Mutation Mutation is defined as any permanent heritable
Mutation • Mutation is defined as any permanent, heritable change in sequence or arrangement of genomic DNA. • Somatic mutations occur in body cells, and cannot be passed on to offspring • A germline mutation occurs in the sex cells, and can be passed to offspring

Sources of DNA mutation • ‘Endogenous’ versus ‘exogenous’ • Endogenous sources include inherent instability of DNA molecule, errors of replication, errors in homologous pairing and recombination, and gene conversion. • Exogenous sources include ionizing radiation, UV radiation, mutagens/intercolators. – Exposures and damage followed by faulty or incomplete repair mechanisms.

Types of point mutations (single base changes) • Missense = point mutation that results in a codon that codes for a different amino acid • Nonsense = point mutation that results in a premature stop codon, which can truncate a protein • Silent mutations = point mutation that results in the same or different amino acid, but with no functional change in the protein

Other types of mutations • Frameshift = insertion or deletion of one or more nucleotides, such that the translational reading frame is shifted • Splice-site mutation = a mutation that inhibits the normal function of a splice donor or acceptor site • Regulatory mutations = affect where or how often a gene is expressed • Expansion or contraction of codon repeats by strand slippage

Other types of mutation • Gene amplification = duplication of a chromosomal region, increasing the dosage of the genes located within that region • Chromosomal translocations = interchange of genetic parts from nonhomologous chromosomes. • Interstitial deletions = an intra-chromosomal deletion that removes a segment of DNA from a single chromosome, thereby juxtaposing previously distant genes. • Chromosomal inversions = reversing the orientation of a chromosomal segment. • Loss of heterozygosity = loss of one allele in an organism that previously had two different alleles.

Strand slippage during replication can lead to net insertion or deletion
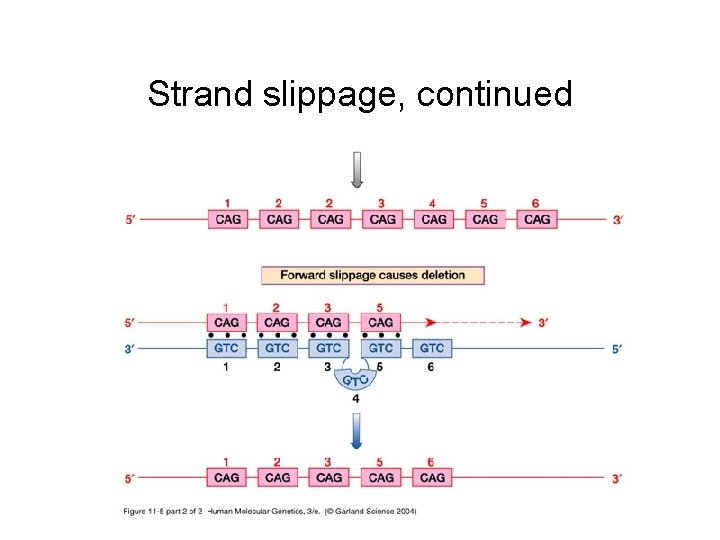
Strand slippage, continued

