Myeloma Webinar Dr Donna Reece Dr Suzanne Trudel

Myeloma Webinar Dr. Donna Reece Dr. Suzanne Trudel 09 April 2018

Today’s Topics • Overview: Dr. Donna Reece – What is a Research Chair? • Goals of the Bloom Chair – Overview of Bloom Chair Research Program activities at Princess Margaret • New immunotherapy initiatives in myeloma: Dr. Donna Reece – The “basics” of immunotherapy – What is available now? – What does the future hold? • The importance of “Bench to Bedside” research: Dr. Suzanne Trudel – What does “precision medicine” mean? – Specific research initiatives in myeloma

What is a Research Chair? • A research chair is an investment in research – Created by financial contributions for targeted support in a defined field – The designated scholar should establish a culture of creativity and innovation • The Bloom Chair for Myeloma Research was generously established by David and Molly Bloom in 2009 • The purposes of this Chair are to: – Support a “bench to beside approach” = use basic laboratory research to improve the longevity of people affected by myeloma – Provide “stepping stones to cure” CURE

The Princess Margaret Myeloma Team • Patient demographics – Plasma cell disorders: Multiple myeloma, light chain amyloidosis, Waldenstrom’s macroglobulinemia – ~ 350 new referrals are seen per year – ~ 220 stem cell transplants are performed per year • Half performed in Outpatient Day Hospital – Over 2000 myeloma patients seen at least once per year – Over 8000 clinic visits occur per year • Clinical Program – 6 myeloma physicians • Drs. Reece, Chen, Kukreti, Tiedemann and Prica – – – 2 stem cell transplant nurse coordinators 2 -3 Fellows (hematologists doing further training in myeloma) 1 outpatient nurse practitioner 3 stem cell transplant nurse practitioners 1 inpatient physician hospitalist 5 clinic nurses

Princess Margaret Myeloma Research Program: Components Laboratory Research Program Percentiles Smallest 1% 31 5% 43 42 10% 45 43 Obs 52 25% 49. 5 44 Sum of Wgt. 52 50% 56. 5 Mean 55. 55769 Largest Std. Dev. 8. 474676 75% 62 68 90% 65 70 Variance 71. 82014 95% 70 Skewness -. 389775 99% 71 Kurtosis 2. 882401 Percentiles Smallest 1% 31 Princess Margaret Myeloma Database Clinical Research Program Better care/improved longevity for myeloma patients

Princess Margaret Myeloma Research Program: Components Laboratory Research Program 2 Clinical Scientists • • Percentiles Smallest 1% 31 5% 43 42 10% 45 43 Obs 52 25% 49. 5 44 Sum of Wgt. 52 50% 56. 5 Mean 55. 55769 Largest Std. Dev. 8. 474676 75% 62 68 90% 65 70 Variance 71. 82014 95% 70 Skewness -. 389775 99% 71 Kurtosis 2. 882401 Percentiles Smallest 1% 31 Princess Margaret Myeloma Database Dr. Suzanne Trudel Dr. Rodger Tiedemann 9 laboratory research personnel • • • 4 Post-docs Clinical Research 4 lab technicians Program 2 graduate students (Masters, Ph. D) Better care/improved longevity for myeloma patients

Princess Margaret Myeloma Research Program: Components Laboratory Research Program 2 Clinical Scientists • • Dr. Suzanne Trudel Dr. Rodger Tiedemann Percentiles Smallest 1% 31 5% 43 42 10% 45 43 Obs 52 25% 49. 5 44 Sum of Wgt. 52 50% 56. 5 Mean 55. 55769 Largest Std. Dev. 8. 474676 75% 62 68 90% 65 70 Variance 71. 82014 95% 70 Skewness -. 389775 99% 71 Kurtosis 2. 882401 Percentiles Smallest 1% 31 Princess Margaret Myeloma Database 1 Database Manager 1. 5 Database coordinators 9 laboratory research personnel • • • 4 Post-docs Clinical Research 4 lab technicians Program 2 graduate students (Masters, Ph. D) Better care/improved longevity for myeloma patients

