MULTIPHOTON IONIZATION AND DISSOCIATION OF DIAZIRINE Experiment Dr



- Slides: 19

MULTIPHOTON IONIZATION AND DISSOCIATION OF DIAZIRINE Experiment: Dr. Igor Fedorov Theory: Dr. Lucas Koziol Dr. Andrew Mollner Prof. Hanna Reisler J. Phys. Chem. A Publication Date (Web): April 2, 2009 (Article) DOI: 10. 1021/jp 900204 g $$$: AFOSR and DOE Prof. Anna Krylov

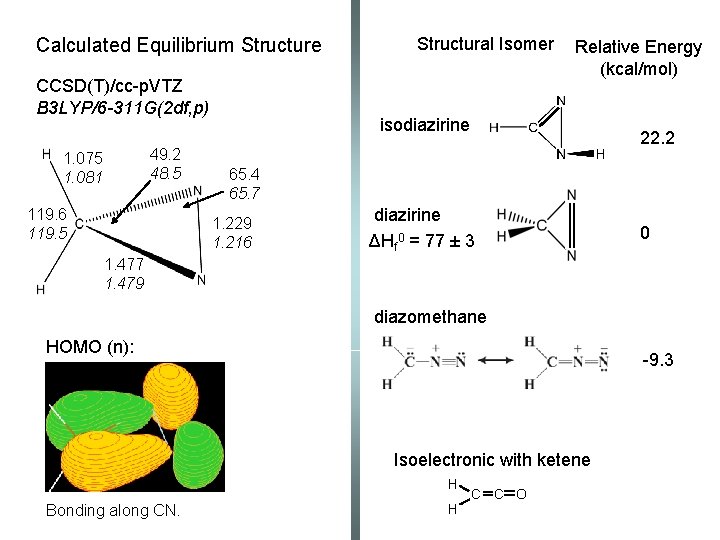
Calculated Equilibrium Structure CCSD(T)/cc-p. VTZ B 3 LYP/6 -311 G(2 df, p) 49. 2 48. 5 1. 075 1. 081 119. 6 119. 5 Structural Isomer Relative Energy (kcal/mol) isodiazirine 22. 2 65. 4 65. 7 1. 229 1. 216 diazirine ΔHf 0 = 77 ± 3 0 1. 477 1. 479 diazomethane HOMO (n): -9. 3 Isoelectronic with ketene H Bonding along CN. H C C O

Why Diazirine? • Source for methylene diradical, : CH 2 – Theory of : CH 2 quite advanced – Electronic states and conical intersections calculated – Experimental results are sparse • Similarity to diazomethane and ketene. – Interesting excited state dynamics