Examples of actual mutations in disease related genes
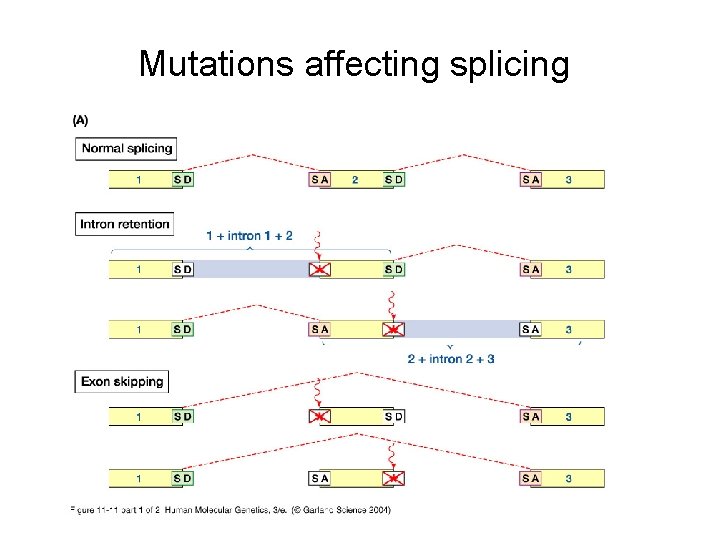
Mutations affecting splicing
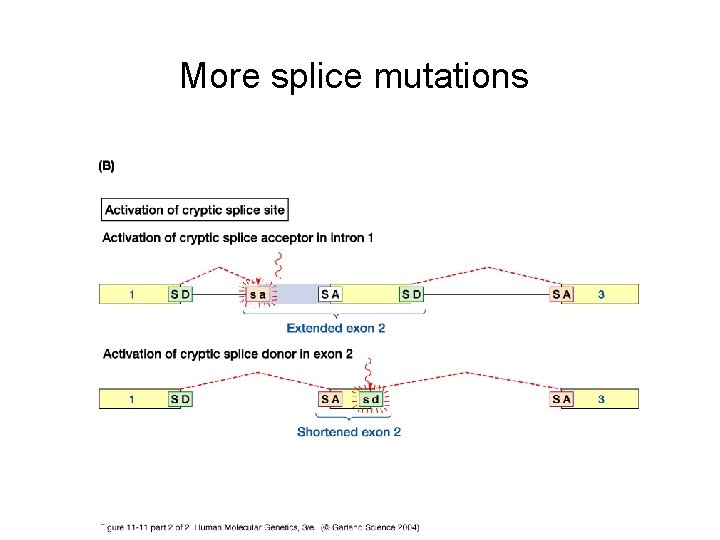
More splice mutations

Cancer, basic concepts • Cancer is not one disease, but many • Related by abnormal cell growth and proliferation with the potential for metastasis • 1/3 to 1/2 of Americans will develop cancer • As burden of infectious and other disease decreases, relative importance of cancer greater • Cancer is clonal in origin • ‘Natural’ end state of multi-cellular organism?

Cancer, basic concepts • Cancer is ALWAYS a genetic disease, but is only occasionally a hereditary disease • Distinction between germline and somatic mutation • Hereditary and sporadic cancers involve mutations in same classes of genes • Depending upon tumor type, ~5 to 10% of cancer cases are due to a strong hereditary predisposition

Cancer, selected definitions • Neoplasm is a mass or tumor • Malignant means that a tumor may invade tissue locally and/or metastasize • Metastasis is the spreading of cancer cells to distant sites • Benign tumors do not metastasize, but may still be very dangerous

Cancer types, broad categories • Carcinoma is derived from epithelial tissue – Ductal carcinoma – Ovarian carcinoma – Colorectal carcinoma • Sarcoma, from mesenchymal tissue – Bone, muscle, connective tissue, soft tissue • Hematopoietic and lymphoid affect marrow, blood, and lymphatic system

Proto-oncogenes • Normal proto-oncogenes promote cell division • When abnormally activated, become oncogenic. – An oncogene is a gene that, when mutated or expressed at high levels, helps turn a normal cell into a tumor cell • Dominant at cell level, single mutant allele enough to cause problems

Proto-oncogenes in sporadic and hereditary cancers • SOMATIC abnormal activation of proto-oncogenes is a common feature of many tumors, sporadic and inherited • GERMLINE mutation in proto-oncogene leading to hereditary cancer syndrome is apparently rare – Often inviable? – Best known example, MEN 2, AD cancer syndrome caused by mutations in ret proto-oncogene – Parathyroid, adrenal tumors – Very high penetrance, prevention by prophylactic thyroidectomy in childhood

Tumor suppressor genes • Normal functions broadly divided – ‘Gatekeepers’ directly down-regulate cell growth (cycle checkpoints, contact inhibition) – ‘Caretakers’ maintain genomic integrity and have indirect effect (repair of DNA damage, replication errors, chromosome breaks) • Loss of function of both alleles • Recessive at cellular level

