Naming Compounds and Writing Formulas Acids Bases I

Naming Compounds and Writing Formulas Acids & Bases I II IV

A. Acid Definition z. Acids y. Compounds that form H+ in water. y. Formulas usually begin with ‘H’. z. Examples: y. HCl – hydrochloric acid y. HNO 3 – nitric acid y. H 2 SO 4 – sulfuric acid

B. Acid Nomenclature

B. Acid Nomenclature

B. Acid Nomenclature z. HBr y 2 elements, -ide hydrobromic acid z. H 2 CO 3 y 3 elements, -ate carbonic acid z. H 2 SO 3 y 3 elements, -ite sulfurous acid
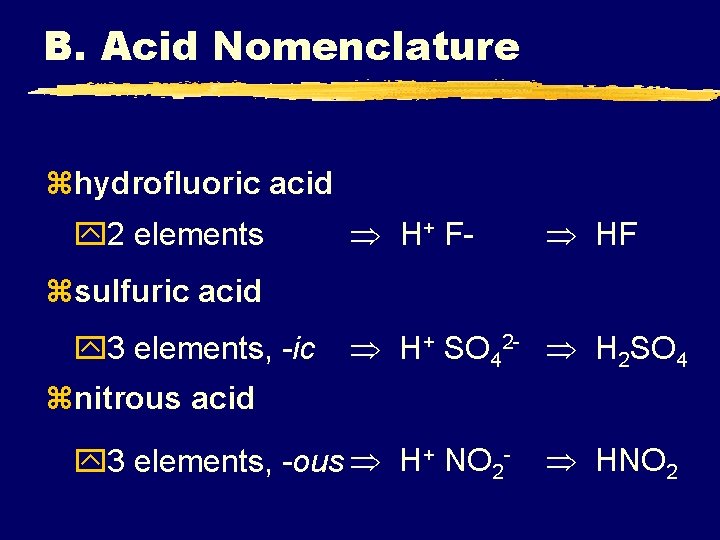
B. Acid Nomenclature zhydrofluoric acid y 2 elements H+ F- HF zsulfuric acid y 3 elements, -ic H+ SO 42 - H 2 SO 4 znitrous acid y 3 elements, -ous H+ NO 2 - HNO 2

C. Base Definition z. Base y. Compounds that form OH- in water. y. Formulas usually end with ‘OH’. z. Examples: y. Na. OH – sodium hydroxide y. Ba(OH)2 – barium hydroxide y. NH 3 – ammonia

D. Base Nomenclature SAME RULES AS APPLIED TO IONIC COMPOUNDS!!! z. Write the name of the metallic cation first. z. Write the name of the hydroxide. z. Use Roman numerals to show the ion’s charge if more than one is possible. (Will only use if dealing with transition metals)
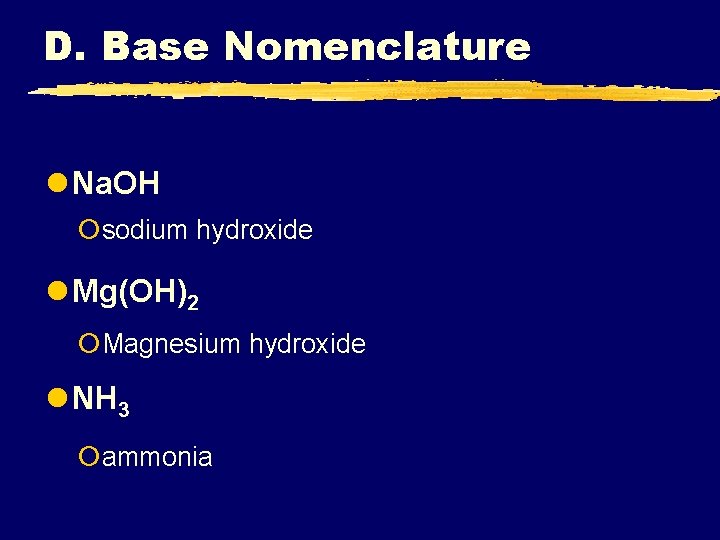
D. Base Nomenclature l Na. OH ¡sodium hydroxide l Mg(OH)2 ¡Magnesium hydroxide l NH 3 ¡ammonia

D. Base Nomenclature Name the following ionic compounds: z. Fe(OH)3 = y. Iron(III) hydroxide z. KOH = z. Al(OH)3 = y. Potassium hydroxide y. Aluminum hydroxide
- Slides: 10