Name Dr Pramod B Thakur Class S Y

Name: Dr. Pramod B. Thakur Class: S. Y. B. Sc Subject: Inorganic Chemistry Title of Topic: Comparative Chemistry of Transition Metals

SYBSC-Paper-II, Unit-II Paper Code-USCH 302 Comparative Chemistry of Transition Metals Teacher: Dr. Pramod B. Thakur

Difference between the properties of elements of 3 d series and 4 d, 5 d series • • • Ambiguous Electronic configuration Ability to form cluster compounds Stability of oxidation state Geometry of Co-ordination compound Atomic and Ionic size Magnetic properties
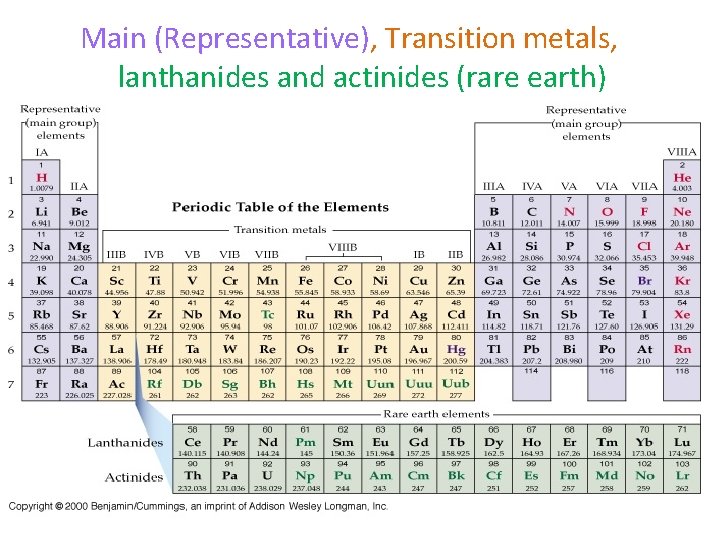
Main (Representative), Transition metals, lanthanides and actinides (rare earth)

1. Ambiguous Electronic configuration 3 d series • • • Sc (Z = 21), [Ar] 3 d 1 4 s 2 Ti (Z = 22), [Ar] 3 d 2 4 s 2 V (Z = 23), [Ar] 3 d 3 4 s 2 Cr (Z = 24), [Ar] 3 d 5 4 s 1 Mn (Z = 25), [Ar] 3 d 54 s 2 Fe (Z = 26), [Ar] 3 d 6 4 s 2 Co (Z = 27), [Ar] 3 d 7 4 s 2 Ni (Z = 28), [Ar] 3 d 8 4 s 2 Cu (Z = 29), [Ar 3 d 10 4 s 1 Zn (Z = 30), [Ar] 3 d 10 4 s 2 [Ar] = 18
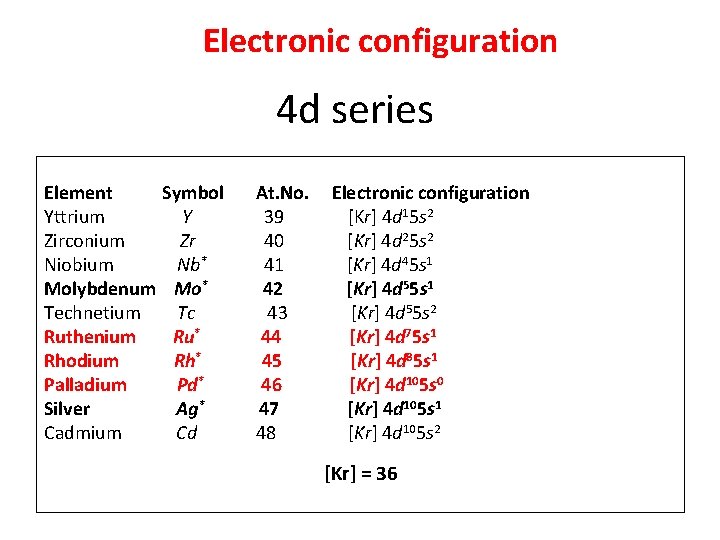
Electronic configuration 4 d series Element Yttrium Zirconium Niobium Molybdenum Technetium Ruthenium Rhodium Palladium Silver Cadmium Symbol Y Zr Nb* Mo* Tc Ru* Rh* Pd* Ag* Cd At. No. 39 40 41 42 43 44 45 46 47 48 Electronic configuration [Kr] 4 d 15 s 2 [Kr] 4 d 25 s 2 [Kr] 4 d 45 s 1 [Kr] 4 d 55 s 2 [Kr] 4 d 75 s 1 [Kr] 4 d 85 s 1 [Kr] 4 d 105 s 0 [Kr] 4 d 105 s 1 [Kr] 4 d 105 s 2 [Kr] = 36
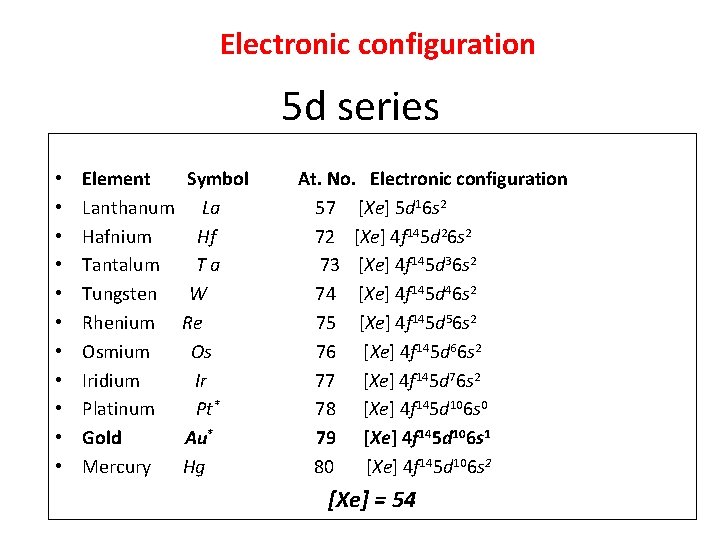
Electronic configuration 5 d series • • • Element Lanthanum Hafnium Tantalum Tungsten Rhenium Osmium Iridium Platinum Gold Mercury Symbol La Hf Ta W Re Os Ir Pt* Au* Hg At. No. Electronic configuration 57 [Xe] 5 d 16 s 2 72 [Xe] 4 f 145 d 26 s 2 73 [Xe] 4 f 145 d 36 s 2 74 [Xe] 4 f 145 d 46 s 2 75 [Xe] 4 f 145 d 56 s 2 76 [Xe] 4 f 145 d 66 s 2 77 [Xe] 4 f 145 d 76 s 2 78 [Xe] 4 f 145 d 106 s 0 79 [Xe] 4 f 145 d 106 s 1 80 [Xe] 4 f 145 d 106 s 2 [Xe] = 54

