Naming Acids and Bases Binary Acids Binary acids

Naming Acids and Bases


Binary Acids • Binary acids: made up of only two elements - hydrogen and one other element.

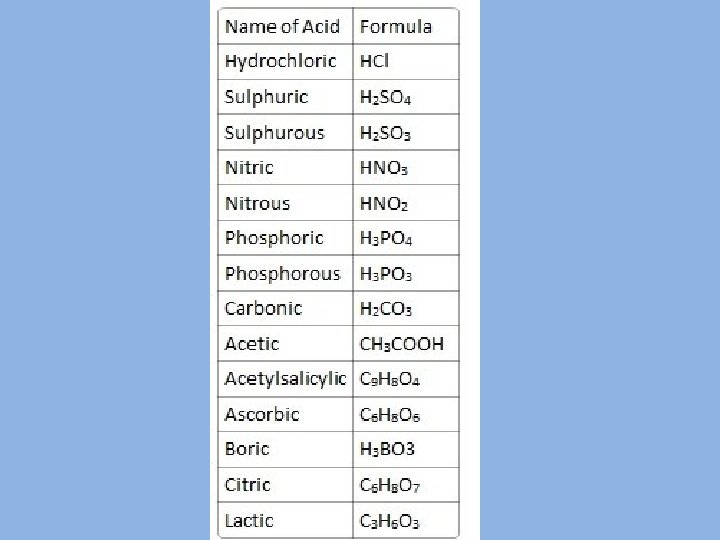
Naming binary acids: – Begin with the prefix hydro. – Determine the "stem" - part of the name of the element that combines with hydrogen. – Add the suffix- ic. • Examples: – HCl - hydro chlor ic - hydrochloric acid – HBr - hydro brom ic - hydrobromic acid
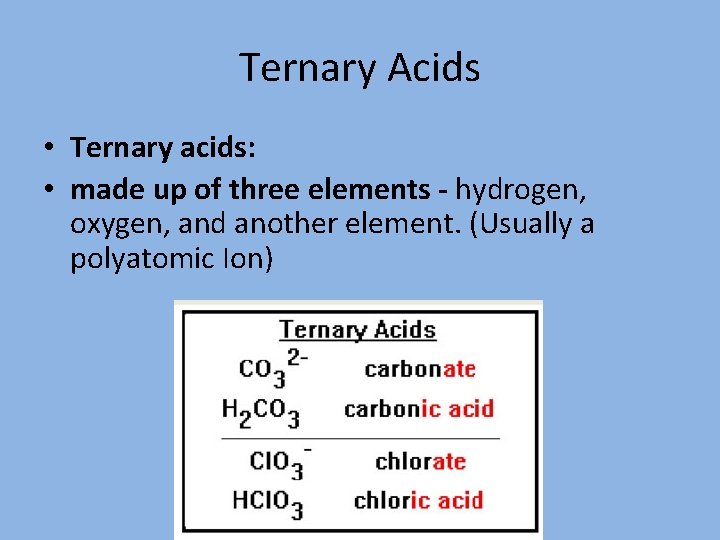
Ternary Acids • Ternary acids: • made up of three elements - hydrogen, oxygen, and another element. (Usually a polyatomic Ion)

Naming ternary acids: – Determine the "stem" - part of the name of the third element. – The most common acid is given the suffix ic. – Add the prefix per for the acid with one more oxygen. – The suffix ous is given to the acid with one less oxygen. – Add the prefix hypo for the acid with two less oxygen atoms.

Examples of Ternary Acids – HCl. O 4 - per chlor ic - perchloric acid - one more oxygen atom. – HCl. O 3 - chlor ic - chloric acid - the most common form of the acid. – HCl. O 2 - chlor ous - chlorous acid - one less oxygen atom. – HCl. O - hypo chlor ous - hypochlorous acid - two less oxygen atoms.

Other important terms: • Amphoteric - a substance that acts as either acid or base, depending on what it reacts with. – Water is the most common amphoteric substance. In the presence of a proton donor, it acts like a base. In the presence of a proton acceptor, it acts like an acid. • Anhydrous - without water. – Anhydrides are substances that have had water removed. – Example: Taking the water out of Ba(OH)2 leaves Ba. O.

Practice : Write the anhydrous form of the following Acids 1. 2. 3. 4. H 2 SO 3 H 2 C 2 O 4 H 3 PO 4 H 4 C 2 O 2

Acid anhydride • Acid anhydride: an oxide that produces an acid when dissolved in water. – Oxides of nonmetals are acid anhydrides. – Example: SO 2 + H 2 O H 2 SO 3 (an acid)

Practice : Write the formula of the compound formed when these are dissolved in water. 1. SO 3 2. NO 2

Practice : Write the formula of the compound formed when these are dissolved in water. 1. H 2 SO 4 2. HNO 3

Basic anhydride • Base anhydride: an oxide that produces a base in when dissolved in water. • Oxides of metals are basic anhydrides. • Example: Na 2 O + H 2 O 2 Na. OH (a base)