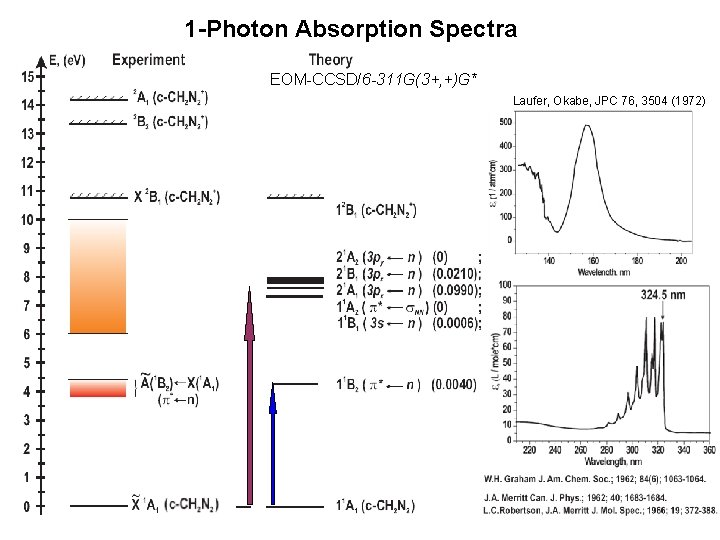
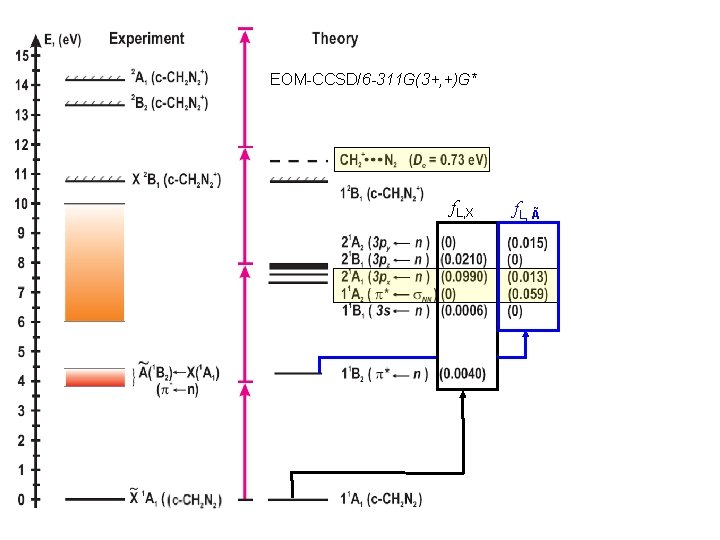
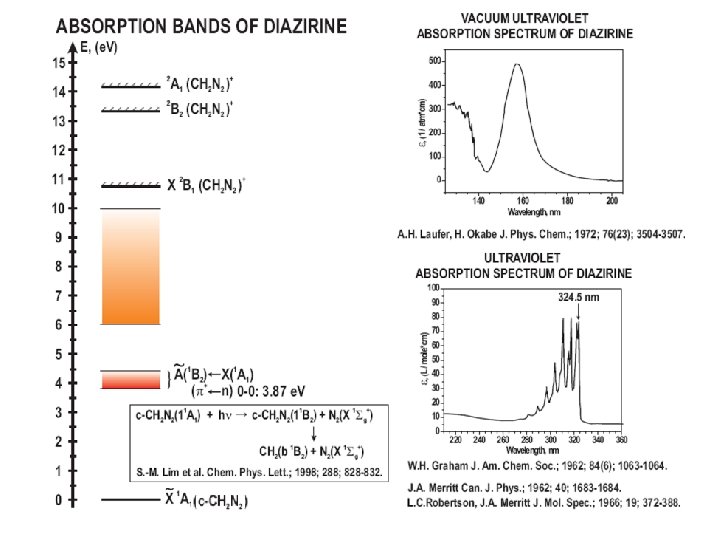
1 -Photon Absorption Spectra EOM-CCSD/6 -311 G(3+, +)G* Laufer, Okabe, JPC 76, 3504 (1972)

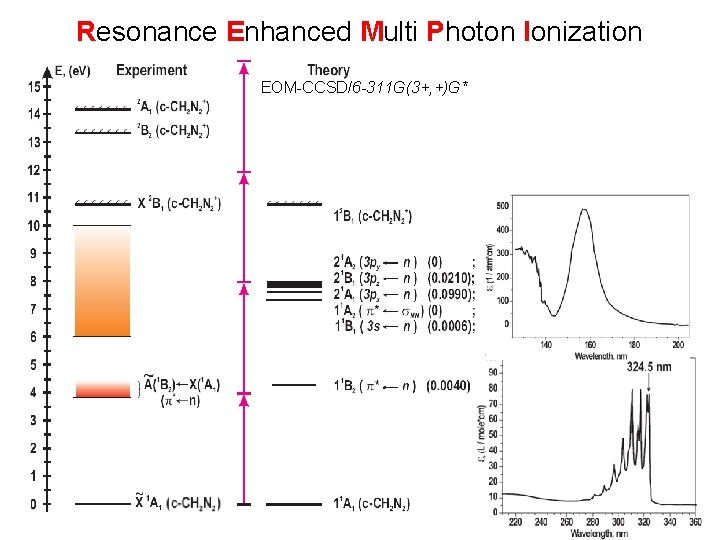
Resonance Enhanced Multi Photon Ionization EOM-CCSD/6 -311 G(3+, +)G*

