Multiple Myeloma 2013 Update A Keith Stewart MB

Multiple Myeloma: 2013 Update A. Keith Stewart MB. Ch. B. , MBA Anna Maria and Vasek Polak Professor of Cancer Research Dean for Research Mayo Clinic in Arizona Genomies Scottsdale, Arizona Rochester, Minnesota Jacksonville, Florida
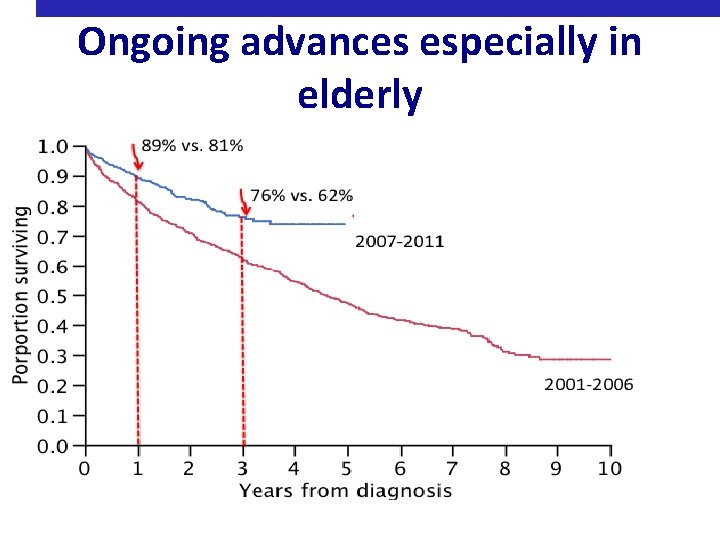
Ongoing advances especially in elderly
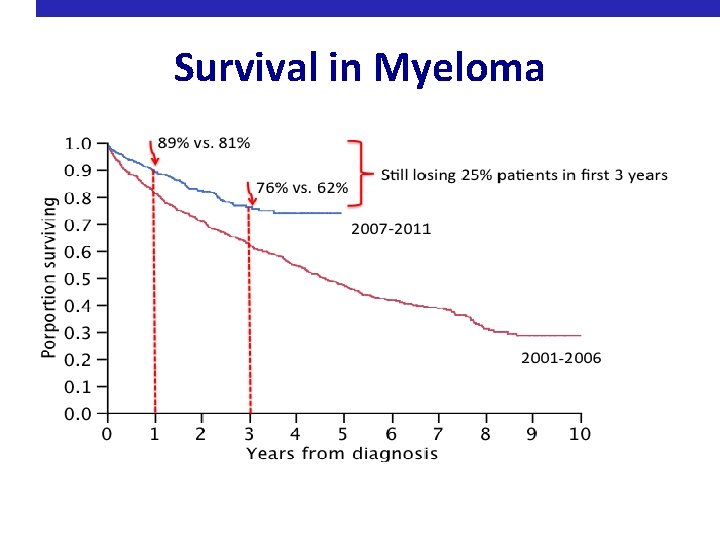
Survival in Myeloma
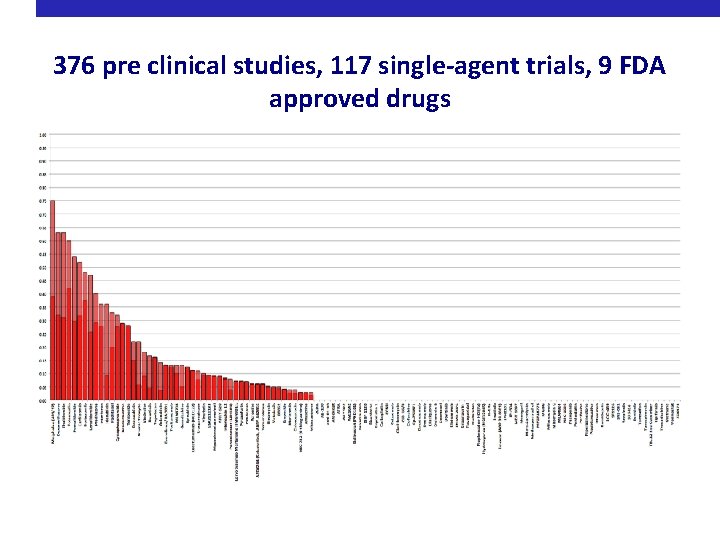
376 pre clinical studies, 117 single-agent trials, 9 FDA approved drugs
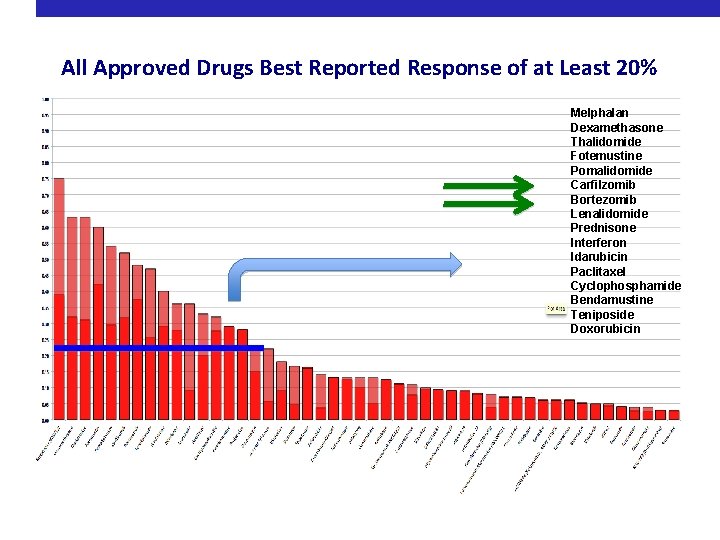
All Approved Drugs Best Reported Response of at Least 20% Melphalan Dexamethasone Thalidomide Fotemustine Pomalidomide Carfilzomib Bortezomib Lenalidomide Prednisone Interferon Idarubicin Paclitaxel Cyclophosphamide Bendamustine Teniposide Doxorubicin

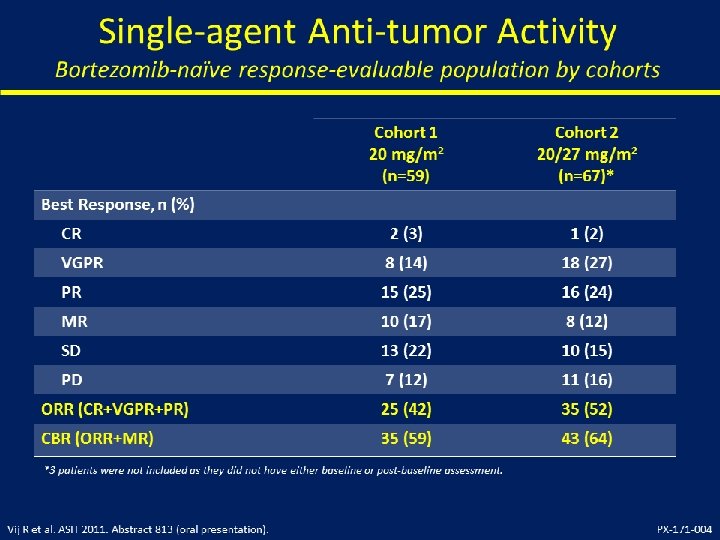
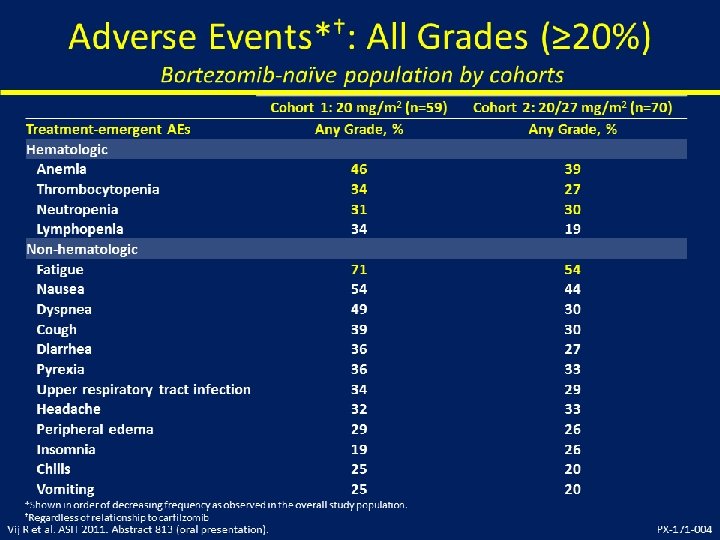
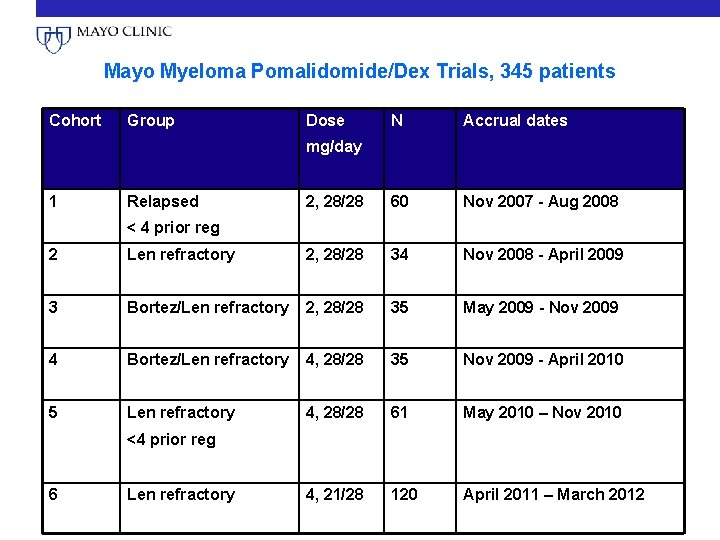
