Nanofabrication H Hau Wang Argonne National Laboratory Materials

Nanofabrication H. Hau Wang Argonne National Laboratory Materials Science Division hau. wang@anl. gov Symposium on Digital Fabrication Aug 12, 2005 Argonne National Laboratory is managed by The University of Chicago for the U. S. Department of Energy

Why going Nano? 1 nm = 10 -9 m n Chemistry – higher reactivity High surface area – Good for chemical and biochemical sensors Higher sensitivity and faster response time Good for heterogeneous catalysis The larger the surface area, the higher the reactivity. High level of reaction control n Material – better mechanical strength UNCD Ultra-nano-crystalline diamond (ANL/CNM) Ultra-thin and tough coating - prolong equipment life time Carbon-nanotubes – Ultra-high mechanical strength n Physics – quantum confined phenomena and designed physical properties Quantum dots, Near field optics, plasmonics High density magnetic storage – nanoclusters Ideal spring nanomagnet - High magnetization and coercivity 2
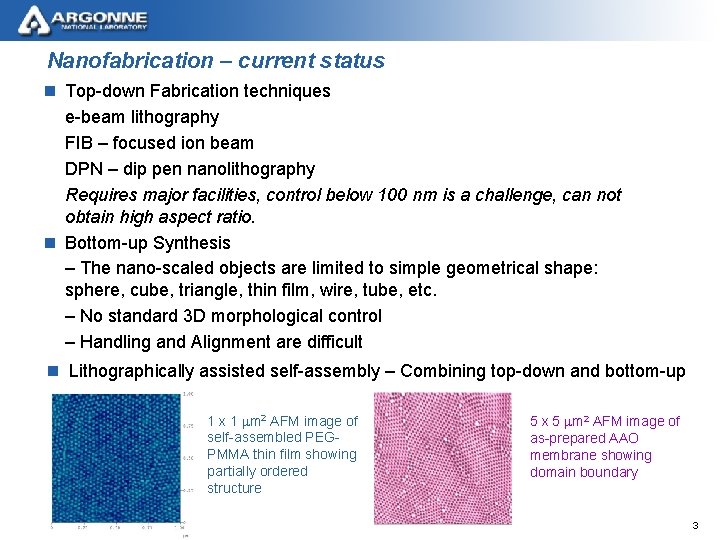
Nanofabrication – current status n Top-down Fabrication techniques e-beam lithography FIB – focused ion beam DPN – dip pen nanolithography Requires major facilities, control below 100 nm is a challenge, can not obtain high aspect ratio. n Bottom-up Synthesis – The nano-scaled objects are limited to simple geometrical shape: sphere, cube, triangle, thin film, wire, tube, etc. – No standard 3 D morphological control – Handling and Alignment are difficult n Lithographically assisted self-assembly – Combining top-down and bottom-up 1 x 1 mm 2 AFM image of self-assembled PEGPMMA thin film showing partially ordered structure 5 x 5 mm 2 AFM image of as-prepared AAO membrane showing domain boundary 3

Nanotubes n AAO/Al 2 O 3/V 2 O 5 nanostructure has been prepared for heterogeneous catalysis studies. n Pd nanotubes showed very good hydrogen sensing capability. Before ALD After ALD Cross-Section of Coated AAO 200 nm Bi NT 4

Nanoparticles n Au, Co/Pt Nanoparticles – Magnetic storage (X. M. Lin et al. ) 5

Dynamics of Monolayer Nanocrystal Self-assembling Process in Liquid Film To understand the dynamics of highly ordered nanocrystal monolayer through evaporation of a nanocrystal colloidal droplet. (J. Phys. Chem. B 2001, 105, 3353) Grazing Incidence Small Angle X-ray Scattering (GISAXS) § Kinetic effects can play an important role in nanocrystal self-assembly. § Fast initial evaporation rate can induce nanocrystals accumulate at the liquid-air interface and undergo a 3 D to 2 D structural transition to form highly ordered nanocrystal monolayer. Xiao-Min Lin MSD/CHM Suresh Narayanan, Jin Wang Advanced Photon Source 6

50 nm Au Nanowires n Various metallic (Au, Ag, Cu, etc. ), superconductive (Pb), ferromagnetic (Co, Ni, etc. ) nanowires have been made with anodized aluminum oxide (AAO) templates. Co 20 nm/Cu 20 nm n Multi-segmented (Co/Cu), (Au/Ni – Mirkin, Science 2005) nanowires have been prepared with pulsed electro-deposition. n Multi-segmented Si. Ni/Si/Si. Ni nanowires have been fabricated into integrated nanowire circuits. (Lieber, Nature 2005) http: //WWW. anl. gov/ Materials Science Division, MSD Center for Nanoscale Materials, CNM M AP FM 1 (Co) NM (Cu) FM 2 (Co) R Parallel AP H Parallel DR/R H 7
- Slides: 7