Princess Margaret Myeloma Research Program: Components 1 Research Manager 12 Research Coordinators 5 Research Nurses Percentiles Smallest 1% 31 5% 43 42 10% 45 43 Obs 52 25% 49. 5 44 Sum of Wgt. 52 50% 56. 5 Mean 55. 55769 Largest Std. Dev. 8. 474676 75% 62 68 90% 65 70 Variance 71. 82014 95% 70 Skewness -. 389775 99% 71 Kurtosis 2. 882401 Percentiles Smallest 1% 31 Laboratory Research Program Princess Margaret Myeloma Database • 10 -12 new trials opened each year Clinical Research • Each trial lasts 3 -8 years Program • 45 active clinical trials • We design, implement and lead innovative national and international trials Better care/improved longevity for myeloma patients

Princess Margaret Myeloma Research Program: Components UPDATES for 2018 Laboratory Research Program Percentiles Smallest 1% 31 5% 43 42 10% 45 43 Obs 52 25% 49. 5 44 Sum of Wgt. 52 50% 56. 5 Mean 55. 55769 Largest Std. Dev. 8. 474676 75% 62 68 90% 65 70 Variance 71. 82014 95% 70 Skewness -. 389775 99% 71 Kurtosis 2. 882401 Percentiles Smallest 1% 31 Princess Margaret Myeloma Database Immunotherapy Database has + Clinical Research gone national= Precision medicine MCRN Database ! Program Better care/improved longevity for myeloma patients

Immunotherapy for Myeloma Is Here
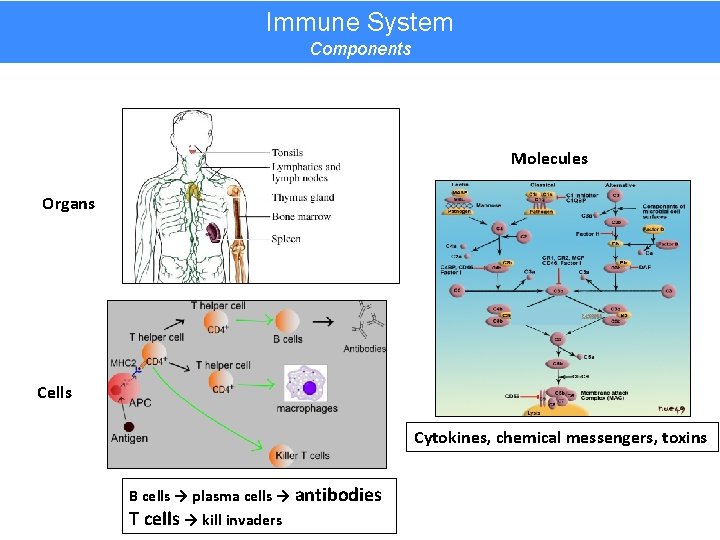
Immune System Components Molecules Organs Cells Cytokines, chemical messengers, toxins B cells → plasma cells → antibodies T cells → kill invaders
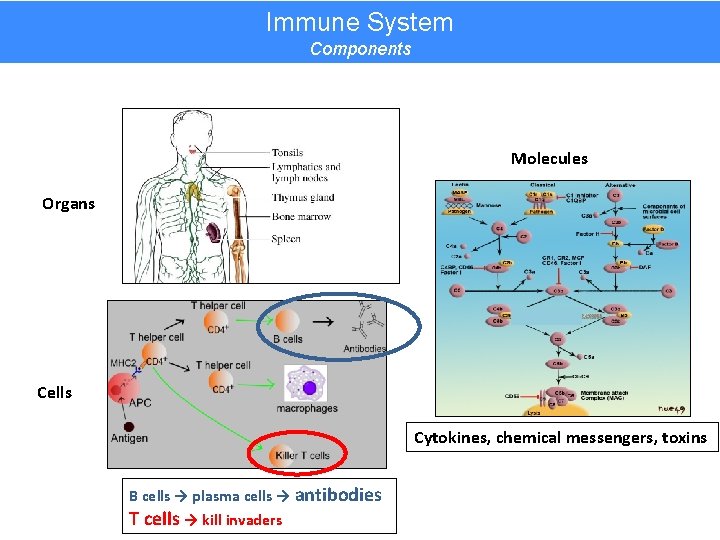
Immune System Components Molecules Organs Cells Cytokines, chemical messengers, toxins B cells → plasma cells → antibodies T cells → kill invaders

