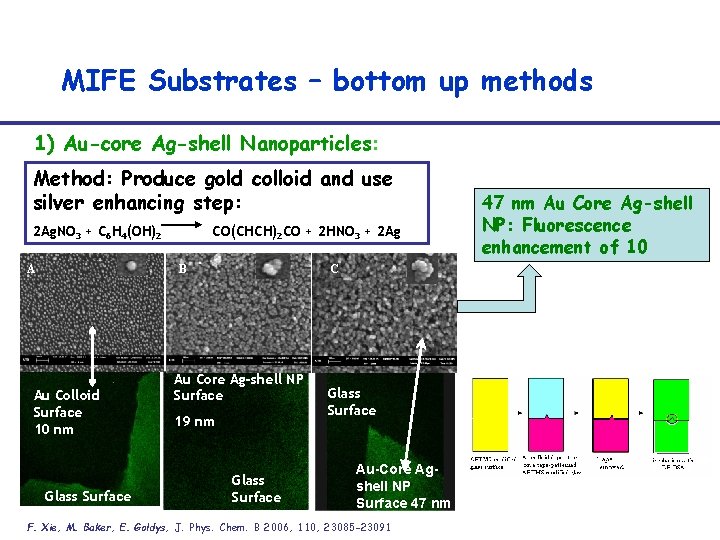
MIFE Substrates – bottom up methods 1) Au-core Ag-shell Nanoparticles: Method: Produce gold colloid and use silver enhancing step: 2 Ag. NO 3 + C 6 H 4(OH)2 A CO(CHCH)2 CO + 2 HNO 3 + 2 Ag B Au Colloid Surface 10 nm Glass Surface C Au Core Ag-shell NP Surface 19 nm Glass Surface Au-Core Agshell NP Surface 47 nm F. Xie, M. Baker, E. Goldys, J. Phys. Chem. B 2006, 110, 23085 -23091 47 nm Au Core Ag-shell NP: Fluorescence enhancement of 10
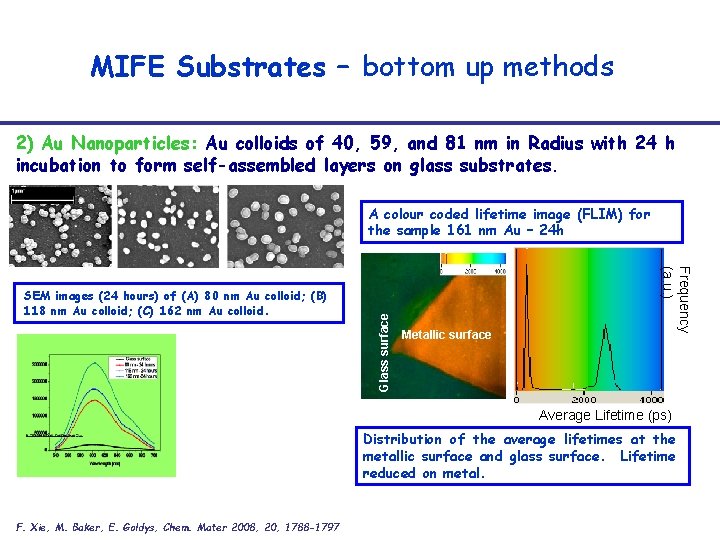
MIFE Substrates – bottom up methods 2) Au Nanoparticles: Au colloids of 40, 59, and 81 nm in Radius with 24 h incubation to form self-assembled layers on glass substrates. Metallic surface Frequency (a. u. ) SEM images (24 hours) of (A) 80 nm Au colloid; (B) 118 nm Au colloid; (C) 162 nm Au colloid. Glass surface A colour coded lifetime image (FLIM) for the sample 161 nm Au – 24 h Average Lifetime (ps) Distribution of the average lifetimes at the metallic surface and glass surface. Lifetime reduced on metal. F. Xie, M. Baker, E. Goldys, Chem. Mater 2008, 20, 1788 -1797
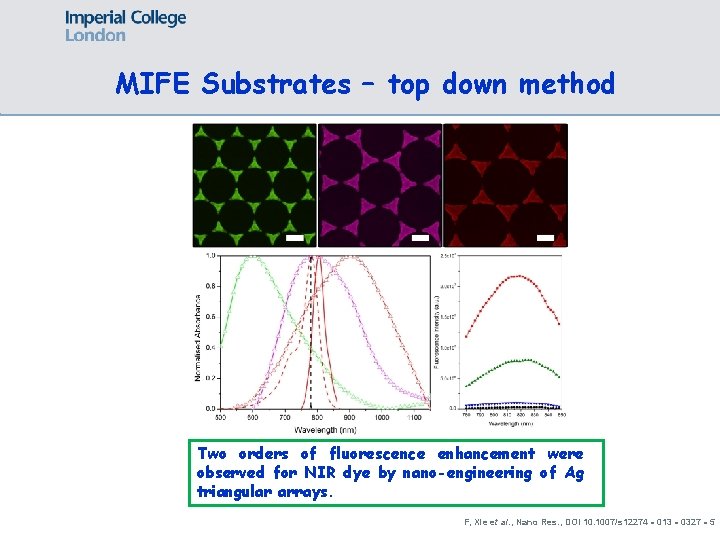