The cancer-car analogy • Mutations in protooncogenes jam on the accelerator • Mutations in tumor suppressor genes – In gatekeepers, cause brakes to fail – In caretakers, sabotage • Loosen nuts and bolts
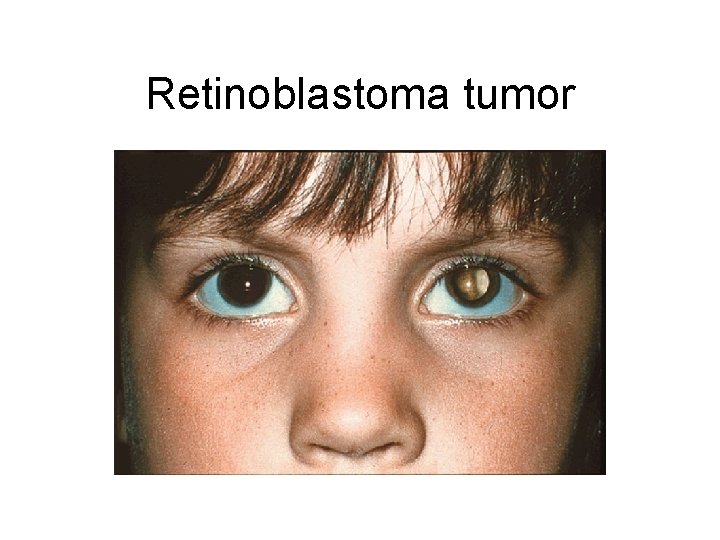
Retinoblastoma tumor
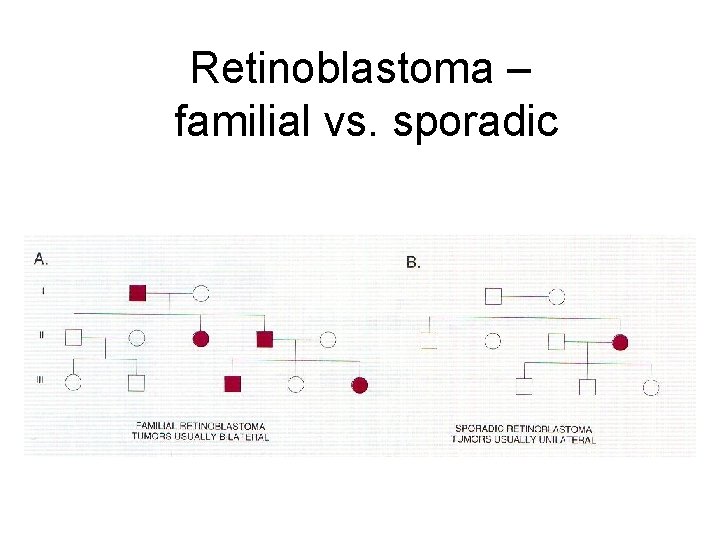
Retinoblastoma – familial vs. sporadic

Retinoblastoma as prototype for tumor-suppressor mutation • Caused by loss of both alleles of crucial gatekeeper, RB 1. • 40% of cases are familial, inherited as autosomal dominant w/ ~90% penetrance. • What is being inherited? – Not cancer per se, but strong single-gene predisposition – Every cell has one RB 1 allele knocked out constitutionally in the germline • Familial cases have earlier onset and may be bilateral
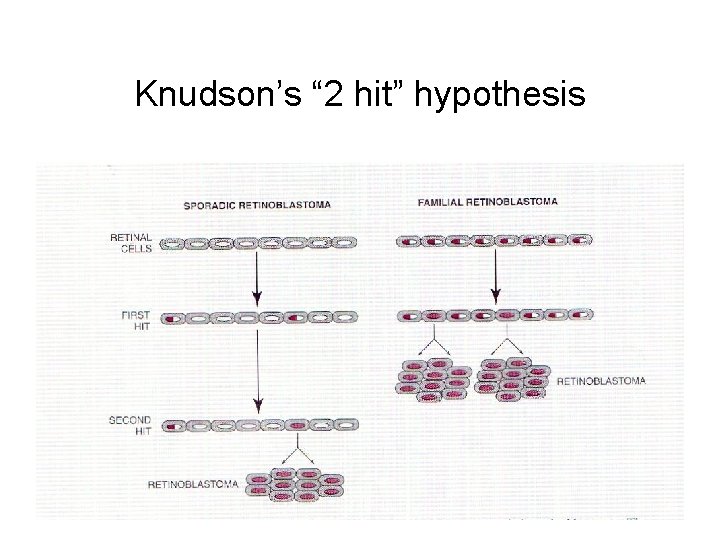
Knudson’s “ 2 hit” hypothesis
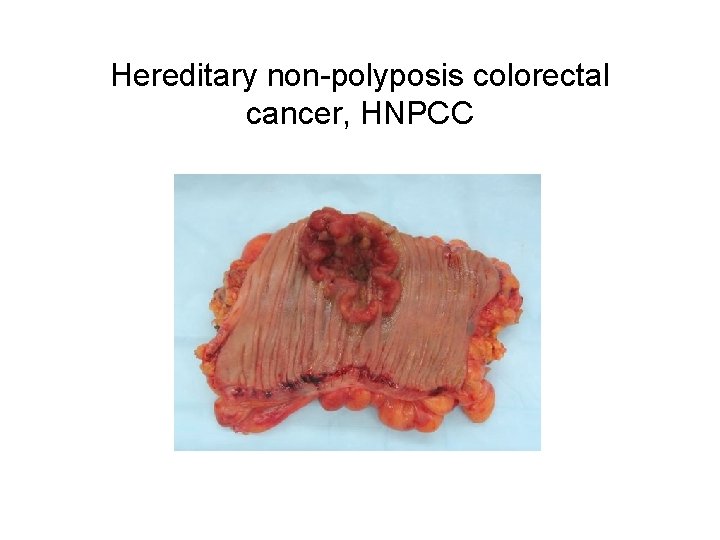
Hereditary non-polyposis colorectal cancer, HNPCC

