Naming Chemical Compounds Binary Ionic Compounds Binary 2

Naming Chemical Compounds

Binary Ionic Compounds Binary = 2 types of elements ¡ Ionic = made of ions ¡ Compound = atoms bonded together ¡

Ionic Compounds Positive ions are attracted to Negative ions ¡ FORMULA UNIT ¡ ¡ Cation always comes first, then anion

Elemental anions ¡ When an element becomes an anion, it starts to end in –ide l l l l Fluoride Chloride Bromide Oxide Sulfide Nitride Phosphide

Type I ¡ The cation has a predictable charge l l l Alkali Metals +1 Alkaline Earth Metals Group 3 A +3 Silver Ag+ Zinc Zn 2+ +2

Type II ¡ The cation has a variable charge l ¡ All Transition metals except for Ag and Zn (for our purposes) The name will have a roman numeral to denote the CHARGE on the cation Fe 2+ Fe 3+ Cu 2+ Iron(II) Ferrous Iron(III) Ferric Copper(I) Cuprous Copper(II) Cupric

Naming ionic compouds (Type I and II) ¡ First name cation l l ¡ Then name anion l ¡ Just the name of element if Type I Name of element and roman numeral with charge on cation if Type II Elemental anions end in “–ide” When determining the formula, remember “swap ‘n’ drop”

Name the following: ¡ ¡ ¡ Na 3 N Mg. Cl 2 Al 2 O 3 ¡ Cu. Cl 2 Pb. Br 4 ¡ ¡ ¡ Sodium nitride Magnesium chloride Aluminum oxide Copper(I) chloride Copper(II) chloride Lead(IV) bromide

Determine the formula of the following ¡ ¡ ¡ ¡ Aluminum chloride Magnesium Oxide Sodium nitride Calcium chloride Copper(I) sulfide Lead (II) bromide Iron(III) oxide ¡ ¡ ¡ ¡ Al. Cl 3 Mg. O Na 3 N Ca. Cl 2 Cu 2 S Pb. Br 2 Fe 2 O 3

Polyatomic Ions Many anions are not just ions of elements. ¡ Polyatomic Ions = many atomed particle with an overall charge ¡ ¡ Mostly nonmetals bonded to Oxygens

Polyatomic Ion Naming ¡ Main polyatomic ion l l ¡ l Ends in “-ite” Example: Chlorite Cl. O 2 - Even fewer oxygens l l ¡ Cl. O 3 - One fewer oxygens l ¡ Ends in “–ate” Example: Chlorate Hypo = under Example: Hypochlorite Cl. O- More oxygens than main ion l l Hyper = over, shortened to “per” Example: Perchlorate Cl. O 4 -

Common Polyatomic Ions NO 2 - Nitrite NO 3 - Nitrate SO 32 - Sulfite SO 42 - Sulfate PO 43 - Phosphate PO 33 - Phosphite

Common Polyatomic Ions Hydrogen can attach to the polyatomic ions as H+ ¡ This adds an H and reduces the overall charge by 1 ¡ PO 43 HPO 4 2 - Phosphate Hydrogen Phosphate H 2 PO 41 - Dihydrogen Phosphate CO 32 - Carbonate HCO 31 - Hydrogen carbonate (bicarbonate)

Name these compounds with PAIs ¡ ¡ ¡ Al. PO 4 Cu(NO 3)2 Na 2 SO 4 Na. HCO 3 Mg 3(PO 4)2 ¡ ¡ ¡ Aluminum phosphate Copper(II) nitrate Sodium sulfate Sodium bicarbonate Magnesium Phosphate

Molecular Compounds Molecular compounds are not bonded by positive and negative ions ¡ They SHARE electrons ¡ Made of two non-metals ¡ Cannot predict or explain charges (no charges, because not LOSING or GAINING electrons – SHARING) ¡

Naming Molecular Compounds (Type III) ¡ ¡ Because there is no way to neutralize the compounds (like for ions) we must determine how many there another way: Use PREFIXES

How to tell if it’s Type III Check the FIRST element – is it a non-metal? ¡ If YES, it’s a Type III ¡ Name the first element as the element ¡ Name the second element ending in “-ide” ¡
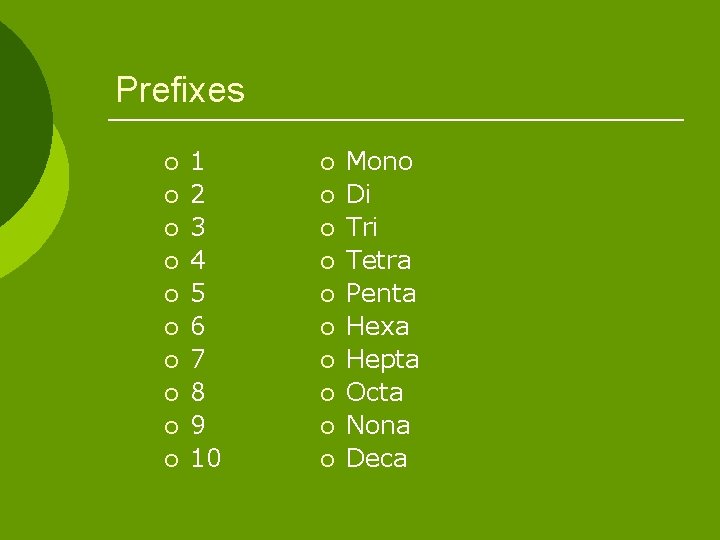
Prefixes ¡ ¡ ¡ ¡ ¡ 1 2 3 4 5 6 7 8 9 10 ¡ ¡ ¡ ¡ ¡ Mono Di Tri Tetra Penta Hexa Hepta Octa Nona Deca