Mayo Myeloma Pomalidomide/Dex Trials, 345 patients Cohort Group Dose N Accrual dates 2, 28/28 60 Nov 2007 - Aug 2008 mg/day 1 Relapsed < 4 prior reg 2 Len refractory 2, 28/28 34 Nov 2008 - April 2009 3 Bortez/Len refractory 2, 28/28 35 May 2009 - Nov 2009 4 Bortez/Len refractory 4, 28/28 35 Nov 2009 - April 2010 5 Len refractory 4, 28/28 61 May 2010 – Nov 2010 4, 21/28 120 April 2011 – March 2012 <4 prior reg 6 Len refractory
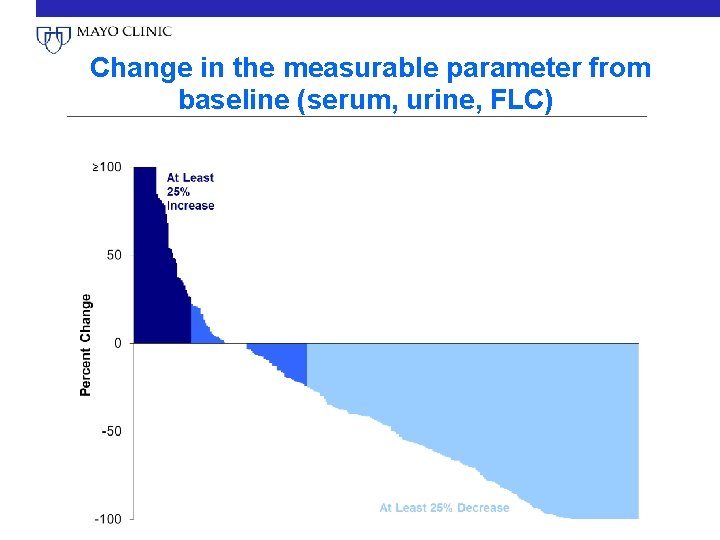
Change in the measurable parameter from baseline (serum, urine, FLC)
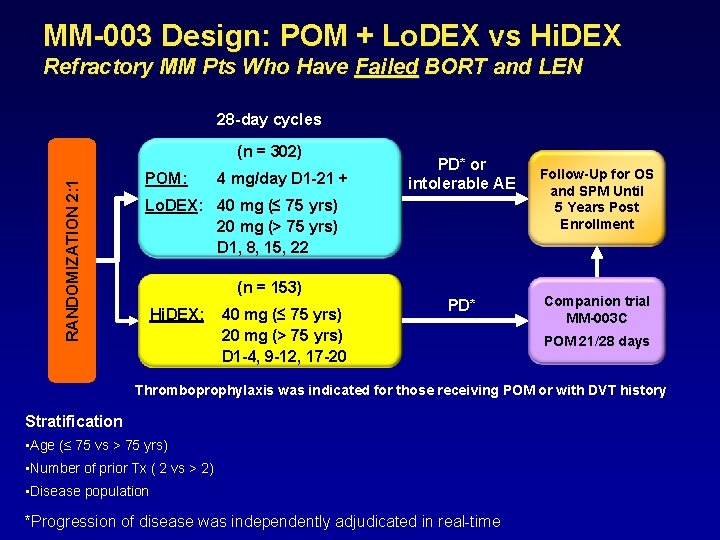
MM-003 Design: POM + Lo. DEX vs Hi. DEX Refractory MM Pts Who Have Failed BORT and LEN 28 -day cycles RANDOMIZATION 2: 1 (n = 302) POM: 4 mg/day D 1 -21 + PD* or intolerable AE Lo. DEX: 40 mg (≤ 75 yrs) 20 mg (> 75 yrs) D 1, 8, 15, 22 (n = 153) Hi. DEX: 40 mg (≤ 75 yrs) 20 mg (> 75 yrs) D 1 -4, 9 -12, 17 -20 PD* Follow-Up for OS and SPM Until 5 Years Post Enrollment Companion trial MM-003 C POM 21/28 days Thromboprophylaxis was indicated for those receiving POM or with DVT history Stratification • Age (≤ 75 vs > 75 yrs) • Number of prior Tx ( 2 vs > 2) • Disease population *Progression of disease was independently adjudicated in real-time