HNPCC, continued • Accounts for ~2 -5% of all colon cancer • AD disease w/ high penetrance – Lifetime risk of crc ~70 -90% (general population ~ – For women, also risk of endometrial and ovarian cancers • Caused by mutations in one of several mismatch repair genes: h. MLH 1, h. MSH 2 family • Defect in ‘caretaker’ function, LOH observed • Genetic testing is available
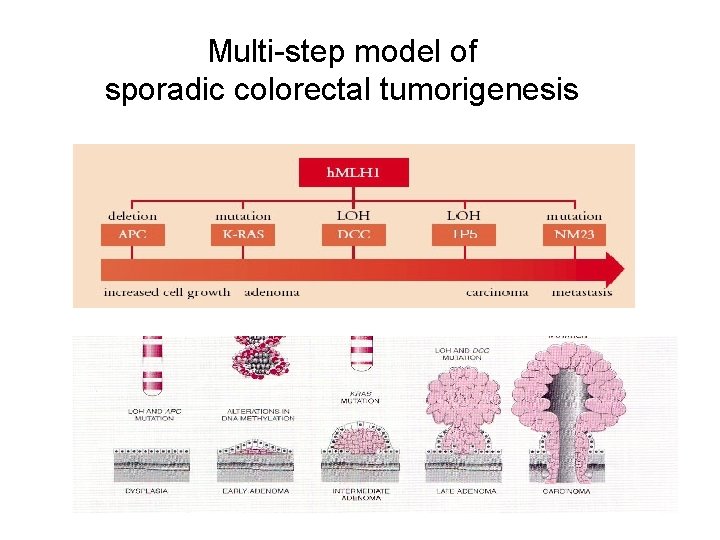
Multi-step model of sporadic colorectal tumorigenesis
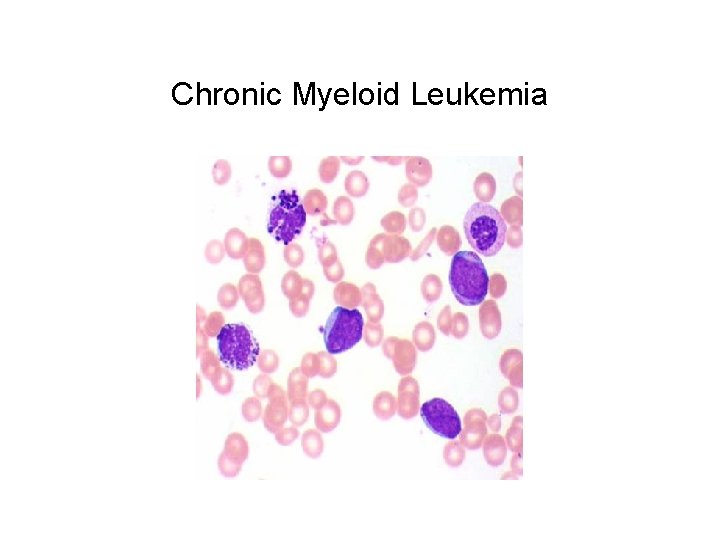
Chronic Myeloid Leukemia