Using prefixes If there is only one of the first element, you may omit the “mono” ¡ You may not omit any other prefixes ¡ The more electronegative element goes last (upper right hand corner) ¡ F, O, N, Cl, ¡

Name the following ¡ ¡ ¡ PCl 3 CO 2 NO N 2 O 4 N 2 F 5 OF 2 ¡ ¡ ¡ Phosphorus trichloride Carbon dioxide Nitrogen monoxide Dinitrogen tetroxide Dinitrogen pentafluoride Oxygen difluoride

Naming Practice! For each of the following compounds, determine whether it is type I, II, or III. 1. 2. 3. 4. 5. 6. 7. 8. Fe. Cl 3 Na. Cl Cu(OH)2 N 2 O Ba. SO 4 PO 5 (NH 4)CO 3 Sn. Br 2

Naming Practice! For each of the following compounds, determine the CHARGE on the cation (+1, +2, +3, or +4) 1. 2. 3. 4. 5. 6. Fe. Cl 3 Cu(OH)2 Cr 2 S 3 Ni. S Pb(NO 3)2 Sn. Cl 4 1. 3 2. 2 3. 3 4. 2 5. 2 6. 4

Acids All acids have the H+ ion in them as the cation. ¡ All acids have “acid” in their name ¡ Consider Hydrochloric Acid ¡ HCl ¡

Naming Acids ¡ If the anion DOES NOT CONTAIN OXYGEN: l l l ¡ Prefix hydro. Root name of anion element Suffix –ic Examples l l HF H 2 S Hydrofluoric Acid Hydrosulfuric Acid Hydrooxygenic acid

Naming Acids ¡ If the anion CONTAINS OXYGEN (ie: is a polyatomic ion) l l Root name of the PAI If anion ends in -ate, ¡ l acid ends in –ic If anion ends in –ite, ¡ acid ends in –ous

Naming Acids Examples ¡ ¡ ¡ ¡ H 2 SO 4 H 2 SO 3 HNO 2 HC 2 H 3 O 2 ¡ Phosphoric Acid Carbonic Acid ¡ ¡ ¡ Sulfuric Acid Sulfurous Acid Nitric Acid Nitrous Acid Acetic Acid H 3 PO 4 H 2 CO 3

Organic Molecules Carbon based ¡ Naming is based on number of carbons ¡ Functional groups ¡ l ¡ Sets of molecules attached to the carbons Naming is also based on what functional groups are where

Alkanes The simplest organic molecules are Alkanes. ¡ Carbons are all single bonded to each other ¡ When not bonded to another carbon, bonded to a Hydrogen ¡ All alkanes end in -ane ¡


Prefixes ¡ Note that the prefixes pertain to all types of carbon based molecules with any types of functional groups or chains • Meth • Eth • Prop • But • Pent • Hex • Hept • Oct

Alkenes and Alkynes ¡ ¡ A functional group can be a set of atoms, or the way they are bonded. Alkenes l ¡ Carbons have double bonds between them Alkynes l Carbons have triple bonds between them

Alkenes and Alkynes ¡ To Name l l l All alkenes end with –ene. All alkynes end with –yne Number the carbons in the longest chain. Carbon 1 is at the end of the chain closest to the first functional group Put the number that the bond is on, then the chain length (some name by putting the number BEFORE the functional group ending)
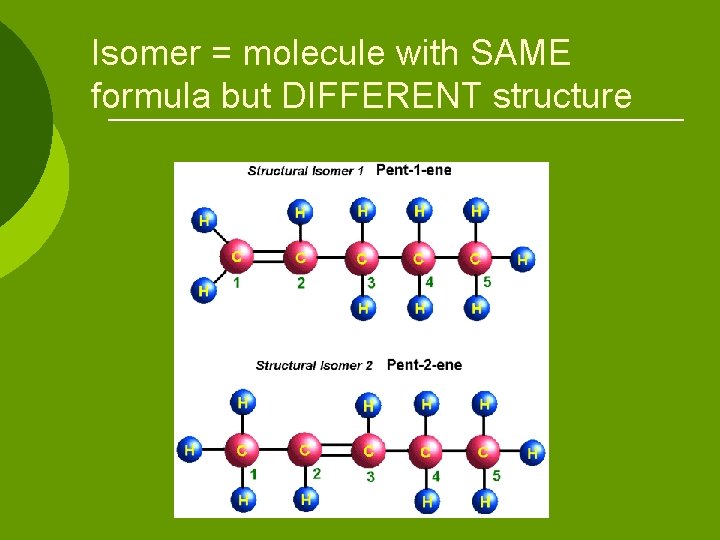
Isomer = molecule with SAME formula but DIFFERENT structure

