MULTIPLE BONDS AND NAMING MOLECULES OF COVALENT BONDS

MULTIPLE BONDS AND NAMING MOLECULES OF COVALENT BONDS Chemistry Unit 3, Day 6
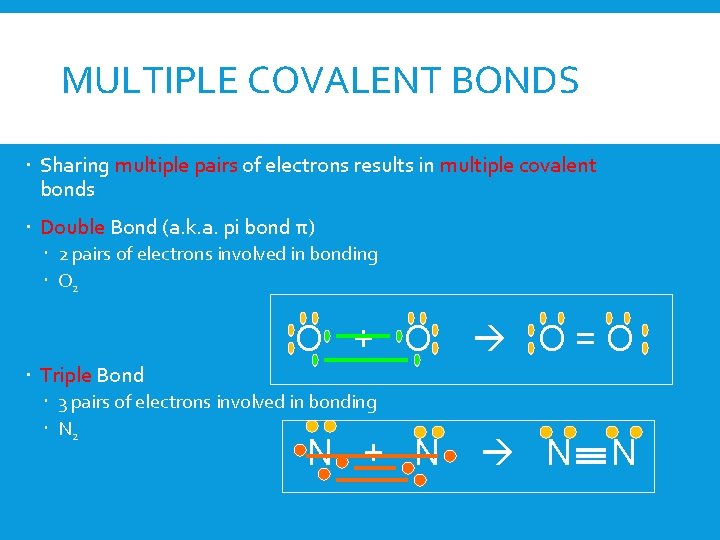
MULTIPLE COVALENT BONDS Sharing multiple pairs of electrons results in multiple covalent bonds Double Bond (a. k. a. pi bond π) 2 pairs of electrons involved in bonding O 2 O + O O=O Triple Bond 3 pairs of electrons involved in bonding N 2 N + N N N

NAMING BINARY MOLECULAR COMPOUNDS There are three rules 1. The first element is given its full name 2. The second element uses the root of its name, but the suffix –ide 3. Both elements use prefixes to express the number of atoms present, but the first element never uses the prefix mono-
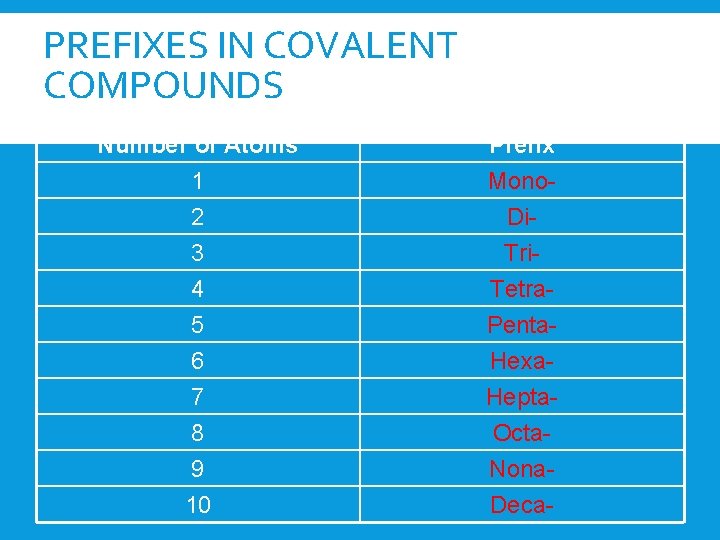
PREFIXES IN COVALENT COMPOUNDS Number of Atoms 1 2 3 Prefix Mono. Di. Tri- 4 5 6 7 8 9 10 Tetra. Penta. Hexa. Hepta. Octa. Nona. Deca-

EXAMPLE PROBLEMS N 2 O – covalent bond 1. First element = nitrogen 2. Second element = oxygen Change oxygen to oxide Dinitrogen monoxide 3. Add prefixes for the number of atoms CO – covalent bond 1. First element = carbon 2. Second element = oxygen Change oxygen to oxide Carbon monoxide 3. Add prefixes for the number of atoms

NAME THE FOLLOWING COMPOUNDS: 1. CO 2 Carbon dioxide 2. SO 2 Sulfur dioxide 3. NF 3 Nitrogen trifluoride 4. CCl 4 Carbon tetrachloride 5. P 2 O 5 Diphosphorous pentaoxide

PROVIDE THE FOLLOWING FORMULAS: 1. Dihydrogen monoxide H 2 O 2. Chlorine trifluoride Cl. F 3 3. Diphosphorous trioxide P 2 O 3 4. Disulfur decafluoride S 2 F 10

GUIDED PRACTICE – THINK, PAIR, SHARE Work with a partner. Give the name And the Lewis Structure: 1. 2. 3. 4. Si. F 4 HBr B 2 H 4 NBr 3 Work with your partner. Give the compound and the Lewis Structure: 1. 2. 3. 4. Sulfur Dichloride Iodine Monofluoride Dicarbon Dihydride Silicon Dioxide

INDEPENDENT PRACTICE – EXIT TICKET 1. Provide the name: C 2 H 6 2. Provide the Lewis Structure: C 2 H 6 3. Provide the formula: Tricarbon octahydride 4. Provide the Lewis Structure: Tricarbon octahydride
- Slides: 9