Naming Transition Metal Cations Pull Out Blank Periodic

Naming Transition Metal Cations
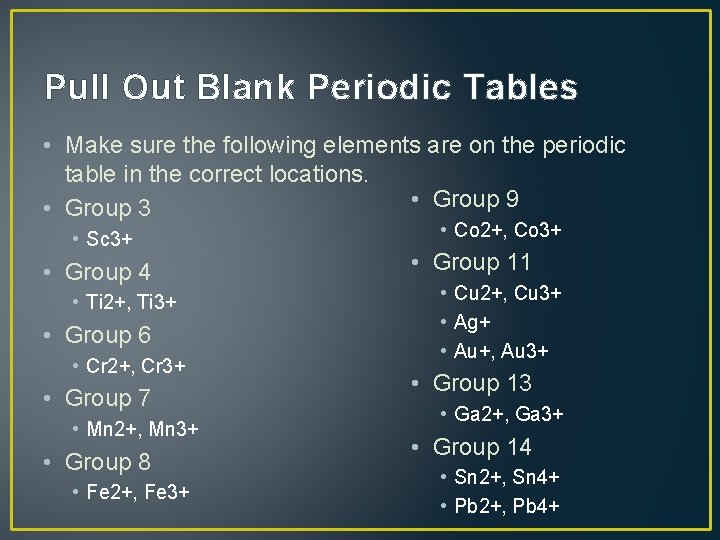
Pull Out Blank Periodic Tables • Make sure the following elements are on the periodic table in the correct locations. • Group 9 • Group 3 • Sc 3+ • Group 4 • Ti 2+, Ti 3+ • Group 6 • Cr 2+, Cr 3+ • Group 7 • Mn 2+, Mn 3+ • Group 8 • Fe 2+, Fe 3+ • Co 2+, Co 3+ • Group 11 • Cu 2+, Cu 3+ • Ag+ • Au+, Au 3+ • Group 13 • Ga 2+, Ga 3+ • Group 14 • Sn 2+, Sn 4+ • Pb 2+, Pb 4+

Transition Metals • For elements that have more than one charge (oxidation value), roman numerals are used within parentheses. • The number within the parentheses is equal to the charge of the element. • Transition metals are a sub category of metals; therefore their charge is always positive.

Identifying the Charge of Transition Metals • Within Ionic Compounds, Transition Metals must be bonded with a non-metal. • Non-metal elements have a set oxidation value. • The charge of a transition metal can be identified by working backwards. • Examples • Cu. O • Co. Cl 3 • Fe 2 O 3

• The oxidation number of a monatomic ion is the same as its charge. • Alkali metals exhibit only an oxidation state of +1 in compounds. • Alkaline earth metals exhibit only an oxidation state of +2 in compounds. Oxidation Rules

• The sum of the oxidation numbers is zero for an electrically neutral compound. • All halogens have a – 1 oxidation number in compounds, except when with oxygen or other halogens where their oxidation numbers can be positive. • Hydrogen is always assigned a +1 oxidation number in compounds, except when it is in a hydride form, where its charge is – 1. Oxidation Rules
- Slides: 6