Naming Alkanes and Hydrocarbons City and Islington College

Naming Alkanes and Hydrocarbons City and Islington College Students Name: …………………………………. .
Topic Chemistry Aims Ø This resource introduces Students to naming Alkanes and Hydrocarbons Level 3 Method Power. Point slides ALL SLIDES ARE HANDOUTS and TESTS (apart from slide 2, which will be hidden) – Go through the slides with the Students, and ask them to fill in the answers Equipment Ø Ø Ø Laptop & Projector Pens Hand-Outs Duration >30 Minutes

Alkanes, Alkenes & Arenes All the above groups of compounds are hydrocarbons – they only contain the element’s carbon and hydrogen. These compounds are obtained from crude oil when it undergoes fractional distillation. Naming hydrocarbons Number of carbon atoms Name begins with - 1 Meth 2 Eth 3 Prop 4 But 5 Pent 6 Hex 7 Hept 8 Oct

Alkanes, Alkenes & Arenes Alkanes These compounds have the general formula Cn. H 2 n+2 Fill out the table below. The names end in –ane. n = 1 2 3 4 5 6 7 8 Formula Name

Alkanes, Alkenes & Arenes Structural Formula Carbon always forms 4 covalent bonds. Hydrogen always forms 1 covalent bond. eg Methane Ethane Draw the structural formula for the following: Propane Pentane

Alkanes, Alkenes & Arenes Rules for naming Hydrocarbons Name the longest chain _________________________________________________________ Name any side chains giving them the lowest possible number _________________________________________________________ Name any functional groups giving them the lowest possible number _________________________________________________________ Branch Names Name of branch Formula Methyl CH 3 - Ethyl CH 3 CH 2 -
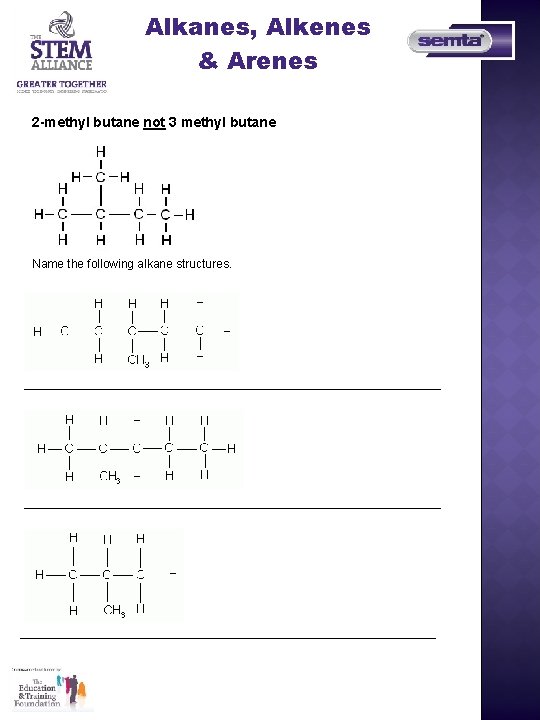
Alkanes, Alkenes & Arenes 2 -methyl butane not 3 methyl butane Name the following alkane structures.

Alkanes, Alkenes & Arenes Draw the following Alkane Structures 3 Methyl Pentane 2, 2 Methyl Pentane

Alkanes, Alkenes & Arenes Alkenes These hydrocarbons have the general formula Cn. H 2 n Fill out the table below. The name ends in –ene. n = 2 Formula Name 3 4 5 6 7 8

Alkanes, Alkenes & Arenes Structural formula - Recall each carbon has 4 bonds and each hydrogen 1 bond. Eg: ethene but-2 -ene Draw the structural formula of the following. But-1 -ene hex-2 -ene Pent – 1, 3 -diene hex-3 -ene Compounds which contain carbon-carbon double bonds are known as unsaturated compounds. Much margarine is high in polyunsaturated fat – these contain a lot of carbon-carbon double bonds. Arenes These compounds contain the C 6 H 6 ring structure – known as a benzene ring. Benzene is a cacogenic substance and is not used in laboratories. It is much safer to use methyl-benzene which has the old name Toluene

For further information please contact The STEM Alliance enquiries@STEMalliance. uk or visit www. STEMalliance. uk
- Slides: 11