Nano Toxicology Study on CNT Nano Carbons Historical

CONFIDENTIAL Nano Toxicology Study on CNT & Nano Carbons Historical Aspect & Standardization ESI Archamps in 2018 Summer Shuji Tsuruoka, Ph. D. , Prof. Shinshu University, Institute of Carbon Science & Technology Nagano, Japan 2020/9/15 1

Nano Materials Safety Genesis CONFIDENTIAL In Chaos about Nano Materials · Different surface characteristics from bulk chemical substances · Not new chemicals in Law/ IUPAC classifications → High surface reactivity (e. g. titanium oxide as photo catalyst) · No scientific evidence but SF novels generated unknown fear CNTs ü Different properties from bulk carbon ü High mechanical strength and electrical properties ü Fiber shape "Impression" to analogize asbestos Nano-phobia by Rich & Famous ü Erix Drexler: Gray Goo Nano-machine to eat creatures: so-called pseudo scientific review ü Prince Charles publicly showed his concern about nano. ü M. Crichton (Author of Jurassic Park): Prey (It's just a SF horror, but. . . ) CNTs is the second Asbestos!? Oberdorster G. , et al. P&FT 1743 -8977 -2 -8 2005 Key toxicologists addressed appropriate approach on Nano Tox v Proposal for tox evaluation based on precautionary principle, initiation of R&D v The conventional methods are not going to work. v Find standard nano materials Practical CNT Tox study began in July 2005 in London by Dr. J. Moore (WWICS) & S. Tsuruoka (Mitsui Co. Ltd. ) 2020/9/15 2
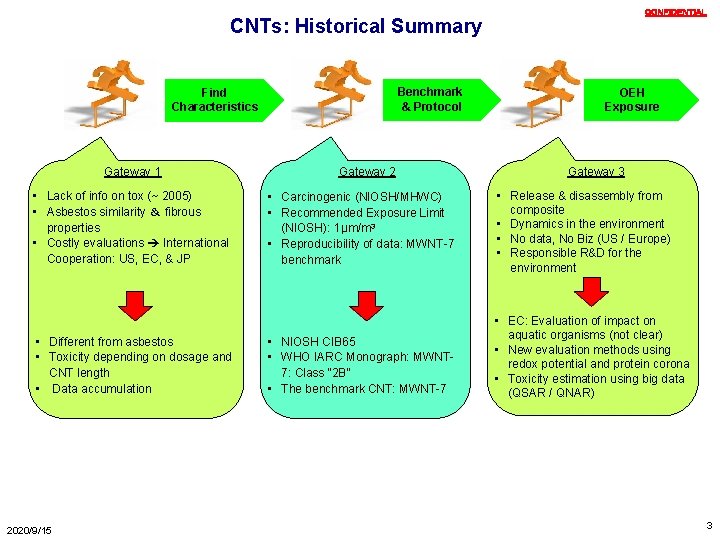
CONFIDENTIAL CNTs: Historical Summary Benchmark & Protocol Find Characteristics Gateway 1 • Lack of info on tox (~ 2005) • Asbestos similarity & fibrous properties • Costly evaluations International Cooperation: US, EC, & JP • Different from asbestos • Toxicity depending on dosage