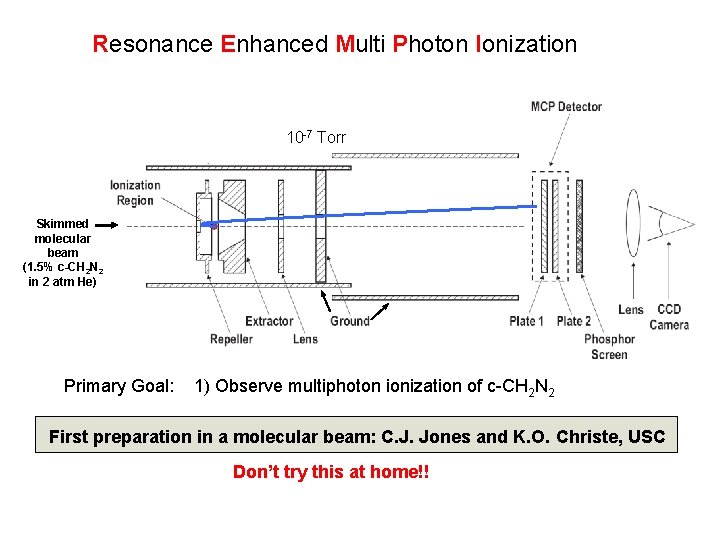
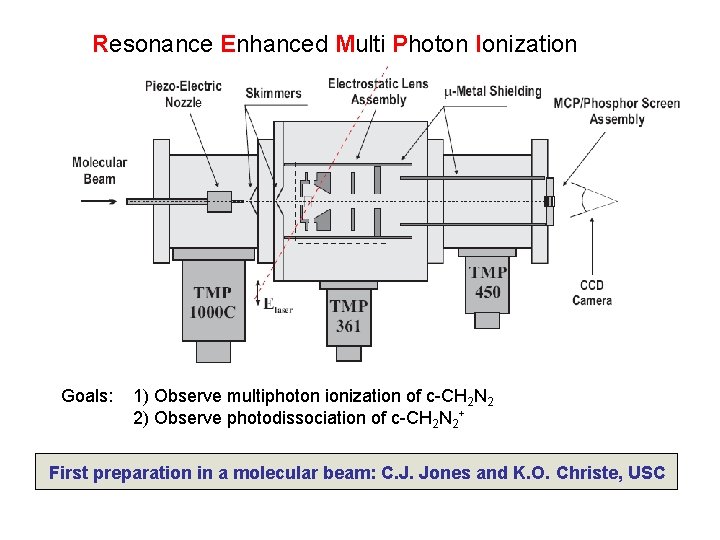
Resonance Enhanced Multi Photon Ionization 10 -7 Torr Skimmed molecular beam (1. 5% c-CH 2 N 2 in 2 atm He) Primary Goal: 1) Observe multiphoton ionization of c-CH 2 N 2 First preparation in a molecular beam: C. J. Jones and K. O. Christe, USC Don’t try this at home!!

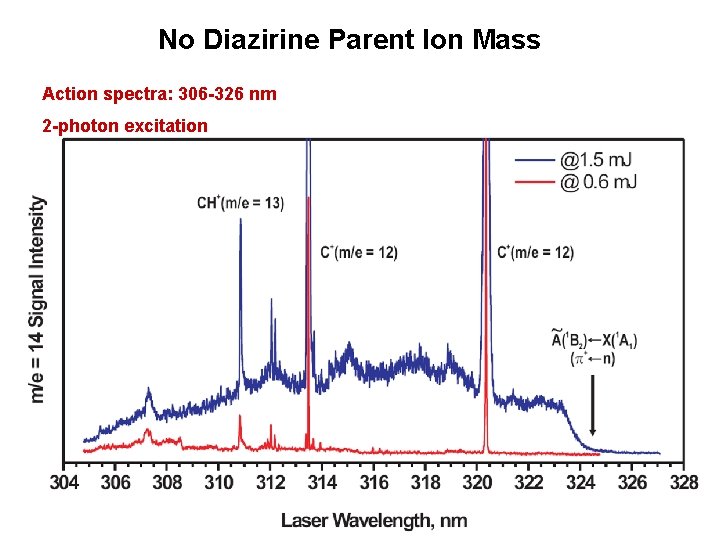
No Diazirine Parent Ion Mass Action spectra: 306 -326 nm 2 -photon excitation