MIFE Substrates – top down method Two orders of fluorescence enhancement were observed for NIR dye by nano-engineering of Ag triangular arrays. F, Xie et al. , Nano Res. , DOI 10. 1007/s 12274‐ 013‐ 0327‐ 5
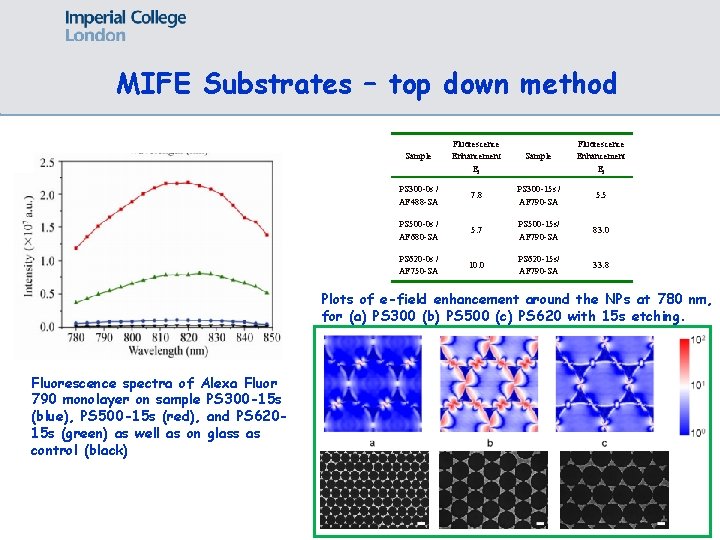
MIFE Substrates – top down method Sample Fluorescence Enhancement Ef PS 300 -0 s / AF 488 -SA 7. 8 PS 300 -15 s / AF 790 -SA 5. 5 PS 500 -0 s / AF 680 -SA 5. 7 PS 500 -15 s/ AF 790 -SA 83. 0 PS 620 -0 s / AF 750 -SA 10. 0 PS 620 -15 s/ AF 790 -SA 33. 8 Plots of e-field enhancement around the NPs at 780 nm, for (a) PS 300 (b) PS 500 (c) PS 620 with 15 s etching. Fluorescence spectra of Alexa Fluor 790 monolayer on sample PS 300 -15 s (blue), PS 500 -15 s (red), and PS 62015 s (green) as well as on glass as control (black)
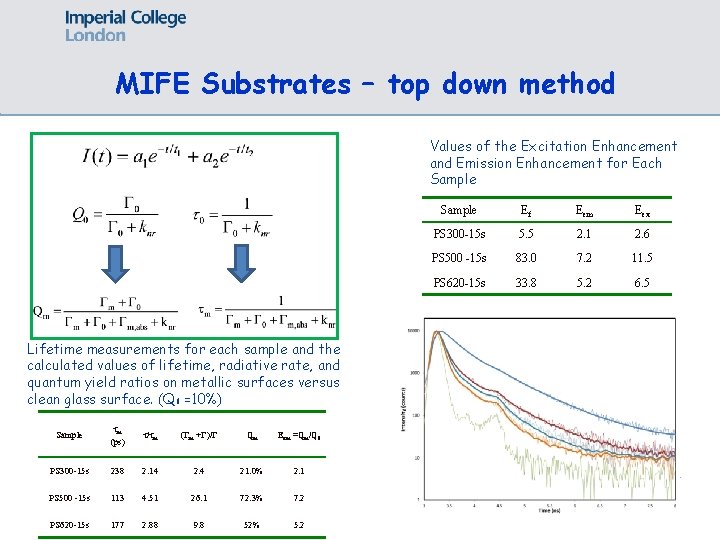
MIFE Substrates – top down method Values of the Excitation Enhancement and Emission Enhancement for Each Sample Lifetime measurements for each sample and the calculated values of lifetime, radiative rate, and quantum yield ratios on metallic surfaces versus clean glass surface. (Q 0 =10%) Sample m (ps) / m ( m + )/ Qm Eem =Qm/Q 0 PS 300 -15 s 238 2. 14 2. 4 21. 0% 2. 1 PS 500 -15 s 113 4. 51 26. 1 72. 3% 7. 2 PS 620 -15 s 177 2. 88 9. 8 52% 5. 2 Sample Ef Eem Eex PS 300 -15 s 5. 5 2. 1 2. 6 PS 500 -15 s 83. 0 7. 2 11. 5 PS 620 -15 s 33. 8 5. 2 6. 5