and CNT length • Data accumulation 2020/9/15 Gateway 2 • Carcinogenic (NIOSH/MHWC) • Recommended Exposure Limit (NIOSH): 1µm/m 3 • Reproducibility of data: MWNT-7 benchmark • NIOSH CIB 65 • WHO IARC Monograph: MWNT 7: Class “ 2 B” • The benchmark CNT: MWNT-7 OEH Exposure Gateway 3 • Release & disassembly from composite • Dynamics in the environment • No data, No Biz (US / Europe) • Responsible R&D for the environment • EC: Evaluation of impact on aquatic organisms (not clear) • New evaluation methods using redox potential and protein corona • Toxicity estimation using big data (QSAR / QNAR) 3
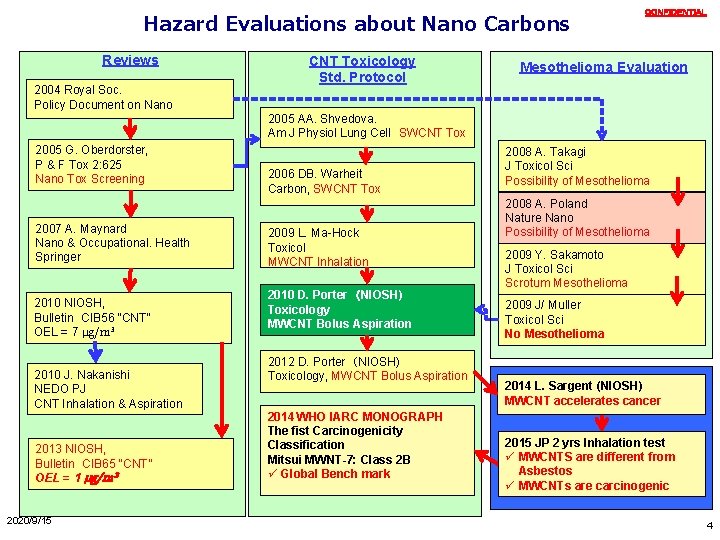
Hazard Evaluations about Nano Carbons Reviews 2004 Royal Soc. Policy Document on Nano CNT Toxicology Std. Protocol CONFIDENTIAL Mesothelioma Evaluation 2005 AA. Shvedova. Am J Physiol Lung Cell SWCNT Tox 2005 G. Oberdorster, P & F Tox 2: 625 Nano Tox Screening 2007 A. Maynard Nano & Occupational. Health Springer 2010 NIOSH, Bulletin CIB 56 “CNT” OEL = 7 μg/m³ 2010 J. Nakanishi NEDO PJ CNT Inhalation & Aspiration 2013 NIOSH, Bulletin CIB 65 “CNT” OEL = 1 μg/m³ 2020/9/15 2006 DB. Warheit Carbon, SWCNT Tox 2009 L. Ma-Hock Toxicol MWCNT Inhalation 2010 D. Porter (NIOSH) Toxicology MWCNT Bolus Aspiration 2012 D. Porter (NIOSH) Toxicology, MWCNT Bolus Aspiration 2014 WHO IARC MONOGRAPH The fist Carcinogenicity Classification Mitsui MWNT-7: Class 2 B ü Global Bench mark 2008 A. Takagi J Toxicol Sci Possibility of Mesothelioma 2008 A. Poland Nature Nano Possibility of Mesothelioma 2009 Y. Sakamoto J Toxicol Sci Scrotum Mesothelioma 2009 J/ Muller Toxicol Sci No Mesothelioma 2014 L. Sargent (NIOSH) MWCNT accelerates cancer 2015 JP 2 yrs Inhalation test ü MWCNTS are different from Asbestos ü MWCNTs are carcinogenic 4

