Myotonic dystrophy DM Suhail Abdulla Al Rukn 19

Myotonic dystrophy DM Suhail Abdulla Al. Rukn 19 -03 -2008

Outline - Overview of repeat expansion disorders. - Introduction to DM - Etiology and pathophysiology of DM - Model mechanisms for myotonic dystrophy: - Haploinsufficiency of DMPK - Haploinsufficiency of SIX 5 and neighboring genes - RNA pathogenesis

Learning Objective -Introduction to repeat expansion disorders and the different between the unstable expanding repeats in noncoding regions and ustable expanding repeats in coding regions. -The pathophysiology of DM, and the three model mechanisms for myotonic dystrophy.

Overview of repeat expansion disorders -There are more than 30 neurological disorders that can attribute their pathogenesis to extensions of tandem repeats above a critical size. -Yet theories as to how large repeat arrays can cause such highly variable diseases are unresolved.

Overview of repeat expansion disorders -Most of the expansion disorders exhibit a delayed onset form of their diseases, indicating that they may share mechanisms that postpone clinical expression until later in life. -Depending on where the unstable repeats are located within the gene, the repeat expansion disorders can be classified as having coding or noncoding mutations.

Overview of repeat expansion disorders -Coincidentally, in 1991, the first two triplet repeat expansion disorders discovered revealed examples from both coding and noncoding categories: -The fragile X syndrome was linked to unstable CGG repeats in the noncoding 5′UTR of FMR 1. -Whereas spinobulbar muscular atrophy was associated with unstable coding CAG repeats.

Overview of repeat expansion disorders - DM 1 followed in 1992 as the third trinucleotide repeat expansion disorder discovered and was mapped to CTG repeats in the noncoding 3′UTR of DMPK. -UREDs can be divided into two major classes: -Unstable expanding repeats in noncoding regions. -Unstable expanding repeats in coding regions.

Unstable expanding repeats in noncoding regions. Biochim Biophys Acta. 2007 Feb; 1772(2): 195 -204. Epub 2006 Jun 20.

Unstable expanding repeats in coding regions. Biochim Biophys Acta. 2007 Feb; 1772(2): 195 -204. Epub 2006 Jun 20.

Introduction -Myotonic dystrophy (DM) is the most common muscular dystrophy in adults, and is the second most common muscular dystrophy after Duchenne muscular dystrophy. -Onset is usually in the second or third decade. -The prevalence of DM is 1 in 8000 in the general population
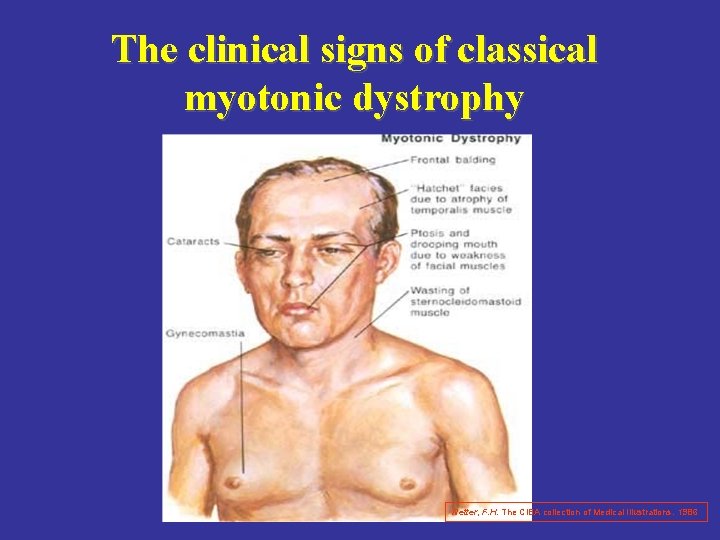
The clinical signs of classical myotonic dystrophy Netter, F. H. The CIBA collection of Medical Illustrations. 1986

Types • The genetic causes of three forms of DM have been identified: – DM 1, also known as Steinert's disease – DM 2, also known as proximal myotonic myopathy (PROMM) 1994 – Congenital myotonic dystrophy (CMy. D)
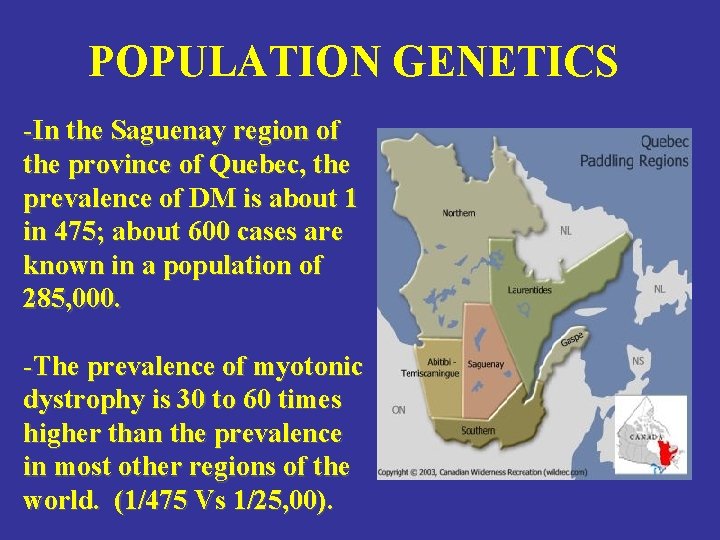
POPULATION GENETICS -In the Saguenay region of the province of Quebec, the prevalence of DM is about 1 in 475; about 600 cases are known in a population of 285, 000. -The prevalence of myotonic dystrophy is 30 to 60 times higher than the prevalence in most other regions of the world. (1/475 Vs 1/25, 00).