Plasmonic Materials for Protein Microarray
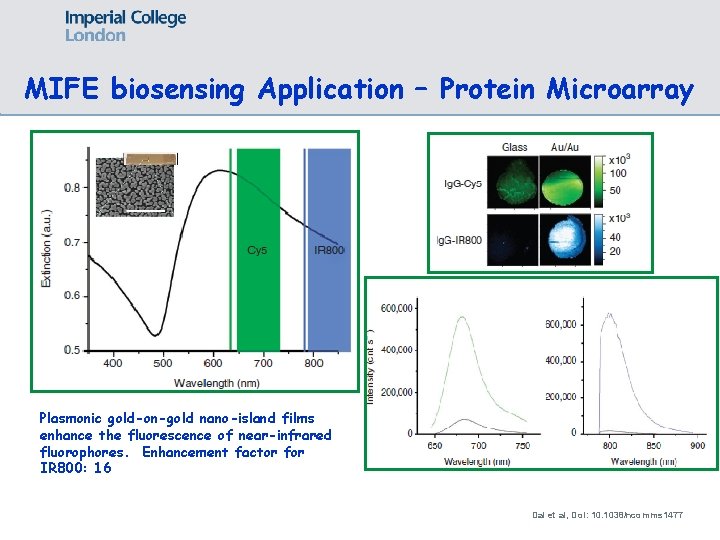
MIFE biosensing Application – Protein Microarray Plasmonic gold-on-gold nano-island films enhance the fluorescence of near-infrared fluorophores. Enhancement factor for IR 800: 16 Dai et al, Do. I: 10. 1038/ncomms 1477
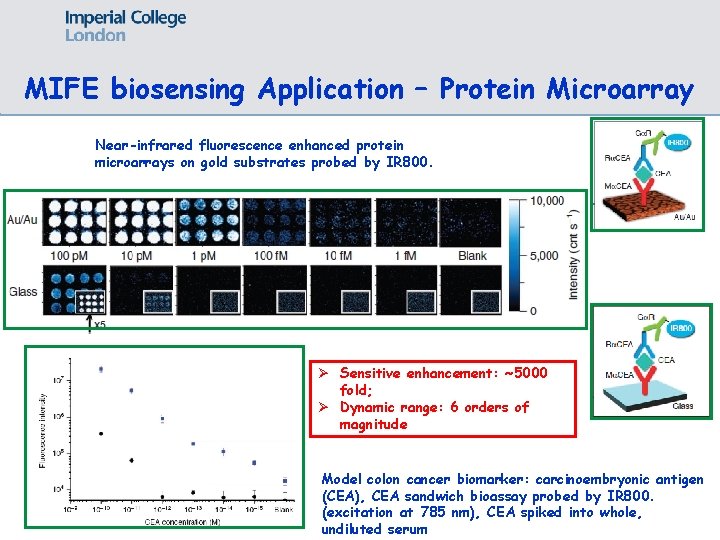
MIFE biosensing Application – Protein Microarray Near-infrared fluorescence enhanced protein microarrays on gold substrates probed by IR 800. Ø Sensitive enhancement: ~5000 fold; Ø Dynamic range: 6 orders of magnitude Model colon cancer biomarker: carcinoembryonic antigen (CEA), CEA sandwich bioassay probed by IR 800. (excitation at 785 nm), CEA spiked into whole, undiluted serum
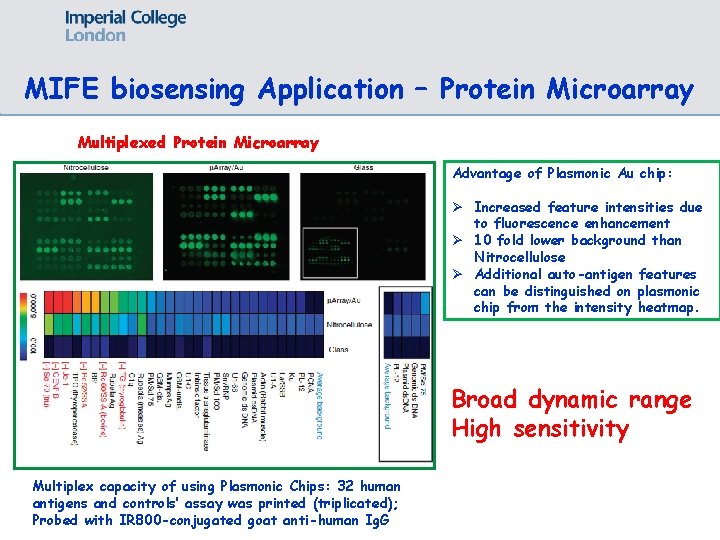
MIFE biosensing Application – Protein Microarray Multiplexed Protein Microarray Advantage of Plasmonic Au chip: Ø Increased feature intensities due to fluorescence enhancement Ø 10 fold lower background than Nitrocellulose Ø Additional auto-antigen features can be distinguished on plasmonic chip from the intensity heatmap. Broad