2008’s News Break-out in US, EU, & JP CONFIDENTIAL All used Mitsui MWNT-7! 2020/9/15 5

CONFIDENTIAL “Why Mitsui (stupidly) supplied CNTs without any condition!? It puts the business at risk!!!” Management says… ü Disclosure ü Contribution ü Commitment 2020/9/15 6
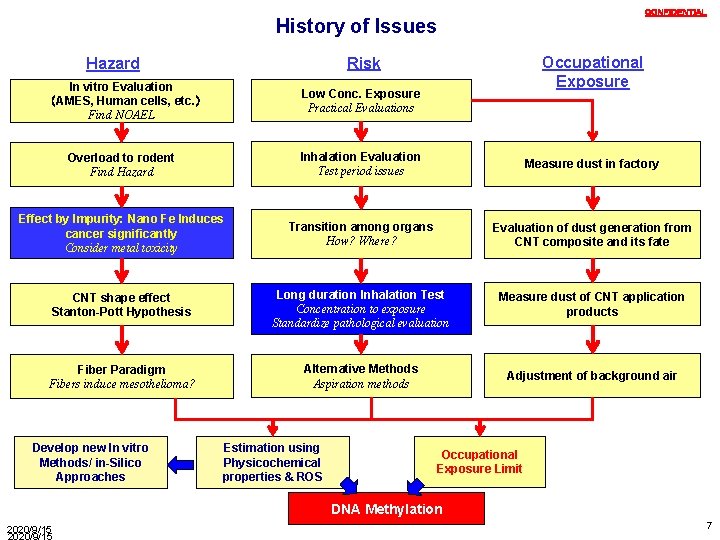
CONFIDENTIAL History of Issues Occupational Exposure Risk Hazard In vitro Evaluation (AMES, Human cells, etc. ) Find NOAEL Low Conc. Exposure Practical Evaluations Overload to rodent Find Hazard Inhalation Evaluation Test period issues Measure dust in factory Effect by Impurity: Nano Fe Induces cancer significantly Consider metal toxicity Transition among organs How? Where? Evaluation of dust generation from CNT composite and its fate CNT shape effect Stanton-Pott Hypothesis Long duration Inhalation Test Concentration to exposure Standardize pathological evaluation Measure dust of CNT application products Fiber Paradigm Fibers induce mesothelioma? Alternative Methods Aspiration methods Adjustment of background air Develop new In vitro Methods/ in-Silico Approaches Estimation using Physicochemical properties & ROS Occupational Exposure Limit DNA Methylation 2020/9/15 7
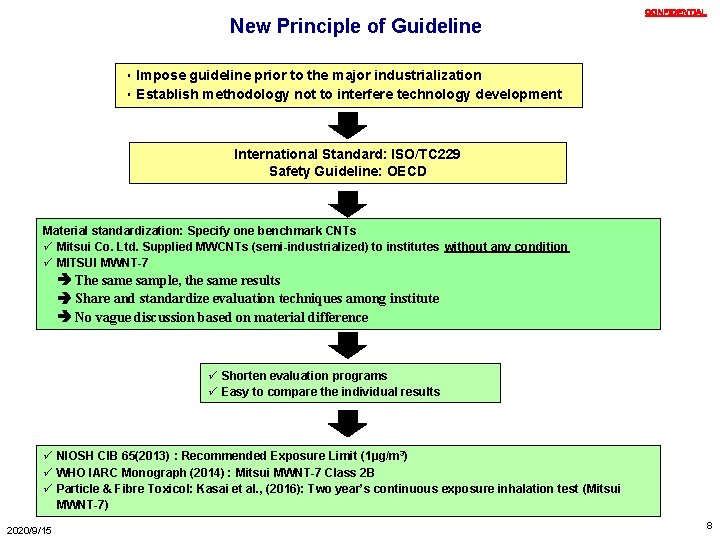
New Principle of Guideline CONFIDENTIAL ・Impose guideline prior to the major industrialization ・Establish methodology not to interfere technology development International Standard: ISO/TC 229 Safety Guideline: OECD Material standardization: Specify one benchmark CNTs ü Mitsui Co. Ltd. Supplied MWCNTs (semi-industrialized) to institutes without any condition ü MITSUI MWNT-7 The sample, the same results Share and standardize evaluation techniques among institute No vague discussion based on material difference ü Shorten evaluation programs ü Easy to compare the individual results ü NIOSH CIB 65(2013):Recommended Exposure Limit (1µg/m 3) ü WHO IARC Monograph (2014) : Mitsui MWNT-7 Class 2 B ü Particle & Fibre Toxicol: Kasai et al. , (2016): Two year’s continuous exposure inhalation test (Mitsui MWNT-7) 2020/9/15 8