Immunotherapy in Myeloma • Antibody treatments – Monoclonal antibodies are injected, travel to the myeloma cells, attach to specific targets on their surface and lead to their destruction – Antibody therapy can consist of • “Naked antibodies” • Conjugated antibodies (antibodies are attached to a toxin to provide additional killing effect on the myeloma cell) • Bi. TES (bispecific T cell engagers) • Cellular therapy – CAR-T (a patient’s T cells are re-engineered to attack and kill myeloma cells)
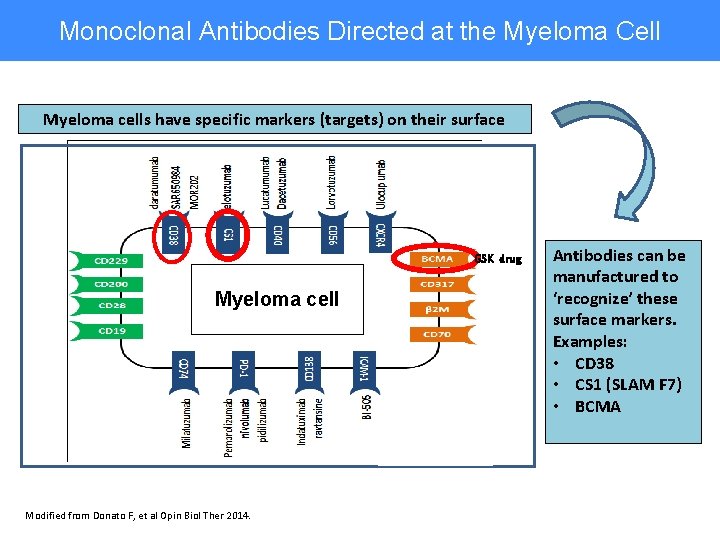
Monoclonal Antibodies Directed at the Myeloma Cell Myeloma cells have specific markers (targets) on their surface GSK drug Myeloma cell Modified from Donato F, et al Opin Biol Ther 2014. Antibodies can be manufactured to ‘recognize’ these surface markers. Examples: • CD 38 • CS 1 (SLAM F 7) • BCMA
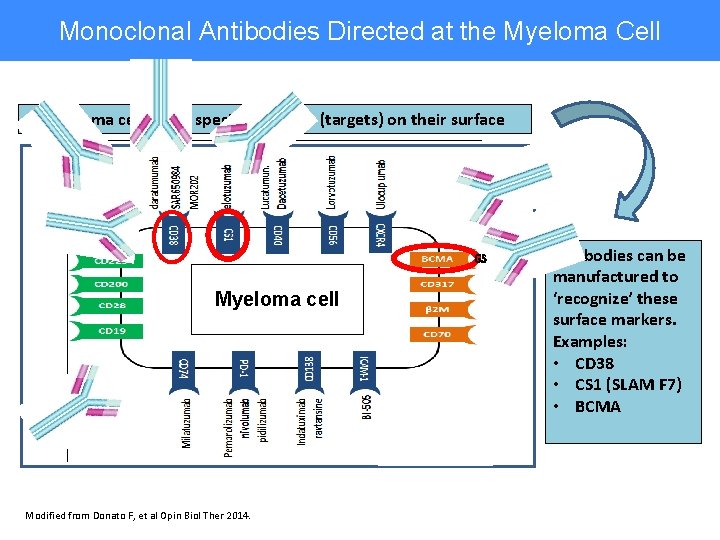
Monoclonal Antibodies Directed at the Myeloma Cell Myeloma cells have specific markers (targets) on their surface GSK drug Myeloma cell Modified from Donato F, et al Opin Biol Ther 2014. Antibodies can be manufactured to ‘recognize’ these surface markers. Examples: • CD 38 • CS 1 (SLAM F 7) • BCMA
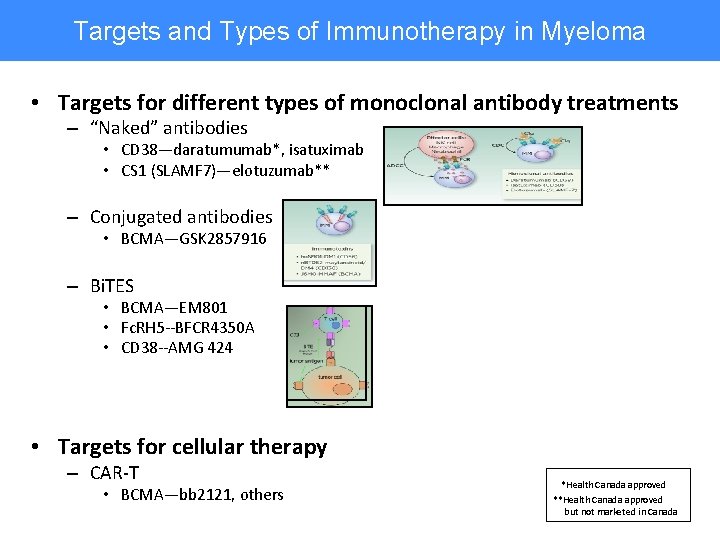
Targets and Types of Immunotherapy in Myeloma • Targets for different types of monoclonal antibody treatments – “Naked” antibodies • CD 38—daratumumab*, isatuximab • CS 1 (SLAMF 7)—elotuzumab** – Conjugated antibodies • BCMA—GSK 2857916 – Bi. TES • BCMA—EM 801 • Fc. RH 5 --BFCR 4350 A • CD 38 --AMG 424 • Targets for cellular therapy – CAR-T • BCMA—bb 2121, others *Health Canada approved **Health Canada approved but not marketed in Canada