Alcohols Have an –OH group attached ¡ Name the carbon chain (prefix and bond) ¡ End in –ol ¡ Indicate which carbon the –OH is on ¡ 2 -propanol ¡ 1 -Butanol ¡ 3 -Octenol ¡

What are the formulas for these alcohols?
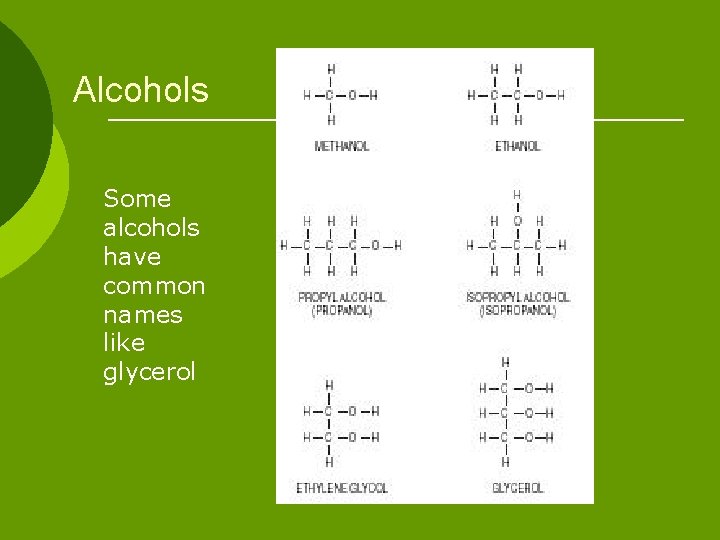
Alcohols Some alcohols have common names like glycerol

Branched Hydrocarbons The longest continuous chain of carbon atoms gives the root name/prefix. ¡ Substituent is named by the number of carbons. ¡ Specify location of substituent by numbering carbon atoms in longest chain. ¡ Substituent often referred to as R or R’ (for second substituent) ¡

Substituent often referred to as R or R’ (for second substituent) ¡ If more than one alkyl group, use prefix di, tri, etc. to alkyl name ¡ Substituents listed in alphabetical order (disregarding prefix) ¡
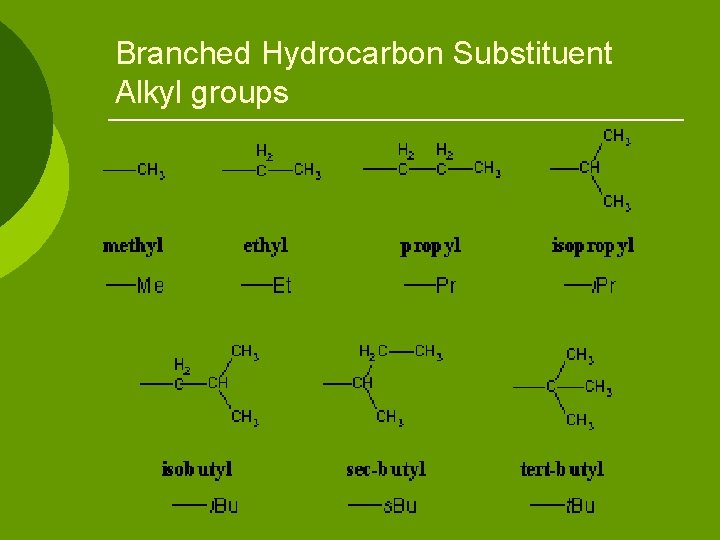
Branched Hydrocarbon Substituent Alkyl groups

What is the name of the compound? Hints • Number Carbon chain from on side closest to first alkyl group • Single carbon branch called methyl, triple carbon group called propyl • More than one branch, use prefixes di-, tri-, etc. 2, 4 dimethyl hexane 4 ethyl, 5 methyl heptane

Tricky ones!!! Each END of a line is a Carbon atom. No H’s are indicated, but they are assumed See if you can name these.

Halohydrocarbons Functional group is a halogen (X = F, Cl, Br, I) ¡ General Formula ¡ l ¡ R–X Naming l l Select longest chain containing halogen Number so that C with halogen gets lowest number

Ethers ¡ Functional group l ¡ General Formula l ¡ -O– R – O – R’ Naming l l l Name the R group as an alkyl Name the R’ group as an alkyl End in ether

Aldehydes ¡ Functional group l -C–H || O ¡ General Formula l R–C–H || O ¡ Naming l l Name the R group Change the ending to -al
- Slides: 44