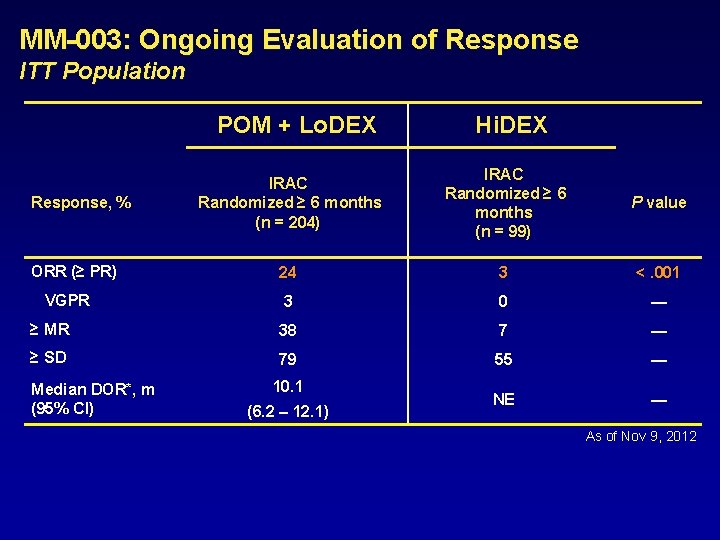
MM-003: Ongoing Evaluation of Response ITT Population POM + Lo. DEX Hi. DEX IRAC Randomized ≥ 6 months (n = 204) IRAC Randomized ≥ 6 months (n = 99) P value 24 3 <. 001 3 0 — ≥ MR 38 7 — ≥ SD 79 55 — NE — Response, % ORR (≥ PR) VGPR Median DOR*, m (95% CI) 10. 1 (6. 2 – 12. 1) As of Nov 9, 2012
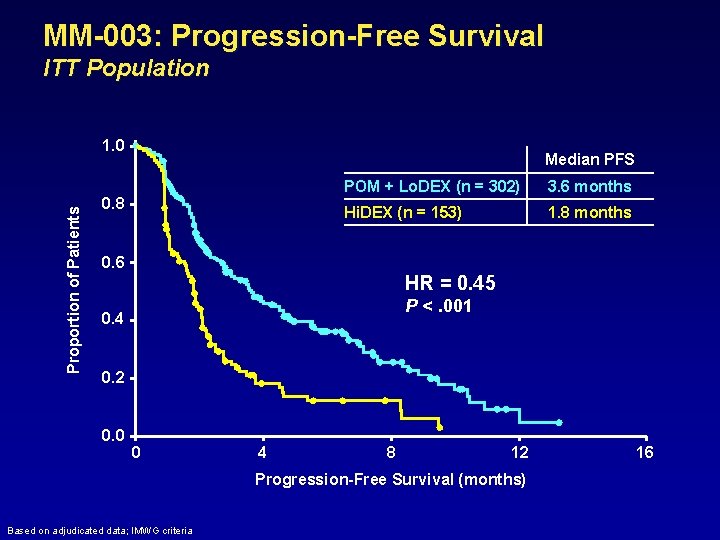
MM-003: Progression-Free Survival ITT Population Proportion of Patients 1. 0 Median PFS 0. 8 POM + Lo. DEX (n = 302) 3. 6 months Hi. DEX (n = 153) 1. 8 months 0. 6 HR = 0. 45 P <. 001 0. 4 0. 2 0. 0 0 4 8 12 Progression-Free Survival (months) Based on adjudicated data; IMWG criteria 16
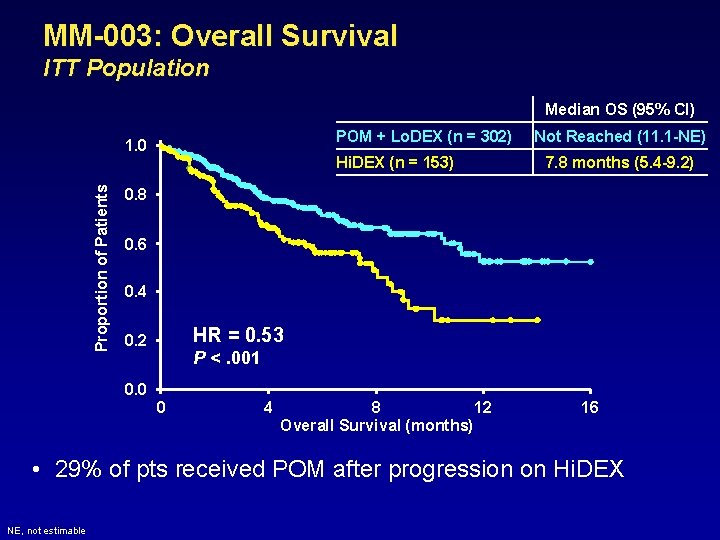
MM-003: Overall Survival ITT Population Median OS (95% CI) POM + Lo. DEX (n = 302) Proportion of Patients 1. 0 Hi. DEX (n = 153) Not Reached (11. 1 -NE) 7. 8 months (5. 4 -9. 2) 0. 8 0. 6 0. 4 HR = 0. 53 0. 2 0. 0 P <. 001 0 4 8 12 Overall Survival (months) 16 • 29% of pts received POM after progression on Hi. DEX NE, not estimable