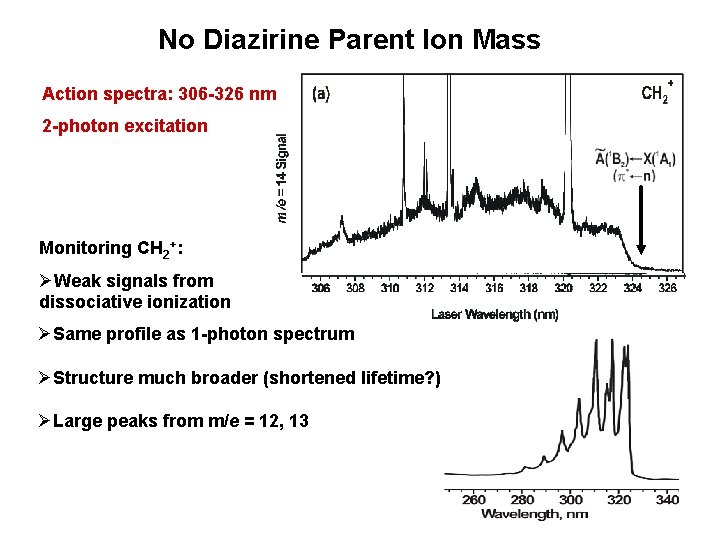
No Diazirine Parent Ion Mass Action spectra: 306 -326 nm 2 -photon excitation Monitoring CH 2+: ØWeak signals from dissociative ionization ØSame profile as 1 -photon spectrum ØStructure much broader (shortened lifetime? ) ØLarge peaks from m/e = 12, 13

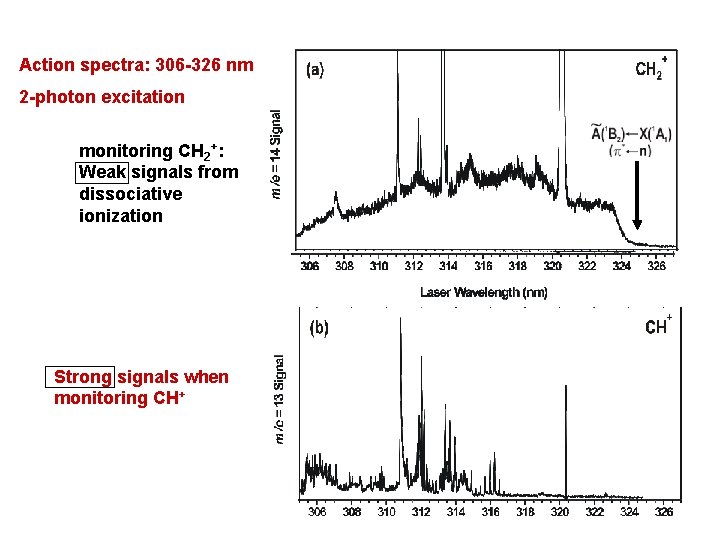
Action spectra: 306 -326 nm 2 -photon excitation monitoring CH 2+: Weak signals from dissociative ionization Strong signals when monitoring CH+