Immunotherapy in Myeloma • None of the new immunotherapies for myeloma are funded yet by the provincial Canadian health care systems – Single-agent daratumumab for advanced myeloma available only through some private insurance plans – Daratumumab + lenalidomide + dex (DRd) and daratumumab + bortezomib + dex (DVd) recommended by p. CODR for those with 13 prior regimens, but not yet funded by provinces • May be covered by some private insurance plans • Therefore, we rely on clinical trials (and compassionate programs, if provided) to secure promising treatments to our patients • What trials are active or planned to deliver immunotherapy to Canadian patients?
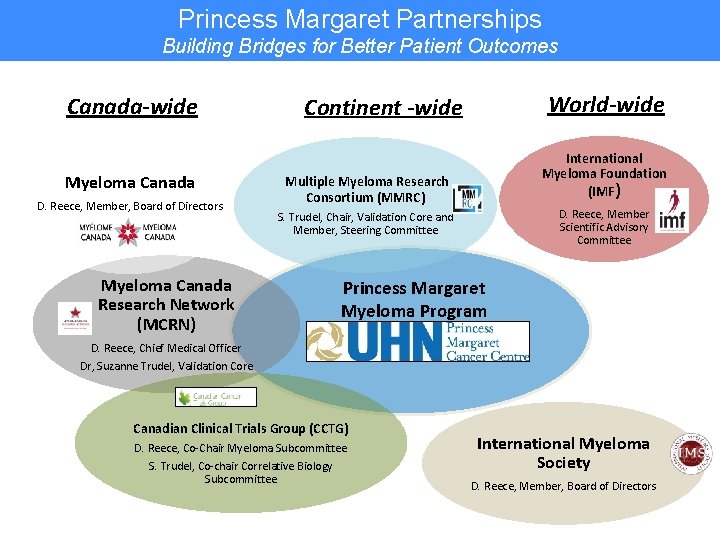
Princess Margaret Partnerships Building Bridges for Better Patient Outcomes Canada-wide Myeloma Canada D. Reece, Member, Board of Directors Myeloma Canada Research Network (MCRN) World-wide Continent -wide International Myeloma Foundation (IMF) Multiple Myeloma Research Consortium (MMRC) D. Reece, Member Scientific Advisory Committee S. Trudel, Chair, Validation Core and Member, Steering Committee Princess Margaret Myeloma Program D. Reece, Chief Medical Officer Dr, Suzanne Trudel, Validation Core Canadian Clinical Trials Group (CCTG) D. Reece, Co-Chair Myeloma Subcommittee S. Trudel, Co-chair Correlative Biology Subcommittee International Myeloma Society D. Reece, Member, Board of Directors