Why & How Mitsui MWNT-7 Becomes the Benchmark? CONFIDENTIAL Let us use SWOT analysis approach Mitsui’s Strength Issues Mitsui’s weak-point Notes Issues Notes 1 Organization One of largest JP corporations ü Trading company 1 PL ü Well-recognized but not experienced ü No specialist about PL 2 Biz ü Investment, trade, venture 2 TOX study ü None among group companies ü Not established in japan 3 CNTs ü Huge Investment & establish R&D/ manuf’ing 3 Management ü Seek collaboration partners 4 Resource ü Skilled & experienced employees 5 Communicat’n ü With key members of Emerging Nano organizations ü Cooperated with the government Mitsui’s Threat Issues Mitsui’s Opportunity Issues Notes 1 Opportunity ü UK-JP Nano Safety Conference in 2005 ü Met with Dr. J. Moore (WWICS, US) ü Took action after Dr. Moore’s suggestion; Visited NIOSH in Morgantown ü Mitsui US (NYC) worked with Tokyo HQ 1 Competitors ü All are major manufact’ers ü Own their Safety Evaluation Div. ü Life long Competitor Mitsubishi Corp has its safety evaluation by collaboration 2 Mitsui’s Crest ü More than 300 yrs History ü Negative images from the past & TV drama 2 Management ü Decided to disclose any negative info to public ü No silo to go forward with safety 3 Nano Tech ü Behind other competitors 3 Sample ü Pilot stage MWCNTs 4 Cost ü First contributor was included into Nat’l projects in US, EU, JP 9 2020/9/15

Change the History CONFIDENTIAL ² Major manufactures hesitated about disclosure, though they address to contribute to the society openly. ² Mitsui broke the “under the table rule among big manufactures”. ² After 2005, manufacturers gradually disclose negative info (but not enough yet). 2020/9/15 10

Overview of CNT Tox Studies CONFIDENTIAL Benchmark is Mitsui MWNT-7 Disclosed info 2020/9/15 1 NIOSH CIB 65 (US) • Exposure concentration criterion in air (Benchmark Concentration) to lung injury (Benchmark Dose) • Recommended Exposure Limit • REL = 1 μg / m 3 (10% increase in disability compared to Natural) • Carcinogenicity: MWNT-7 has promotion of cancer 2 Nanosafety Cluster Compendium (EC) • Nano safety assessment program in Europe FP 7 • Aim for QSAR / QNAR (Safety by calculation) • Evaluation of pharmacokinetics by radioactive CNT: CNT exposed to the lung is distributed in organs in the body. It is not absorbed from the digestive tract. • Pseud-biological test using protein corona to estimate environmental exposure. • Aquatic organism test: Daphnia, algae, zebra fish, rainbow trout. 3 WHO IARC Monograph • First time listing CNT: • MWNT-7 and equivalent products: 2 B • The other MWCNT / SWCNT: “ 3” which does not mean safe. • The result of Ministry of Health, Labor and Welfare (Japan Bioassay) 48 months Inhalation exposure test implies the next revision forecast: 2 B ⇒ 2 A 4 Japan Bioassay Inhalation (48 mo) test (JP) • MWNT-7 Ultra long-term continuous inhalation exposure test: Although MWNT-7 has carcinogenicity (carcinogenesis or cancer promotion), but does not induce mesothelioma. • Toxicity data is almost the same as one time dose (NIOSH method), but accumulation property was confirmed. 11