Conclusions • POM + Lo. DEX significantly improved PFS and OS vs Hi. DEX – Median PFS: 3. 6 vs 1. 8 months • HR = 0. 45; P <. 001 – Median OS: not reached vs 7. 8 months • HR = 0. 53; P <. 001 • Equal benefit in pts refractory to both LEN and BORT • In these heavily pre-treated pts, POM + Lo. DEX was generally well tolerated • POM + Lo. DEX should be considered as a new treatment option for these pts
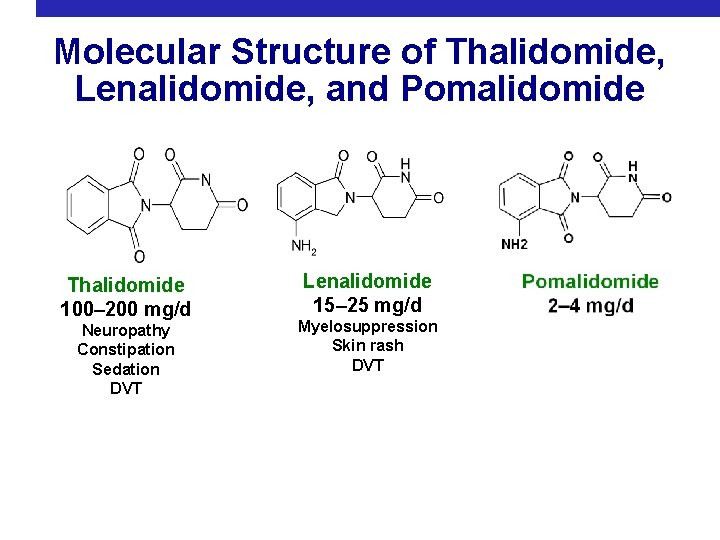
Molecular Structure of Thalidomide, Lenalidomide, and Pomalidomide Thalidomide 100– 200 mg/d Neuropathy Constipation Sedation DVT Lenalidomide 15– 25 mg/d Myelosuppression Skin rash DVT
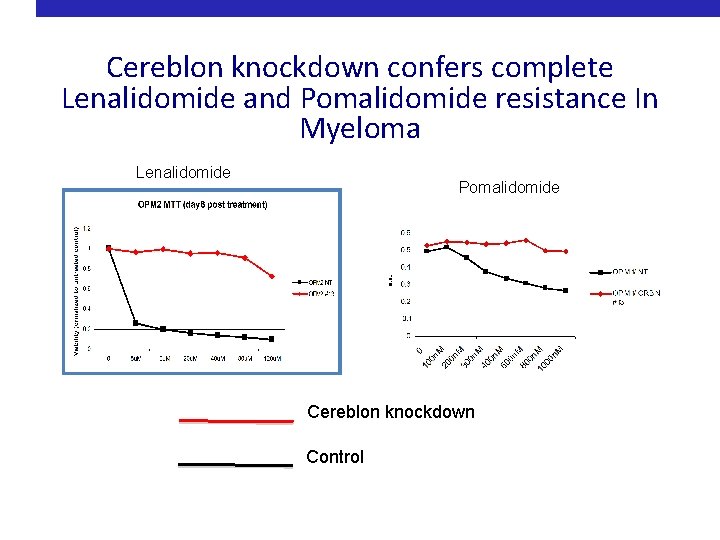
Cereblon knockdown confers complete Lenalidomide and Pomalidomide resistance In Myeloma Lenalidomide Pomalidomide Cereblon knockdown Control
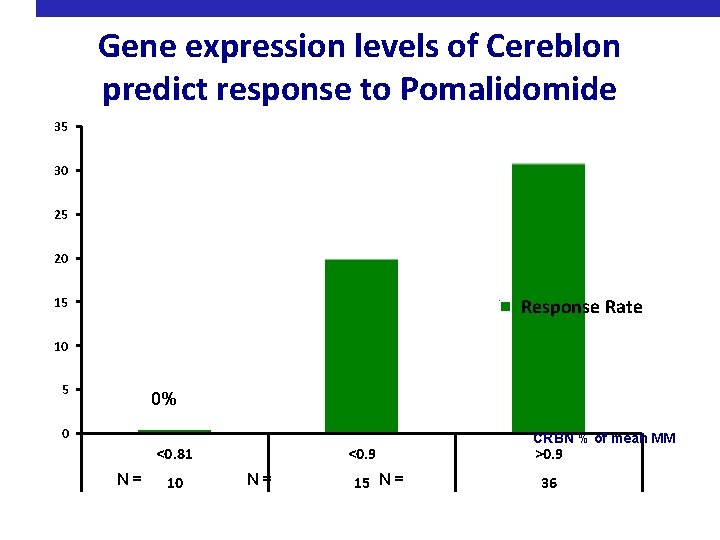
Gene expression levels of Cereblon predict response to Pomalidomide 35 30 25 20 33% 15 10 Response Rate 19% 5 0% 0 <0. 81 N = 10 <0. 9 N = 15 N = CRBN % of mean MM >0. 9 36