The Princess Margaret Myeloma Program and MCRN work closely together • • • Non-profit organization that is partnered with Myeloma Canada • Collaborates with CCTG • Annual Scientific Roundtable has become the “go to“ myeloma meeting in Canada • Mission To conduct innovative Phase I/II clinical trials and translational research in a collaborative manner to improve patient outcomes To publish evidence-based and peer-reviewed consensus statements on the diagnosis and treatment of myeloma To develop a nationwide myeloma patient data base

Daratumumab Studies at Princess Margaret • Industry-sponsored trials – Lenalidomide + dex +/- daratumumab* in newly diagnosed myeloma patients who are not eligible for stem cell transplant – Carfilzomib + dex +/- daratumumab* for myeloma in relapsed after 1 -3 prior regimens – IV vs SQ daratumumab • Investigator-initiated (IIT) trials – MCRN-004: Dara + cyclophosphamide + dex with pomalidomide initially or at next relapse – MCRN: Possible daratumumab rapid IV infusion# • CCTG (NCIC) trials – MY-12(? ): Cyclophosphamide + dex + ixazomib + dara in newly diagnosed patients who are not eligible for stem cell transplant# *Closed to accrual; patients in follow-up #Planned/under discussion
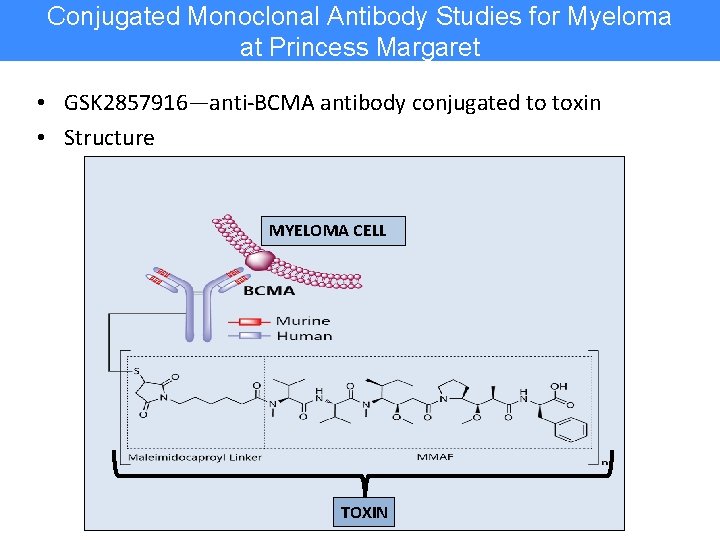
Conjugated Monoclonal Antibody Studies for Myeloma at Princess Margaret • GSK 2857916—anti-BCMA antibody conjugated to toxin • Structure MYELOMA CELL TOXIN