Overview of CNT Tox Studies CONFIDENTIAL Benchmark is Mitsui MWNT-7 Disclosed info 5 Nanosafety Cluster (Nano Mile) • Necessity of European regulatory. • Results are reflected in regulation. • Toxicity evaluation research and data collection to aim for the establishment of QSAR / QNAR. • Nano REACH: Food, cosmetics, general consumer goods, labeling as presently regulated subjects. • Aiming to establish a global standard safety protocol. Not-Disclosed Info 6 Nano. Release • Euro and US collaboration: Evaluation of MWCNT environment scattering from resin composite. • Epoxy and Tire: Partly disclosed at Workshop held at nanosafe 2014. • MWCNT kneading applications: epoxy, tire rubber • Key research institutions: BASF and US EPA • Follow up OECD's research report on next generation tire dust suppression. Others - • • 2020/9/15 Ministry of Health, Labor and Welfare (JP), Tsuda-Hirose team (one publication of article by overdose) Genotoxicity evaluation by Japanese bioassay exposure test results. NIST Environmental Release (Dropping out of resin composite); The full report has not be published yet? NRCWE: Exposure from cutting resin, concrete composite. 12
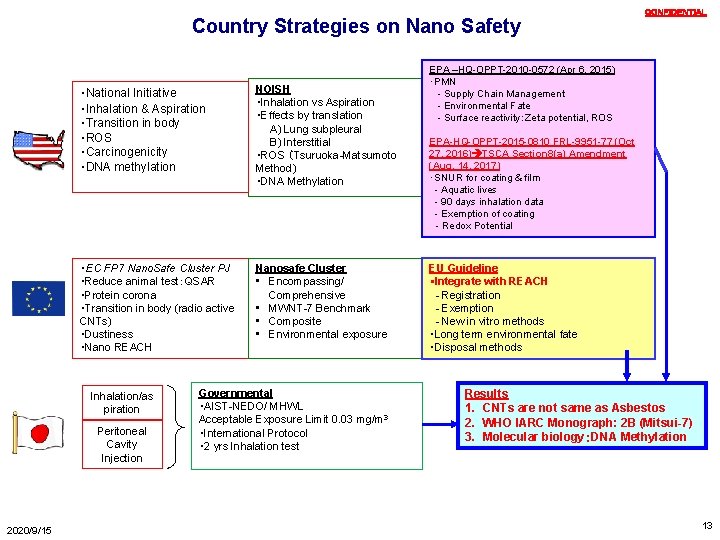
Country Strategies on Nano Safety ・National Initiative ・Inhalation & Aspiration ・Transition in body ・ROS ・Carcinogenicity ・DNA methylation NOISH ・Inhalation vs Aspiration ・Effects by translation A) Lung subpleural B) Interstitial ・ROS (Tsuruoka-Matsumoto Method) ・DNA Methylation ・EC FP 7 Nano. Safe Cluster PJ ・Reduce animal test:QSAR ・Protein corona ・Transition in body (radio active CNTs) ・Dustiness ・Nano REACH Nanosafe Cluster • Encompassing/ Comprehensive • MWNT-7 Benchmark • Composite • Environmental exposure Inhalation/as piration Peritoneal Cavity Injection 2020/9/15 Governmental ・AIST-NEDO/ MHWL Acceptable Exposure Limit 0. 03 mg/m 3 ・International Protocol ・ 2 yrs Inhalation test CONFIDENTIAL EPA –HQ-OPPT-2010 -0572 (Apr 6, 2015) ・PMN - Supply Chain Management - Environmental Fate - Surface reactivity:Zeta potential, ROS EPA-HQ-OPPT-2015 -0810 FRL-9951 -77 (Oct 27, 2016) TSCA Section 8(a) Amendment (Aug. 14, 2017) ・SNUR for coating & film - Aquatic lives - 90 days inhalation data - Exemption of coating - Redox Potential EU Guideline ・Integrate with REACH - Registration - Exemption - New in vitro methods ・Long term environmental fate ・Disposal methods Results 1. CNTs are not same as Asbestos 2. WHO IARC Monograph: 2 B (Mitsui-7) 3. Molecular biology:DNA Methylation 13
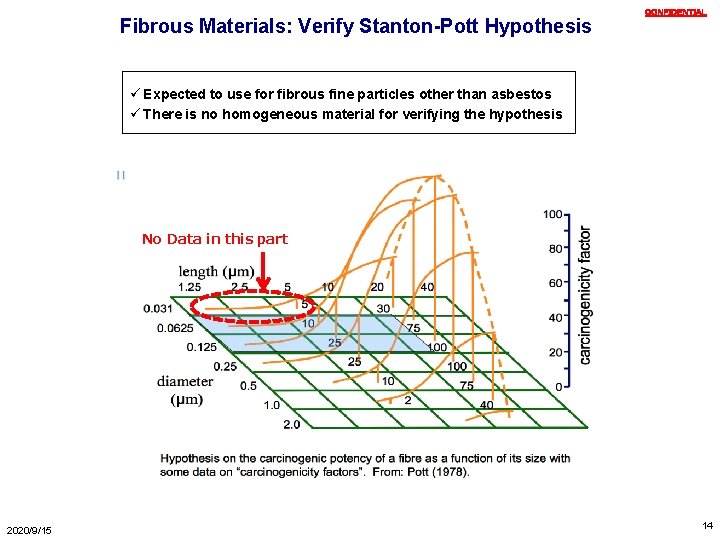
Fibrous Materials: Verify Stanton-Pott Hypothesis CONFIDENTIAL ü Expected to use for fibrous fine particles other than asbestos ü There is no homogeneous material for verifying the hypothesis No Data in this part 2020/9/15 14