Practice : write the formula of the compound formed when these are dissolved in water. 1. 2. Mg. O Al 2 O 3

Practice : write the formula of the compound formed when these are dissolved in water. 1. 2. 2 Mg. OH Al(OH)3

What is a strong Acid? An Acid that is 100% ionized in water. Strong Acids: 100% ionized (completely dissociated) in water. HCl + H 2 O H 3 O+ + Cl- often written as: HCl H+ + Cl-

Strong acids and bases ionize completely in water solution. • This rule-of-thumb can be used : – HCl, HBr, and HI are the only strong binary acids. – In strong ternary acids, the number of oxygen atoms exceeds the number of hydrogen atoms by two or more. Examples are H 2 SO 4 and HNO 3 • Hydroxides of groups 1 and 2, except Be, are strong bases.
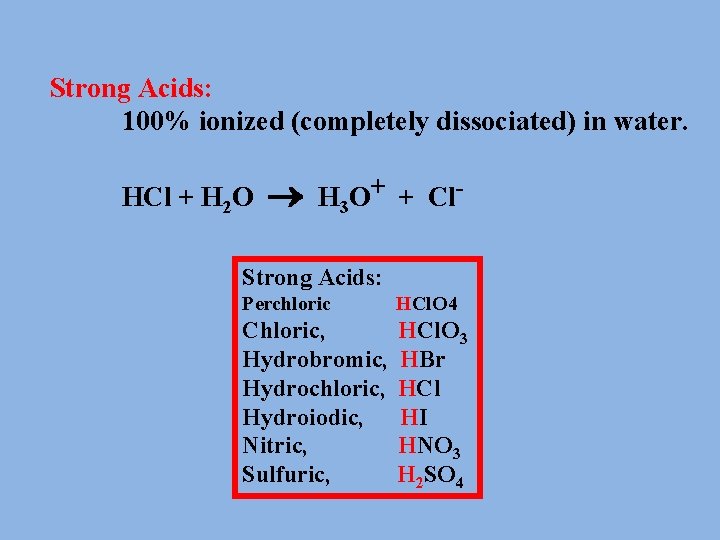
Strong Acids: 100% ionized (completely dissociated) in water. HCl + H 2 O H 3 O+ + Cl- Strong Acids: Perchloric HCl. O 4 Chloric, Hydrobromic, Hydrochloric, Hydroiodic, Nitric, Sulfuric, HCl. O 3 HBr HCl HI HNO 3 H 2 SO 4

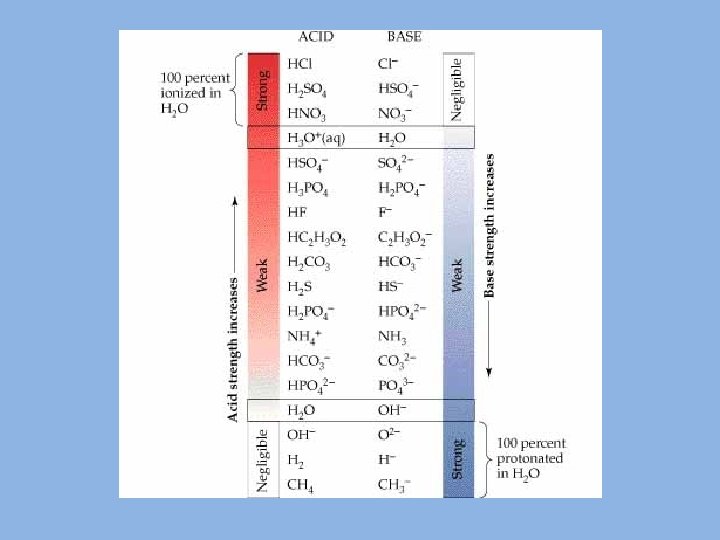
Weak acids and bases ionize only slightly in water solution. • This rule-of-thumb can be used : – Any binary acid not listed above is weak. – A ternary acid is weak if the ratio of oxygen to hydrogen is less than two to one. An example is H 3 PO 4 • Any hydroxide not listed above is a weak base.

Acid – Base Strength Strong Acid - Transfers all protons to water; completely ionizes; strong electrolyte; conjugate base is weaker and has negligible tendency to be protonated. Weak Acid - Fraction of protons transferred to water; partly ionized; weak electrolyte; conjugate base is stronger readily accepting protons from water § As acid strength decreases, base strength increases. § The stronger the acid, the weaker its conjugate base § The weaker the acid, the stronger its conjugate base

Leveling effect The term leveling effect refers to a solvent's ability to level the effect of a strong acid or base dissolved in it. When a strong acid is dissolved in water, it reacts with it to form H 3 O+ in the following reaction (where "HA" is a generic strong acid such as "HCl"): HA + H 2 O -> A- + H 3 O+ Any acid that is stronger than H 3 O+ reacts with H 2 O to form H 3 O+; therefore, no acid can be stronger than H 3 O+ in H 2 O. This is true with any solvent, although usually to a lesser extent than with water. Bases may be also leveled in solvents
- Slides: 23