Conjugated Monoclonal Antibody Studies at Princess Margaret (GSK 2857916) • Industry-sponsored – Dr. Trudel presented results at the ASH 2017 meeting 1 – DREAMM-1 single agent study demonstrated unprecedented response rates and duration of response in heavily treated myeloma patients* – Has led to breakthrough therapy designation by the FDA – Additional industry trials planned BCMA Antibody-Drug Conjugate Reaches 60% Response in Myeloma Jason M. Broderick @jasoncology Published: Wednesday, Dec 13, 2017 Suzanne Trudel, MD The B-cell maturation antigen (BCMA) antibody-drug conjugate GSK 2857916 induced an overall response rate (ORR) of 60% in heavily pretreated patients with multiple myeloma, according to findings from the phase I DREAMM-1 study presented at the 2017 ASH Annual Meeting. The 60% (n = 21; 95% CI, 42. 1 -76. 1) ORR included, 2 complete responses (CRs), 1 stringent CR, 15 very good partial responses, and 3 partial responses. The median progression-free survival was 7. 9 months (95% CI, 3. 1 to NE). In November 2017, the FDA granted GSK 2857916 a breakthrough therapy designation for the treatment of patients with relapsed/refractory multiple myeloma who have failed at least 3 prior lines of therapy, including an anti-CD 38 antibody, and who are refractory to a proteasome inhibitor (PI) and an immunomodulatory agent (IMi. D). • Investigator-initiated (IIT) – MCRN-006: GSK + pomalidomide + dex for patients in relapse after lenalidomide planned for later in 2018 • Ability of Canadian investigator to design and implement this trial in Canada is a major coup for our country’s patients 1 Trudel S, et al. Blood 2017; 130: abstract 741. *Closed to accrual; patients in follow-up
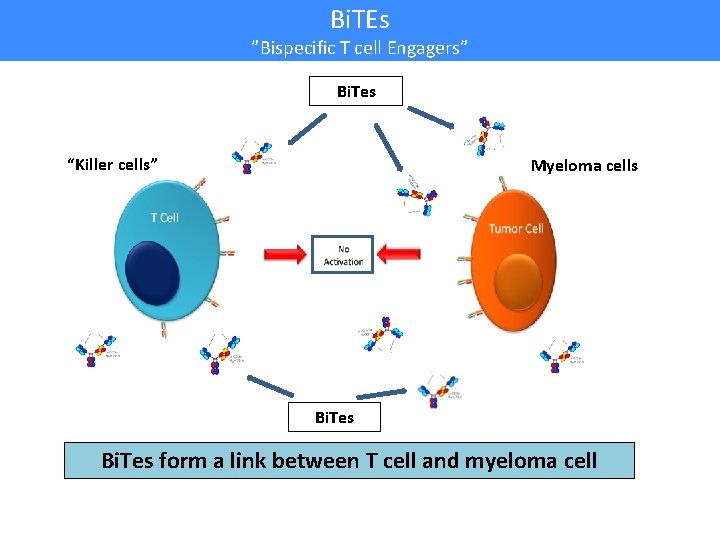
Bi. TEs ”Bispecific T cell Engagers” Bi. Tes “Killer cells” Myeloma cells Bi. Tes form a link between T cell and myeloma cell