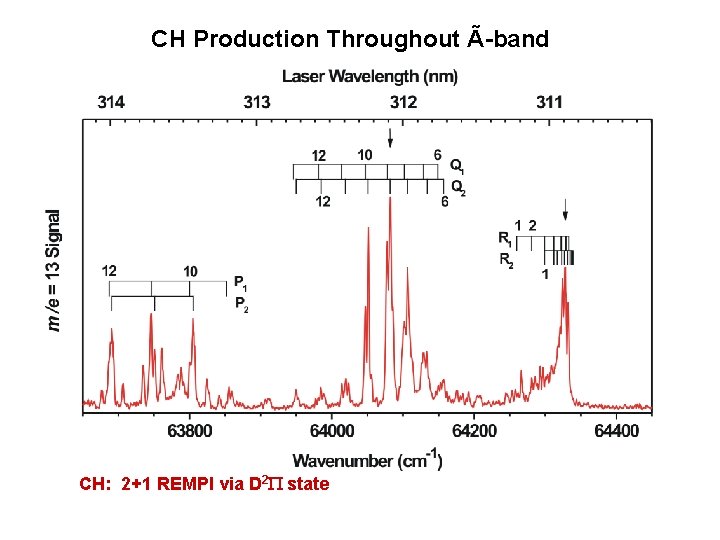
CH Production Throughout Ã-band CH: 2+1 REMPI via D 2 P state

EOM-CCSD/6 -311 G(3+, +)G* f. L, X f. L, Ã

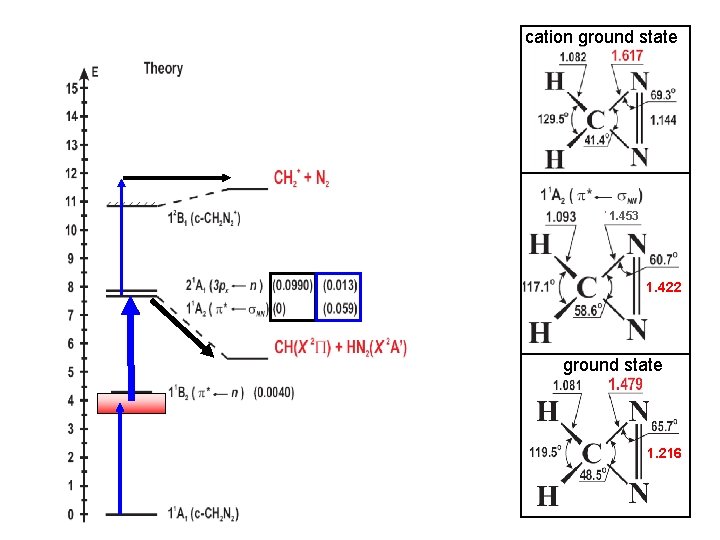
cation ground state 1. 453 1. 422 ground state 1. 216

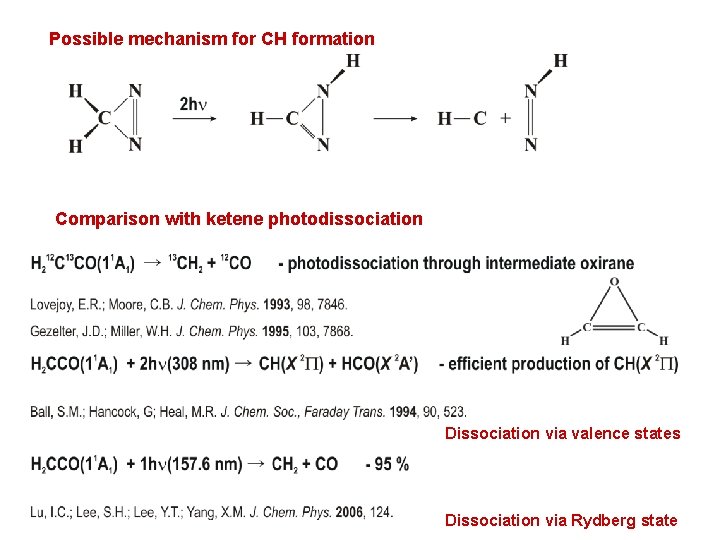
Possible mechanism for CH formation Comparison with ketene photodissociation Dissociation via valence states Dissociation via Rydberg state