Challenge at Stanton-Pott Hypothesis (July 2017 ~) CONFIDENTIAL JP MHWL (NIHS-JP & Nagoya City Univ. ) Siddarmark Group Developed: ü DWCNTs/TWCNTs: Ave D = 3. 3 n. ü Size distribution: Very narrow ü Purity: Not detected metal ü Inhalation exposure test by Taquann method (24 months) ü Dispersion in t-butyl alcohol: almost perfect dispersion ü The amount of spray & measurement are proportional with the Condensation Particle Counter (CPC) ü Measurable END POINT ü Three length: 5, 10, 15 µm ü Condensation Particle Counter (CPC) ü End points:Nano tox parameter, Inflammation, collagen thickening, apoptosis, body weight change, cytokine, neutrophil concentration change, others UK MRC (Dr. T. Chernova) ü Mesothelioma expression by direct injection into lung pleura (2 -24 months) ü Collaboration with Edinburgh University ü Dispersing micrograms CNTs into body fluids END POINT ü Two length (50, 100 µm) ü Examination of hypothesis of Donaldson / Poland: Excretion size from Stoma (expected: length 2 mm) where fibrosis manifests regardless of substance ü Measurement item: Mesothelioma 2018/4/25 15

Taquann Method CONFIDENTIAL A+B+D: Air Pressure Relief Dumper A: Flexible Plastic Film for Insulation B: ULPA Filter Unit C: Main Flow Inlet (cut off during injection) D: Air Pressure Dumper Duct E: Subchamber F: Cartridge Loader/Injector G: Inhalation Chamber (with animal cages) 2018/4/25 16