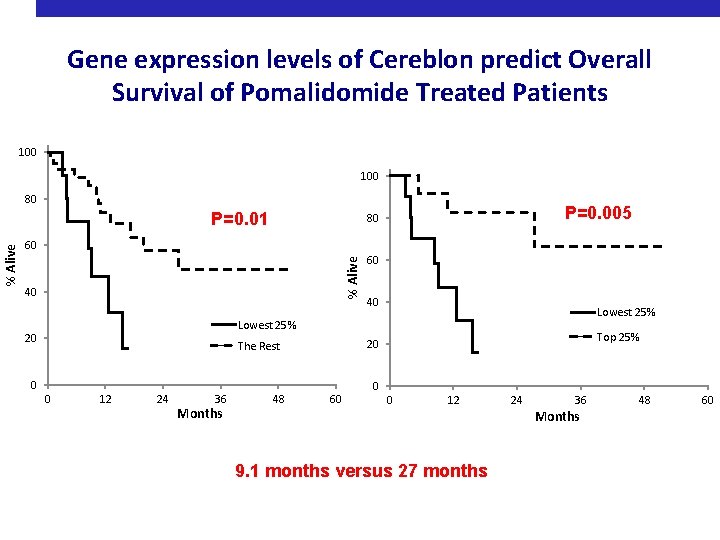
Gene expression levels of Cereblon predict Overall Survival of Pomalidomide Treated Patients 100 80 P=0. 005 80 60 % Alive P=0. 01 40 60 40 Lowest 25% 20 0 0 12 24 36 Months 48 Top 25% 20 The Rest 60 0 0 12 24 36 Months 9. 1 months versus 27 months 48 60
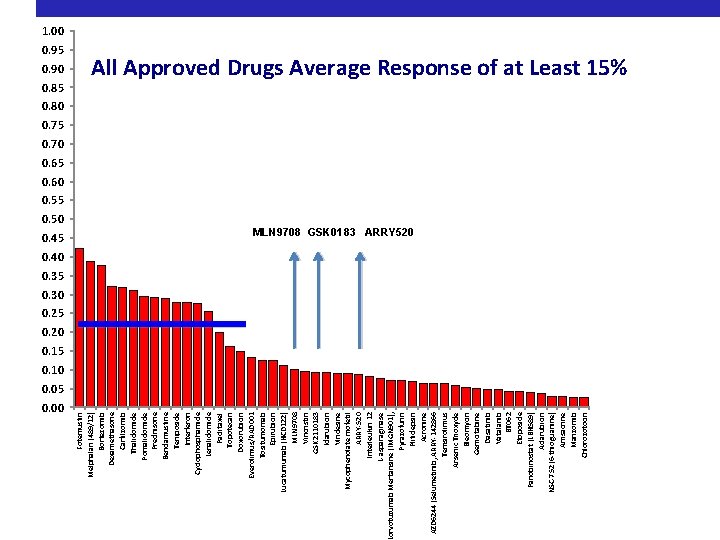
0. 95 0. 90 0. 85 0. 50 0. 45 0. 00 Fotemustin Melphalan (489/12) Bortezomib Dexamethasone Carfilzomib Thalidomide Pomalidomide Prednisone Bendamustine Teniposide Interferon Cyclophosphamide Lenalidomide Paclitaxel Topotecan Doxorubicin Everolimus/RAD 001 Tositumomab Epirubicin Lucatumumab (HCD 122) MLN 9708 Vincristin GSK 2110183 Idarubicin Vindesine Mycophenolate mofetil ARRY-520 Interleukin 12 L-asparaginase orvotuzumab Mertansine (IMGN 901), Pyrazofurin Plitidepsin Acronine AZD 6244 (Selumetinib, ARRY-142866 Temsirolimus Arsenic Trioxyde Bleomycin Gemcitabine Dasatinib Vatalanib BT 062 Etoposide Panobinostat (LBH 589) Aclarubicin NSC-752 (6 -thioguanine) Amsacrine Marizomib Chlorozotocin 1. 