Etiology -DM is autosomal dominant disease. -DM 1 results from an unstable trinucleotide repeat expansion, CTG, in the 3' non-coding region (myotonic dystrophy type-1 protein kinase, DMPK) on chromosome 19 q 13. 3. - DM 2 results from an unstable tetranucleotide (CCTG) repeat expansion in the intron 1 of ZNF 9 on chromosome 3 q 21.

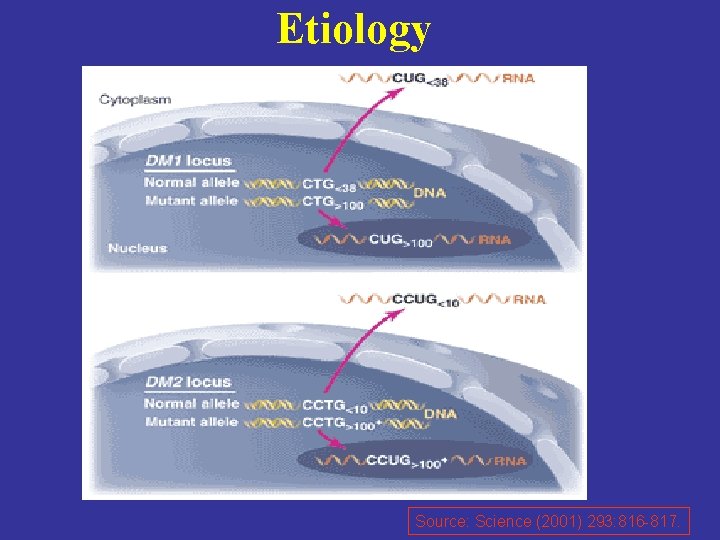
Etiology Source: Science (2001) 293: 816 -817.

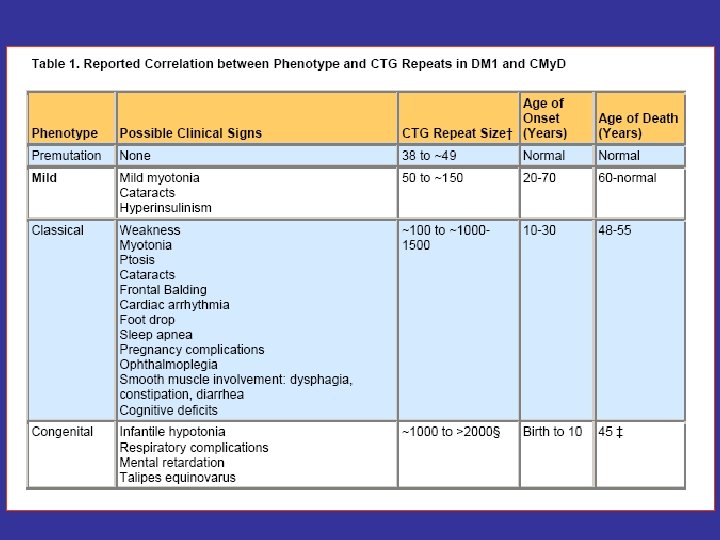
Etiology -The cause of the unstable CTG repeat expansion is unknown; however, it is thought to occur during gametogenesis and is more extensive when coming from a female carrier. - The disease severity generally correlates with repeat length.
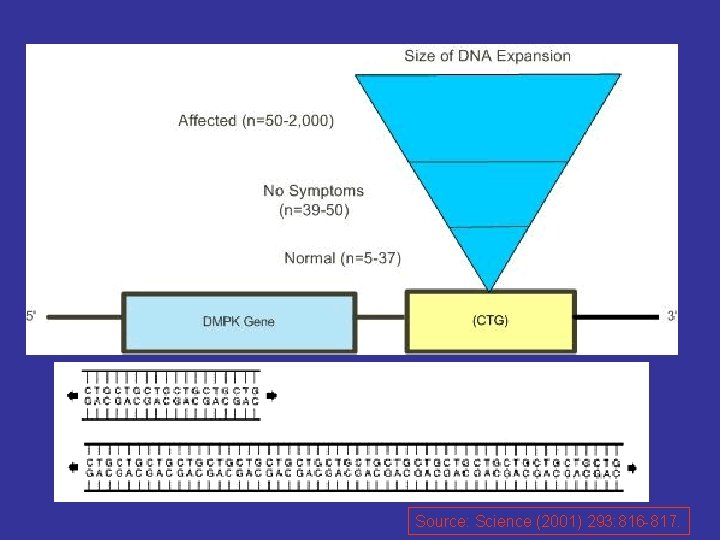
Source: Science (2001) 293: 816 -817.