dynamic range High sensitivity Multiplex capacity of using Plasmonic Chips: 32 human antigens and controls’ assay was printed (triplicated); Probed with IR 800 -conjugated goat anti-human Ig. G

Plasmonic Materials for in vitro Bioimaging
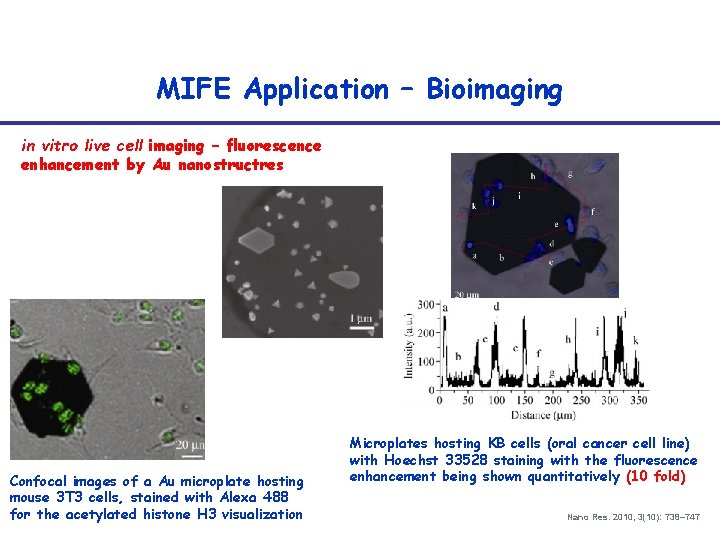
MIFE Application – Bioimaging in vitro live cell imaging – fluorescence enhancement by Au nanostructres Confocal images of a Au microplate hosting mouse 3 T 3 cells, stained with Alexa 488 for the acetylated histone H 3 visualization Microplates hosting KB cells (oral cancer cell line) with Hoechst 33528 staining with the fluorescence enhancement being shown quantitatively (10 fold) Nano Res. 2010, 3(10): 738– 747
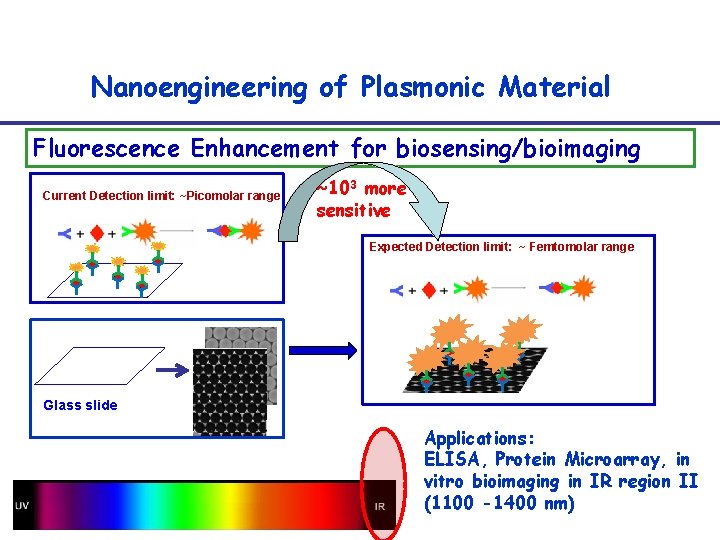
Nanoengineering of Plasmonic Material Fluorescence Enhancement for biosensing/bioimaging Current Detection limit: ~Picomolar range ~103 more sensitive Expected Detection limit: ~ Femtomolar range Glass slide Applications: ELISA, Protein Microarray, in vitro bioimaging in IR region II (1100 -1400 nm)
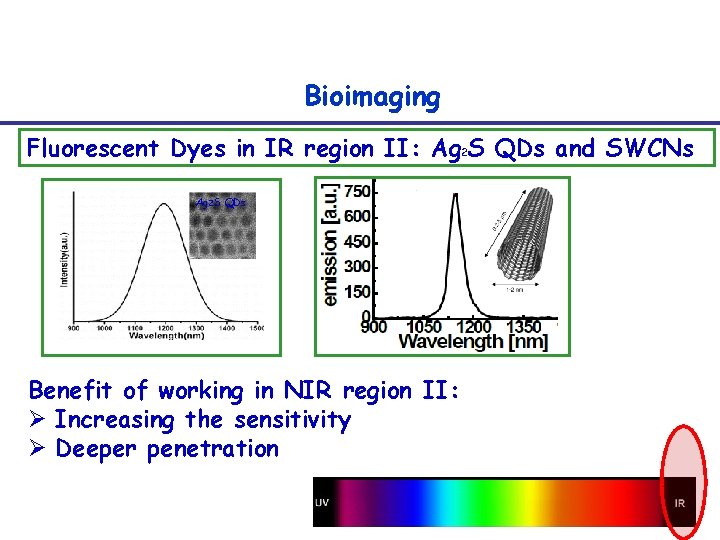
Bioimaging Fluorescent Dyes in IR region II: Ag 2 S QDs and SWCNs Ag 2 S QDs Benefit of working in NIR region II: Ø Increasing the sensitivity Ø Deeper penetration