00 All Approved Drugs Average Response of at Least 15% 0. 80 0. 75 0. 70 0. 65 0. 60 0. 55 MLN 9708 GSK 0183 ARRY 520 0. 40 0. 35 0. 30 0. 25 0. 20 0. 15 0. 10 0. 05
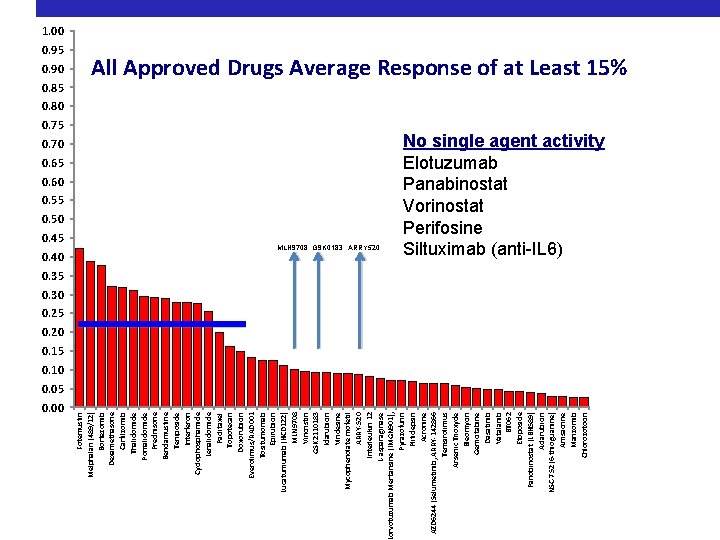
0. 95 0. 90 0. 85 0. 70 0. 65 0. 60 0. 55 0. 50 0. 45 0. 40 0. 00 Fotemustin Melphalan (489/12) Bortezomib Dexamethasone Carfilzomib Thalidomide Pomalidomide Prednisone Bendamustine Teniposide Interferon Cyclophosphamide Lenalidomide Paclitaxel Topotecan Doxorubicin Everolimus/RAD 001 Tositumomab Epirubicin Lucatumumab (HCD 122) MLN 9708 Vincristin GSK 2110183 Idarubicin Vindesine Mycophenolate mofetil ARRY-520 Interleukin 12 L-asparaginase orvotuzumab Mertansine (IMGN 901), Pyrazofurin Plitidepsin Acronine AZD 6244 (Selumetinib, ARRY-142866 Temsirolimus Arsenic Trioxyde Bleomycin Gemcitabine Dasatinib Vatalanib BT 062 Etoposide Panobinostat (LBH 589) Aclarubicin NSC-752 (6 -thioguanine) Amsacrine Marizomib Chlorozotocin 1. 00 All Approved Drugs Average Response of at Least 15% 0. 80 No single agent activity Elotuzumab Panabinostat Vorinostat Perifosine Siltuximab (anti-IL 6) MLN 9708 GSK 0183 ARRY 520 0. 35 0. 30 0. 25 0. 20 0. 15 0. 10 0. 05