Bi. TEs ”Bispecific T cell Engagers” Bi. Tes “Killer cells” Myeloma cells T cells then release substances that enter myeloma cells and kill them
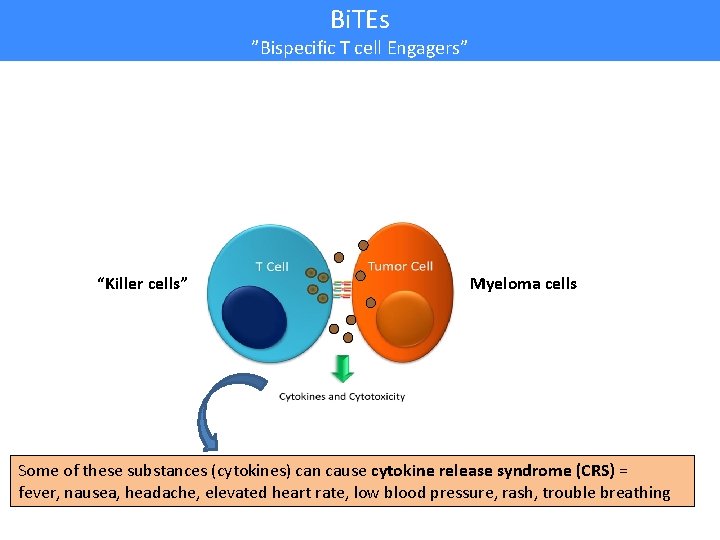
Bi. TEs ”Bispecific T cell Engagers” “Killer cells” Myeloma cells Some of these substances (cytokines) can cause cytokine release syndrome (CRS) = fever, nausea, headache, elevated heart rate, low blood pressure, rash, trouble breathing
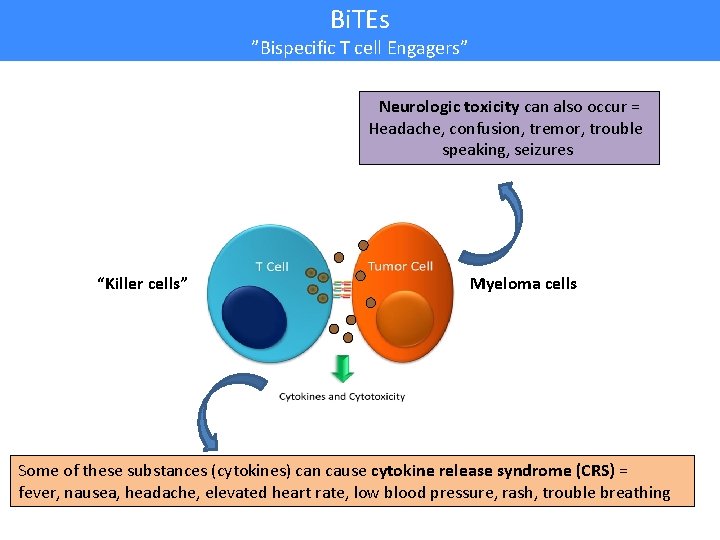
Bi. TEs ”Bispecific T cell Engagers” Neurologic toxicity can also occur = Headache, confusion, tremor, trouble speaking, seizures “Killer cells” Myeloma cells Some of these substances (cytokines) can cause cytokine release syndrome (CRS) = fever, nausea, headache, elevated heart rate, low blood pressure, rash, trouble breathing
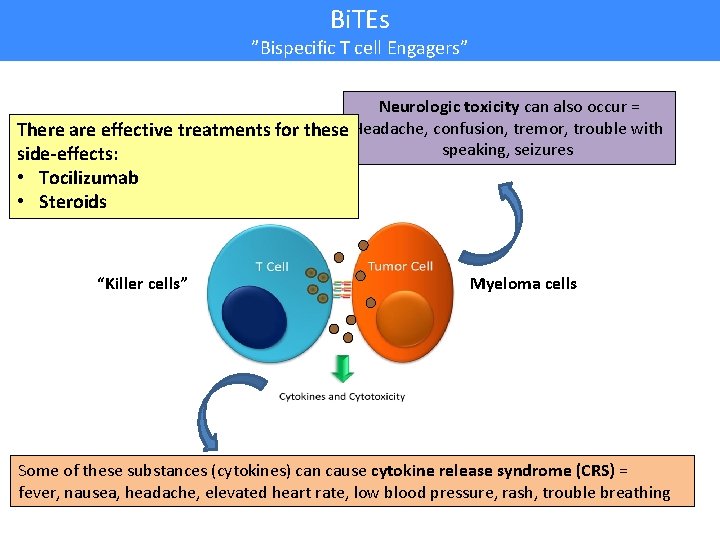
Bi. TEs ”Bispecific T cell Engagers” Neurologic toxicity can also occur = There are effective treatments for these Headache, confusion, tremor, trouble with speaking, seizures side-effects: • Tocilizumab • Steroids “Killer cells” Myeloma cells Some of these substances (cytokines) can cause cytokine release syndrome (CRS) = fever, nausea, headache, elevated heart rate, low blood pressure, rash, trouble breathing

