Naming Ionic Compounds Chemical Bonding IV Lewis Structures

Naming Ionic Compounds Chemical Bonding IV
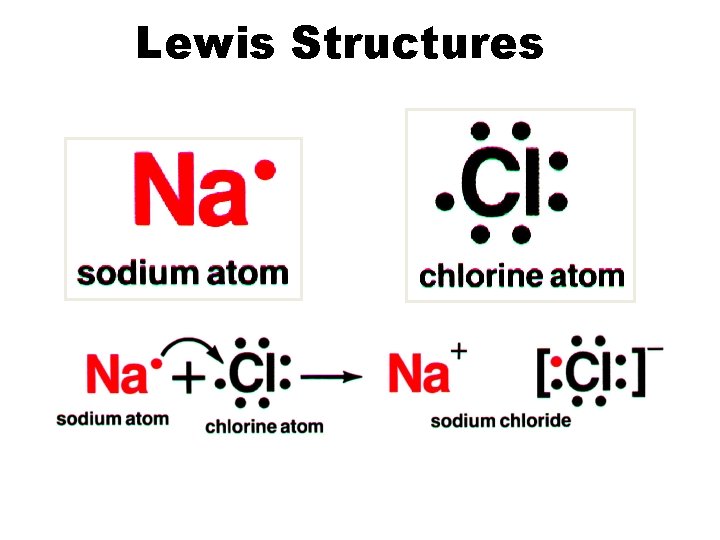
Lewis Structures

How to write a formula with transition metals • When a Transition metal is involved in the bond, you must identify which form of the atom is bonding. • These atoms can have different oxidation numbers. • Look for the Roman Numeral in the name when writing the formula. The Roman numeral is the oxidation number of the transition metal. Copper (II) Fluoride Cu+2 2 F -1 Cu. F 2

Writing Formula Practice Copper (II) Fluoride Formula Cu+2 2 F -1 Cu. F 2 Name Copper (I) Oxide Iron (III) Phosphide Manganese (V) Fluoride Zinc Phosphide Nickel Oxide

Writing Names Practice Copper (II) Fluoride Formula Cu 3 N Ti 3 P 2 Sn. O Mn. F 3 Ti. O 2 Cu+2 2 F -1 Name Cu. F 2

How to write a formula with polyatomic ions • If you have a compound with a polyatomic ion, the name will have a different ending using either (-ate) or (-ite) • Use your polyatomic ions chart to determine the ion • When writing formulas, keep these ions in parentheses if there is more than one polyatomic

Writing Formula Practice Sodium Nitrate Formula Na+1 NO 3 -1 Na. NO 3 Name Magnesium Nitrate Sodium Nitrite Aluminum Phosphate Barium Sulfate Ammonium Fluoride

How to write a name with polyatomic ions • Look for more than two elements. That’s means a Polyatomic is present. • Match up the polyatomics and write the correct names and ending straight from the chart.

Writing Names Practice Sodium Nitrate Formula Na 2 SO 4 Al 2(SO 3)3 Ca. CO 3 NH 4 Cl Cu. C 2 H 3 O 2 Na+1 NO 3 -1 Na. NO 3 Name
- Slides: 9