Model mechanisms for myotonic dystrophy 1) Haploinsufficiency of DMPK 2) Haploinsufficiency of SIX 5 and neighboring genes 3) RNA pathogenesis

RNA toxic gain-of-function model for myotonic dystrophy Biochim Biophys Acta. 2007 Feb; 1772(2): 195 -204. Epub 2006 Jun 20.

Haploinsufficiency of DMPK -Early expression studies were consistent with the hypothesis that the mutation interfered with DMPK production, in that m. RNA and protein levels were reduced in patient muscle and cell culture. -However, DMPK knockout mice showed only a very mild, late-onset myopathy without the multisystemic features of the disease.

Haploinsufficiency of DMPK -The fact that no DMPK point mutations have been associated with a DM phenotype further suggests that the multisystemic features of DM 1 are not simply caused by DMPK haploinsufficiency.

Haploinsufficiency of SIX 5 and neighboring genes -A second proposed mechanism has been that the mutation interferes with expression of multiple genes in the DM 1 region, possibly through regional effects produced by repeat-induced alterations in chromatin structure. -In addition to DMPK and the neighboring homeodomain gene SIX 5, other regional genes suggested to be involved in DM 1 pathogenesis have included myotonic dystrophy gene with WD repeats, DMWD, which is prominently expressed in the testis and brain.

Haploinsufficiency of SIX 5 and neighboring genes -In this model, the multisystemic features of DM 1 would be explained by haploinsufficiency of a number of neighboring genes, with expression level and hence disease severity, dependent on repeat length. -In support of this possibility, Six 5 knockout mice develop cataracts, but without the posterior subcapsular location or the distinctive opacities that are characteristic of cataracts in DM patients.

RNA pathogenesis -A third hypothesized mechanism is that the enlarged CUG-containing transcripts accumulate as intranuclear foci and disrupt cellular function. -Direct support for this model came from a transgenic mouse model, in which the CTG expansion was inserted into the 30 end of the human skeletal actin gene, a gene not directly involved in DM 1 but which is expressed only in skeletal muscle.

RNA pathogenesis -This mouse model expressed an m. RNA with a CUG repeat tract of 250 repeats, and caused the myotonia and myopathic features characteristic of DM 1. -But because CUG-containing transgene expression was limited to skeletal muscle the role of the CUG expansion in the multisystemic features of DM was not addressed.

RNA toxic gain-of-function model for myotonic dystrophy Biochim Biophys Acta. 2007 Feb; 1772(2): 195 -204. Epub 2006 Jun 20.

Pathogenic model of DM 1 and DM 2.
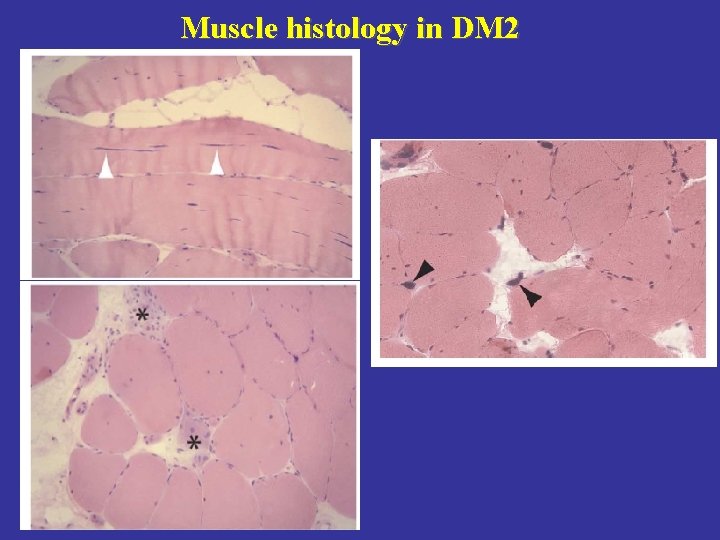
Muscle histology in DM 2

Home Take massage - The severity of the URED’s correlates with repeat length - DM 1 results from an unstable trinucleotide repeat expansion, CTG, DMPK on chromosome 19 q 13. 3. - DM 2 results from an unstable tetranucleotide (CCTG) repeat expansion in the intron 1 of ZNF 9 on chromosome 3 q 21. - The cause of the unstable CTG repeat expansion is unclear but there are few model mechanisms.

Thanks
- Slides: 32