Chronic Myeloid Leukemia • Cancer of the white blood cells • Increased and unregulated growth of myeloid cells in the bone marrow and in the blood • Detected from an acquired chromosomal translocation Philadelphia chromosome • Symptoms include: anemia, increased susceptibility to infections, malaise, low-grade fever

Characteristics of Bcr-Abl • Continuously active • Fusion gene product is a tyrosine kinase • Speeds up cell division • Inhibits DNA repair, which causes genomic instability
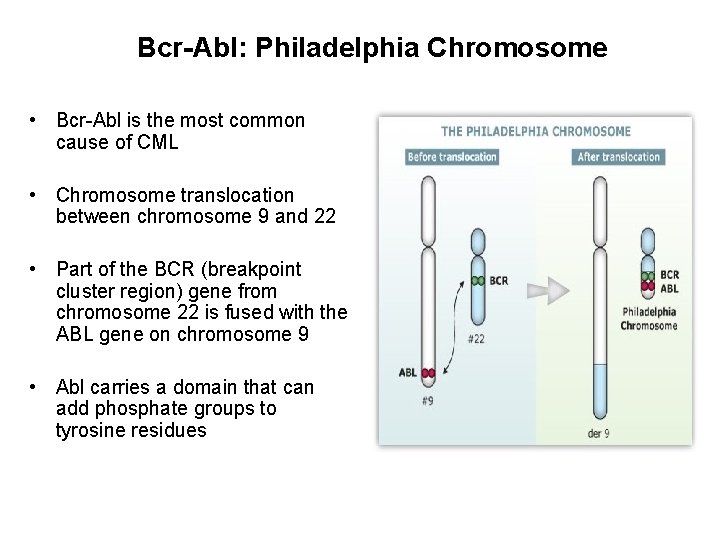
Bcr-Abl: Philadelphia Chromosome • Bcr-Abl is the most common cause of CML • Chromosome translocation between chromosome 9 and 22 • Part of the BCR (breakpoint cluster region) gene from chromosome 22 is fused with the ABL gene on chromosome 9 • Abl carries a domain that can add phosphate groups to tyrosine residues
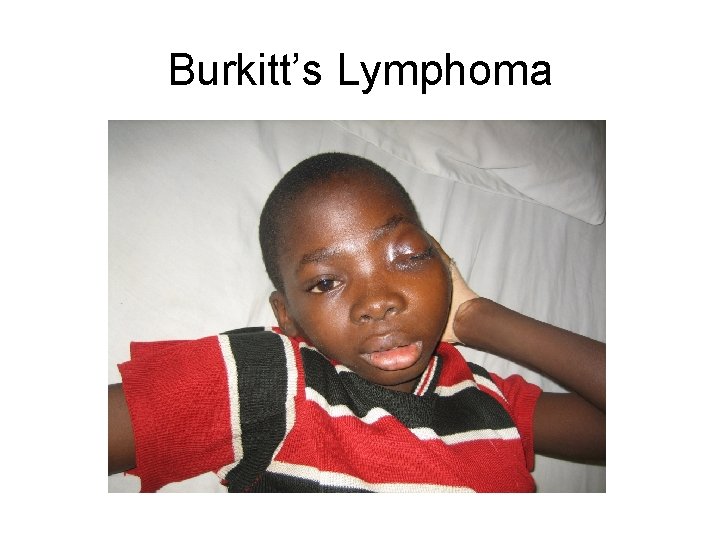
Burkitt’s Lymphoma
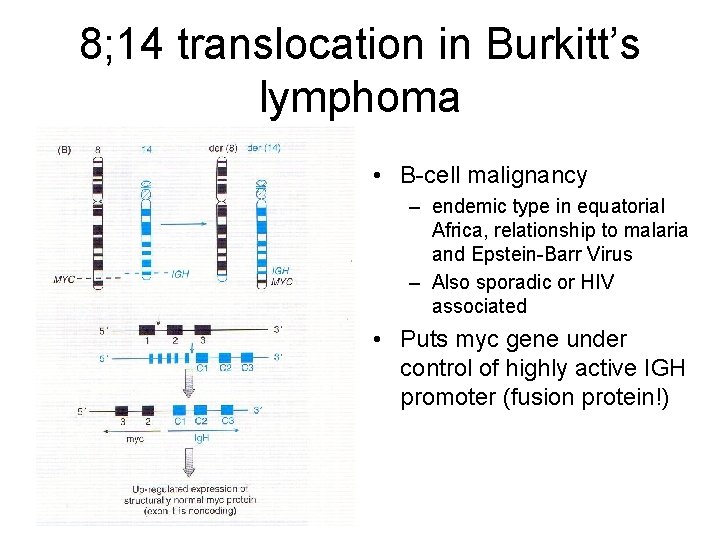
8; 14 translocation in Burkitt’s lymphoma • B-cell malignancy – endemic type in equatorial Africa, relationship to malaria and Epstein-Barr Virus – Also sporadic or HIV associated • Puts myc gene under control of highly active IGH promoter (fusion protein!)
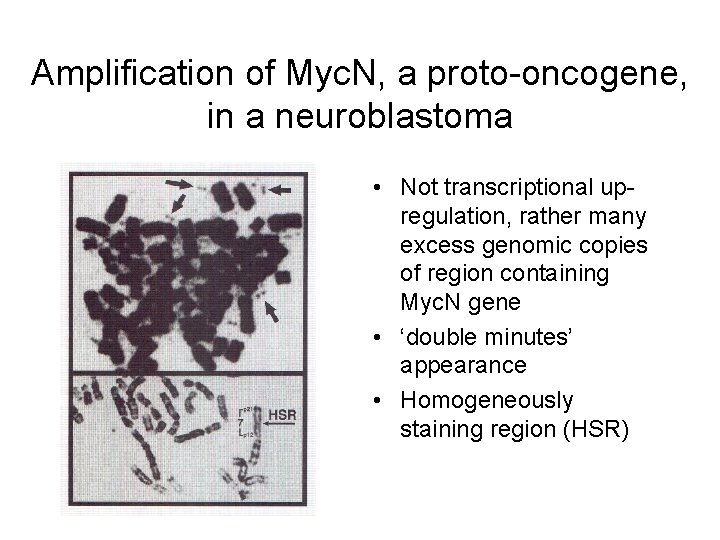
Amplification of Myc. N, a proto-oncogene, in a neuroblastoma • Not transcriptional upregulation, rather many excess genomic copies of region containing Myc. N gene • ‘double minutes’ appearance • Homogeneously staining region (HSR)