DNA Methylation CONFIDENTIAL Definition Reaction in which a methyl group is added to the C 5 position of a dinucleotide of a Cp. G island sequence found in DNA. It is deeply involved in Epigenetics and is thought to be an essential mechanism for accurately shaping the body of complex organisms. This modification is inherited also. It plays an extremely important role in the development of most cancers. Previous Articles on Epigenetics ü Abnormal methylation is seen in cancer & cardiovascular diseases (Hyper- & Hypo -): DNA alteration is observed with specific proteins (IFN-γ) & genes by PM and ozone (2012, 2015) ü Confirmation of hypo-methylation of PARP-1 enzyme gene by nanosilica (2010, 2015) ü Studies on Methylation mutation in MWCNT (2016) 2018/4/25 18
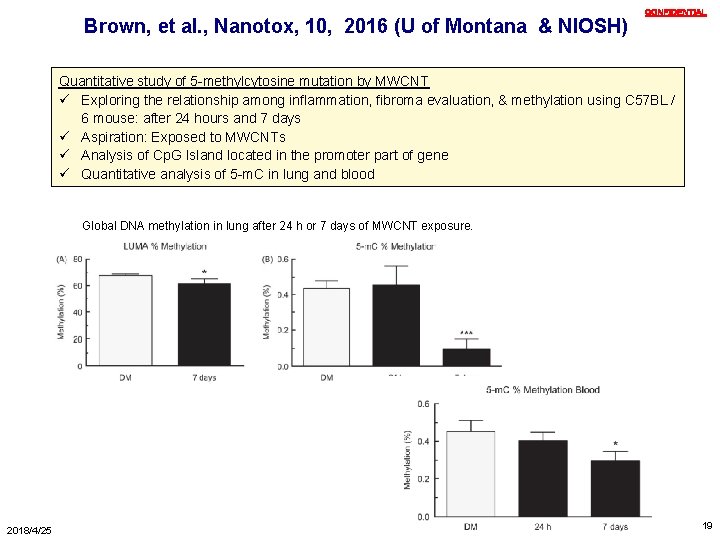
Brown, et al. , Nanotox, 10, 2016 (U of Montana & NIOSH) CONFIDENTIAL Quantitative study of 5 -methylcytosine mutation by MWCNT ü Exploring the relationship among inflammation, fibroma evaluation, & methylation using C 57 BL / 6 mouse: after 24 hours and 7 days ü Aspiration: Exposed to MWCNTs ü Analysis of Cp. G Island located in the promoter part of gene ü Quantitative analysis of 5 -m. C in lung and blood Global DNA methylation in lung after 24 h or 7 days of MWCNT exposure. 2018/4/25 19
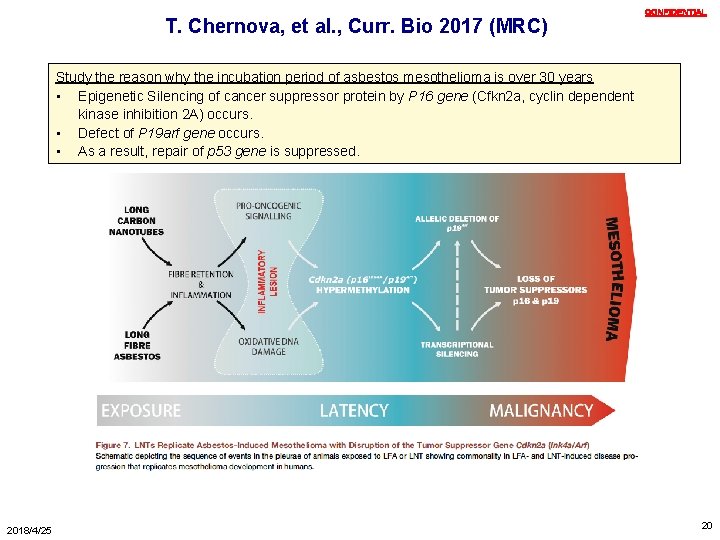
T. Chernova, et al. , Curr. Bio 2017 (MRC) CONFIDENTIAL Study the reason why the incubation period of asbestos mesothelioma is over 30 years • Epigenetic Silencing of cancer suppressor protein by P 16 gene (Cfkn 2 a, cyclin dependent kinase inhibition 2 A) occurs. • Defect of P 19 arf gene occurs. • As a result, repair of p 53 gene is suppressed. 2018/4/25 20

DNA Methylation: Issues CONFIDENTIAL Is it a chemical or physical reaction? ü DNA Methylation occurs by asbestos, nanoparticles, MWCNTs ü Univ. Of Montana (NIOSH, NIH) is Hypo-, MRC is Hyper-Methylation. ü Uo. M: Carcinogenicity, MRC: long term Silencing ü Exposure site; Uo. M is inhalation exposure to the lungs, MRC is Pleural Cavity pleural cavity. Issues to be solved by Methylation of Cp. G ü Shape effect Corresponds with the Stanton-Pott hypothesis? ⇒ Evaluation using DWCNTs with uniform length & diameter: Japan: Exposure (Inhalation exposure), MRC: Injection to Pleural Cavity. ü Threshold: Effect on Cp. G site is genetically retained There is no threshold? ⇒ It is necessary to wait and see Japan & MRC results Methylation evaluation as a nanoparticle toxicity evaluation method ü At present it is insufficient data to satisfy the above-mentioned problem, but it is thought that the prediction of carcinogenicity will be possible, so that the United States and Britain are aggressive to conduct the study. ü It may be a "scientific" indicator for regulation 2020/9/15 21

Summary 2020/9/15 CONFIDENTIAL 1. Possible toxicity classification by CNT length/ diameter 2. WHO IARC Monograph: MWNT - 7 and equivalent: 2 B. The other CNTs: 3. Asbestos nature of MWCNT: Those thicker than MWNT - 7 may be categorized to Thick Fiber Paradigm (WHO standard: diameter around 3 μ and thicker, without upper limit of length) 3. Environmental exposure: Although it is expected to be an extremely low concentration of exposure, emphasis is placed on catalytic reaction by "contact" in addition to "internal uptake" (protein corona test) 4. Verification about Stanton-Pott Hypothesis 5. Biochemical Evaluation: DNA Methylation 22

- Slides: 23