2. Ability to form cluster compounds • Complex involving Metal-Metal bonds are called as metal clusters • Elements of second and third series have greater tendency to form cluster compounds than elements of first transition series. • Eg: Chloride and carbonyls cluster compounds. 3 d Series 4 d series 5 d series

3. Stability of Oxidation state • In first series, lower +2, +3 oxidation state are stable and higher oxidation state are unstable. • In second and third series, lower +2, +3 oxidation state are unstable and higher oxidation state are stable. • Eg: Co+2 is stable, but Rh+2, Ir+2 are not stable, • Re+7, Tc+7, are stable but Mn+7 is not stable 3 d Series 4 d series 5 d series

4. Geometry of Co-ordination Complexes • In second and third series elements forms co -ordination compounds with higher coordination number 7 & 8. • In First series elements do not forms coordination compounds with higher coordination number. 3 d Series 4 d series 5 d series
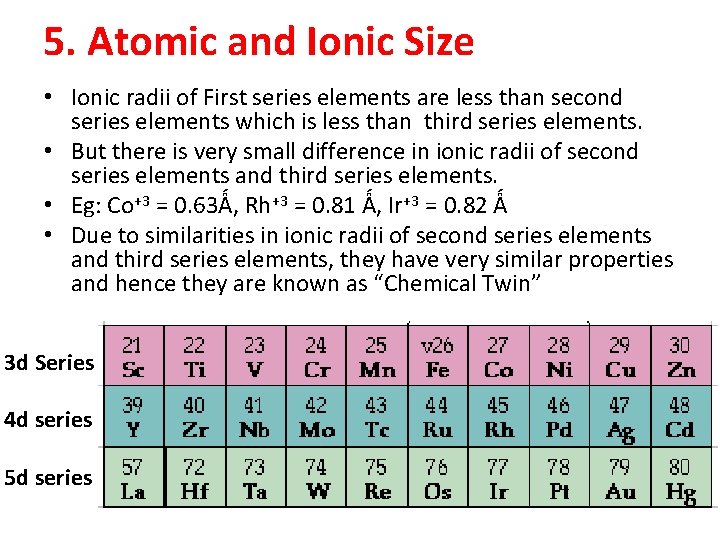
5. Atomic and Ionic Size • Ionic radii of First series elements are less than second series elements which is less than third series elements. • But there is very small difference in ionic radii of second series elements and third series elements. • Eg: Co+3 = 0. 63Ǻ, Rh+3 = 0. 81 Ǻ, Ir+3 = 0. 82 Ǻ • Due to similarities in ionic radii of second series elements and third series elements, they have very similar properties and hence they are known as “Chemical Twin” 3 d Series 4 d series 5 d series
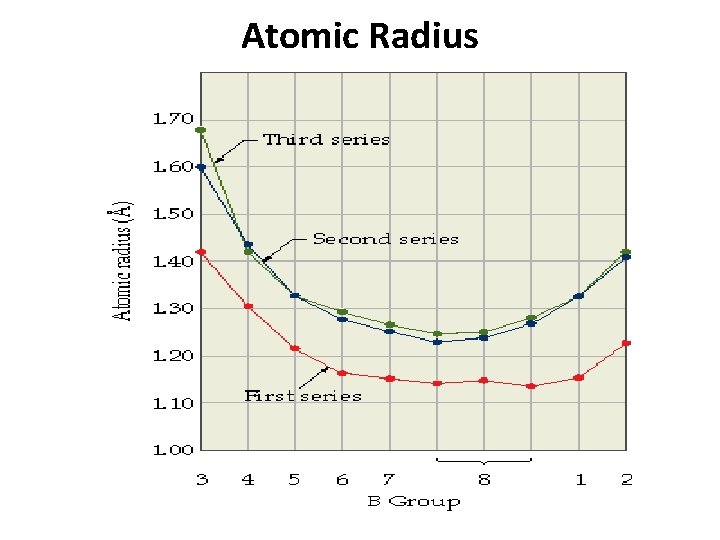
Atomic Radius

6. Magnetism • Magnetic moments of First series elements are calculated only by considering spin motion. • Magnetic moments of Second series elements and Third series elements are calculated by considering both spin motion and orbital motion. 3 d Series 4 d series 5 d series

- Slides: 14