Hereditary breast and ovarian cancer Syndrome (HBOC) • BRCA 1 (chrom 17) and BRCA 2 (chrom 13) – account for ~50% of hereditary br ca and ~90% of hereditary br/ov cancer families • AD transmission • mutations in ~1/500 – 1/800 in most populations, many different mutations

Functions of BRCA 1 and BRCA 2 • repair of DNA damage – key trigger in BRCA 1 activation – Interaction w/ RAD 51 DNA repair protein • cell cycle checkpoint control • ubiquitylation (tagging of proteins for degradation by the proteosome)

BRCA 1 -Associated Cancers: Lifetime Risks • breast cancer, ~50 -85%, often early onset – compare to general population risk ~12% • second breast primary, ~40 -60% • ovarian cancer, ~30 -45% – compare to general population risk ~1 -2% • possible modest increases in colon, pancreatic, and male breast cancers

BRCA 2 -Associated Cancers: Lifetime Risk • • breast cancer, ~50 -85% ovarian cancer, ~10 -20% male breast cancer, 5 -10% Also increased risks of pancreatic cancer and melanoma – BRCA 2 accounts for ~6% of familial pancreatic ca

Cancer and the Environment • “Several obstacles, I believe, prevent us from addressing cancer’s environmental roots. An obsession with genes and heredity is one. ”
- Slides: 37