Bi. Te Studies for Myeloma at Princess Margaret • Industry-sponsored – Phase 1 trial of BFCR 4350 A—anti-Fc. RHS 5/anti-CD 3 antibody (bispecific) will open soon • For patients with no other effective treatment available • Basic research—Dr. Rodger Tiedemann Leveraging his discovery to target the “roots” of myeloma with immunotherapy Tiedemann R, et al. Cancer Cell 2013; 24: 289 -304.
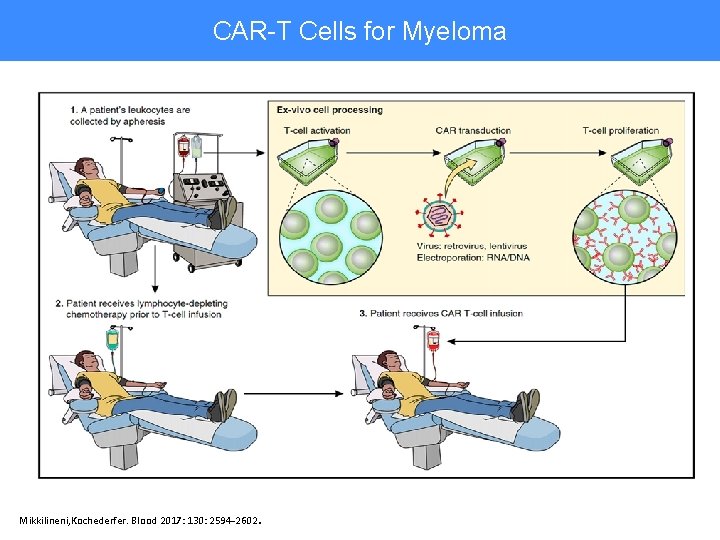
CAR-T Cells for Myeloma CAR-T Cells Therapy Mikkilineni, Kochederfer. Blood 2017: 130: 2594 -2602 .

CAR-T Cells for Myeloma • Promising results of phase 1 bb 2121 trial – Target is BCMA on myeloma cells – At effective dose, 17/18 (94%) responded with 55% CR • • Median of 7 prior treatments 71% progression free at 9 months 71% CRS (tocilizumab given to 4) 2 deaths in remission • Features of upcoming Princess Margaret bb 2121 trial – – Phase 2 study—limited number of patients per site T cells collected ~ 28 days before they are given back High-dose “lymphodepletion” therapy given shortly before CAR-T cell reinfusion 14 day minimum hospitalization required • Encompasses highest risk period for CRS and neurotoxicity • Phase 3 trial comparing BB 2121 with daratumumab-based therapy in advanced myeloma planned by Celgene • Many other institutions are developing their own CAR-T cell products, including Canadian centres Berdeja JG, et al. Blood 2017: 130: Abstract 740 .

Suzanne’s talk
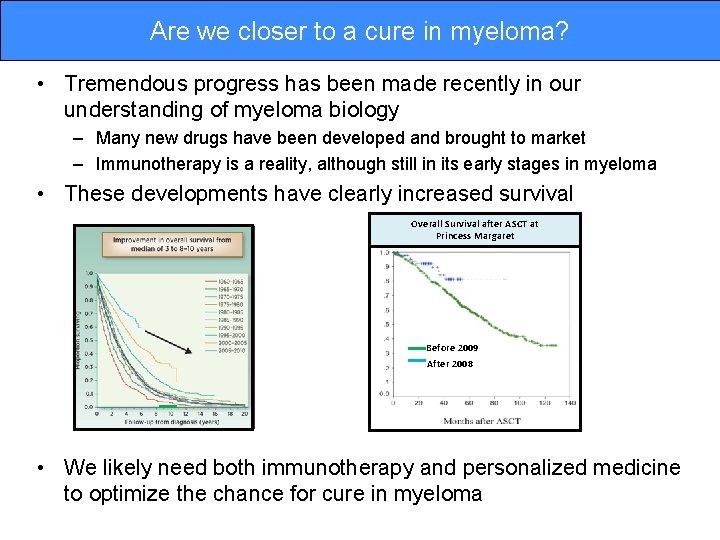
Are we closer to a cure in myeloma? • Tremendous progress has been made recently in our understanding of myeloma biology – Many new drugs have been developed and brought to market – Immunotherapy is a reality, although still in its early stages in myeloma • These developments have clearly increased survival Overall Survival after ASCT at Princess Margaret Before 2009 After 2008 • We likely need both immunotherapy and personalized medicine to optimize the chance for cure in myeloma

Summary and Acknowledgements • The Bloom Chair supports numerous aspects of the Clinical, Database and Laboratory Components of the Princess Margaret Myeloma Research Program and the MCRN • Thank you to the Bloom family, all our other donors and supporters, and, most importantly, our patients and caregivers!
- Slides: 33