Multivalent ions Word breakdown multi means many and
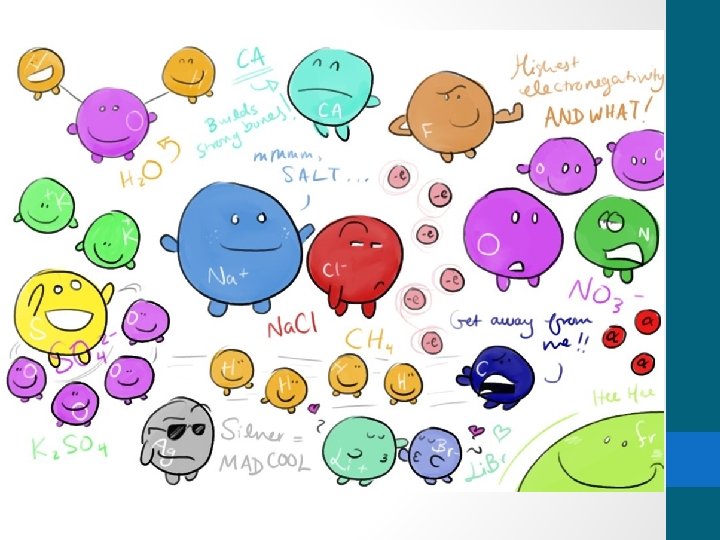

Multivalent ions

Word breakdown!!! • “multi” means many and “valent” refers to valence electrons • Multivalent metals can form two or more different positive ions with different ion charges

• Take out your periodic table or use the on pg. 54 of your textbook. • Find iron (Fe), do you see that there are more than one ion charge? • Iron (Fe) is multivalent b/c it has more than one ion charge listed in the periodic table. • Fe has 2 ion charges: 3+ and 2+

• This means that in some compounds, the iron is Fe 3+ and in other compounds the iron is Fe 2+ • The table always lists the most common ion charge first so for iron, Fe 3+ is more common than Fe 2+ Identify some other multivalent metals. ________________________ What is the most common ion for Cu? , Au? Cu 2+ and Au 3+

• How to distinguish between these different ions? • Use Roman numerals to name each ion. These need to be memorized!

• Examples: • Fe 3+ or iron (III) is pronounced as “iron three” • Fe 2+ or iron (II) is pronounced as “iron two” • Pb 4+ or lead (IV) is pronounced as “lead four” • Cu+ or copper (I) is pronounced as “copper one” When dealing with multivalent metals, you MUST use roman numerals in order to indicate the type of ion that is used.

Rules for writing formulas for compounds with multivalent metals: 1) Identify each ion and its charge 2) Cross the charges 1) Use subscripts to write the formula 3) Double check to make sure that the final formula has the smallest whole number ratio
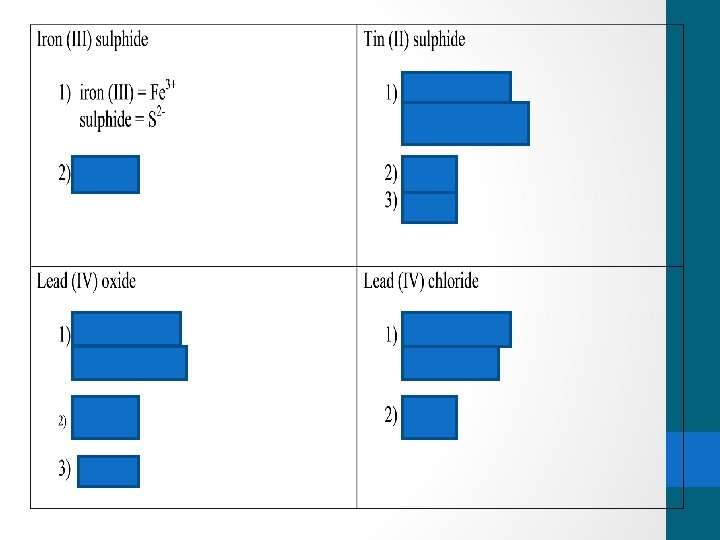

Rules for writing the name for compounds containing multivalent metals: 1) Identify the multivalent metal 1) Identify its different ion forms 2) Note the charge on the negative ion 3) See which ion was used when you tried the “cross the charge” method 4) Write the name using the roman numerals in brackets 5) Write the name of the compound
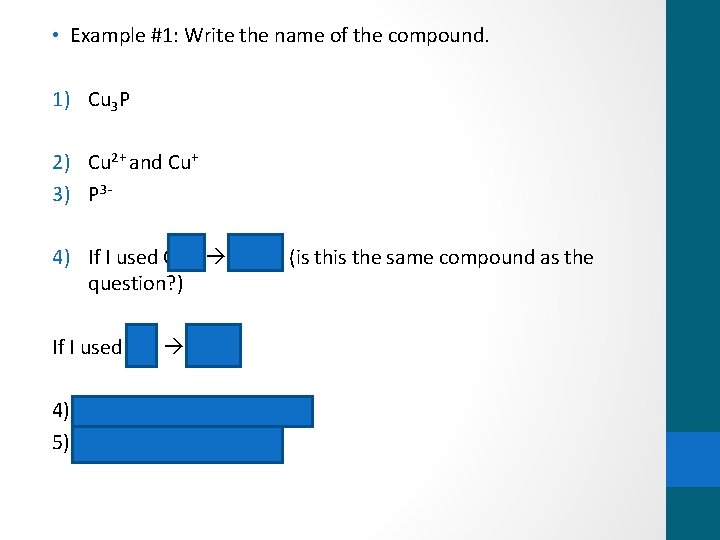
• Example #1: Write the name of the compound. 1) Cu 3 P 2) Cu 2+ and Cu+ 3) P 3 - 4) If I used Cu 2+ Cu 3 P 2 (is the same compound as the question? ) If I used Cu+ Cu 3 P 4) Copper (I) = Cu+ was used 5) Copper (I) phosphide

Practice • Workbook page 47
- Slides: 12