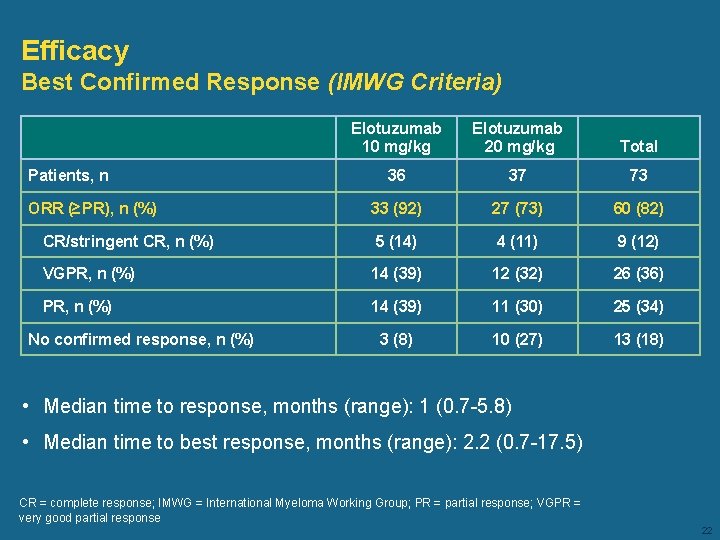
Efficacy Best Confirmed Response (IMWG Criteria) Elotuzumab 10 mg/kg Elotuzumab 20 mg/kg Total 36 37 73 33 (92) 27 (73) 60 (82) CR/stringent CR, n (%) 5 (14) 4 (11) 9 (12) VGPR, n (%) 14 (39) 12 (32) 26 (36) PR, n (%) 14 (39) 11 (30) 25 (34) 3 (8) 10 (27) 13 (18) Patients, n ORR (≥PR), n (%) No confirmed response, n (%) • Median time to response, months (range): 1 (0. 7 -5. 8) • Median time to best response, months (range): 2. 2 (0. 7 -17. 5) CR = complete response; IMWG = International Myeloma Working Group; PR = partial response; VGPR = very good partial response 22

Dinaciclib (SCH 727965) • Dinaciclib is a novel, potent, small molecule inhibitor of cyclin dependent kinases (cdk). • It inhibits cdk 1, cdk 2, cdk 5 and cdk 9 with 50% inhibitory concentrations (IC 50) of 4 n. M, 1 n. M and 4 n. M respectively. • Cyclin D/CDK 4 complexes are inhibited with an IC 50 of 100 n. M.