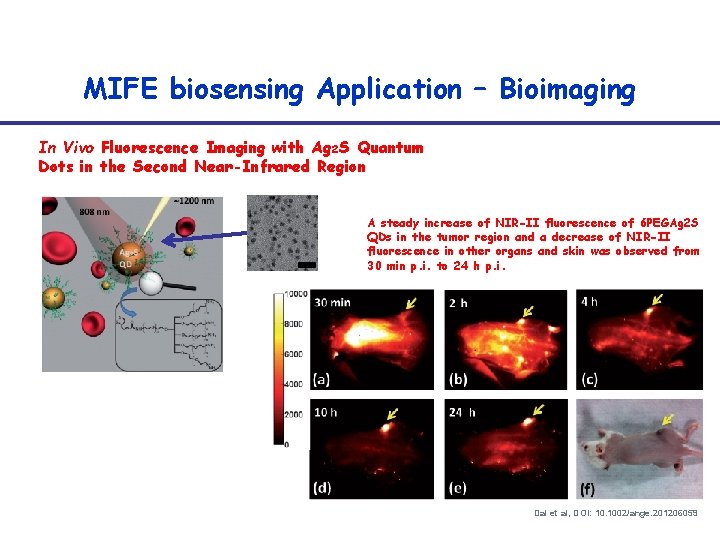
MIFE biosensing Application – Bioimaging In Vivo Fluorescence Imaging with Ag 2 S Quantum Dots in the Second Near-Infrared Region A steady increase of NIR-II fluorescence of 6 PEGAg 2 S QDs in the tumor region and a decrease of NIR-II fluorescence in other organs and skin was observed from 30 min p. i. to 24 h p. i. Dai et al, DOI: 10. 1002/ange. 201206059

MIFE technology – potential applications Plasmonic enhanced bioimaging using super bright NIR II probes? Plasmonic enhanced bioimaging and therapy ? ? Clinical questions where MEF can help? ?

Nanostructures by Nanosphere Lithography Sensing and in vitro bioimaging

Nanostructures by Synthesis Superbright in vivo bioimaging probes


Acknowledgement Collaborators The research areas include electromagnetic modeling, light harvesting, bioengineering, synthetic biology, biomaterials, bioimaging, and biosensing.

Acknowledgement Team members Ioannis Theodore (Postdoc); Jing Pang (Ph. D student); Daniel Price (Ph. D student); Heng Qin (Ph. D student); Zaynab Jaward (Ph. D student); Danyang Wang (MSc); Justin Lim (MEng); Amed Shamso (MSc)

Thank you
- Slides: 35