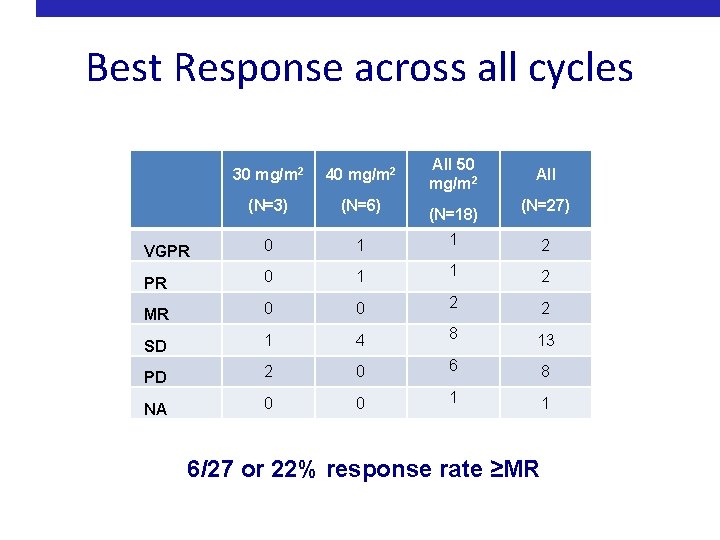
Best Response across all cycles All 50 mg/m 2 30 mg/m 2 40 mg/m 2 (N=3) (N=6) VGPR 0 1 1 2 MR 0 0 2 2 SD 1 4 8 13 PD 2 0 6 8 NA 0 0 1 1 (N=18) All (N=27) 6/27 or 22% response rate ≥MR
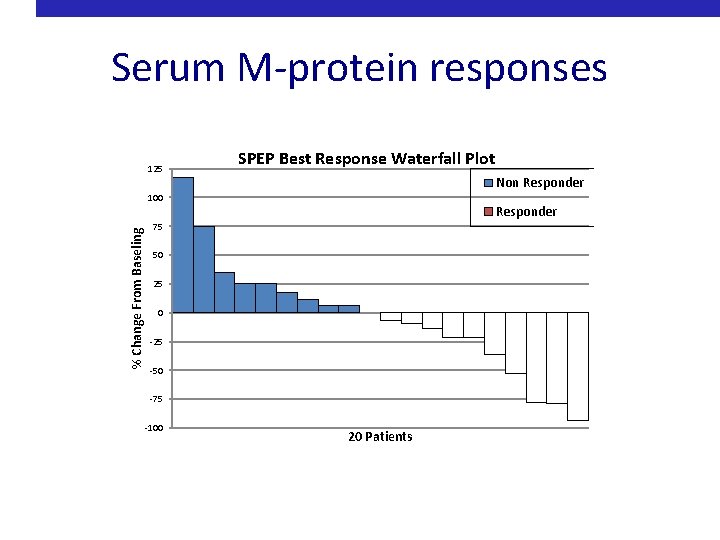
Serum M-protein responses 125 SPEP Best Response Waterfall Plot Non Responder % Change From Baseling 100 Responder 75 50 25 0 -25 -50 -75 -100 20 Patients

Conclusions • Doing better overall - drug combinatons given for longer • High risk disease a major problem • MOA of Lenalidomide better understood • New Agents – Pomalidomide – Carfilzomib – Other investigational
- Slides: 26