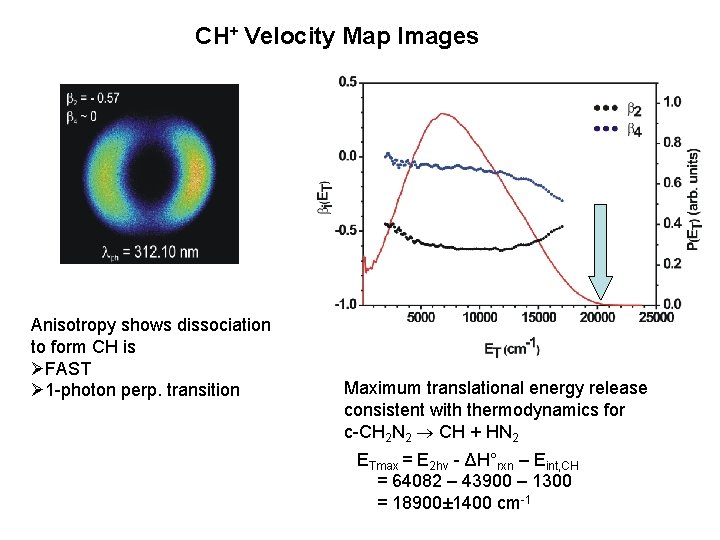
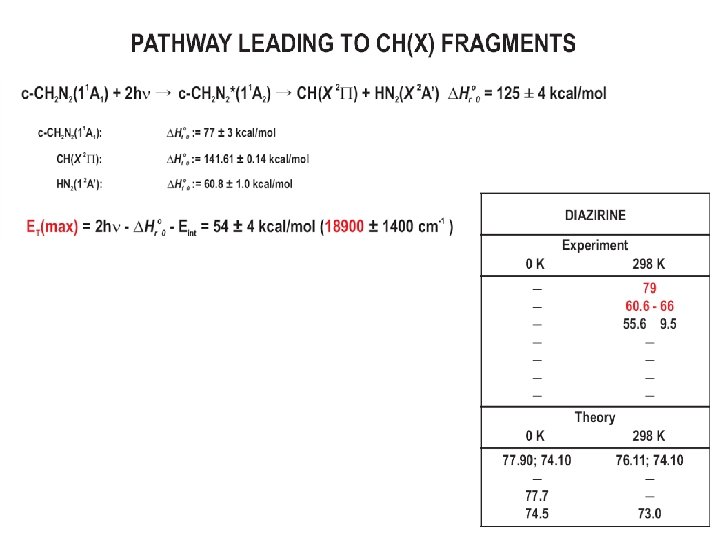
CH+ Velocity Map Images Anisotropy shows dissociation to form CH is ØFAST Ø 1 -photon perp. transition Maximum translational energy release consistent with thermodynamics for c-CH 2 N 2 CH + HN 2 ETmax = E 2 hv - ΔH°rxn – Eint, CH = 64082 – 43900 – 1300 = 18900± 1400 cm-1

Summary ØNo parent ion signal detected by REMPI ØWeak CH 2+ signal from dissociative ionization ØStrong CH+ signal from dissociation from 1 A 2 valence state. Ø CH formation via isomerization to isodiazirine followed by dissociation to CH + HN 2 is consistent with our data ØPES’s involved in CH production are unknown Øisodiazirine dissociation on excited state? ØLarge geometric changes ØMust be very careful going from 1 -photon to 2 -photon ØStates involved can change dramatically ØCH 2 to be continued… J. Phys. Chem. A DOI: 10. 1021/jp 900204 g

Resonance Enhanced Multi Photon Ionization Goals: 1) Observe multiphoton ionization of c-CH 2 N 2 2) Observe photodissociation of c-CH 2 N 2+ First preparation in a molecular beam: C. J. Jones and K. O. Christe, USC

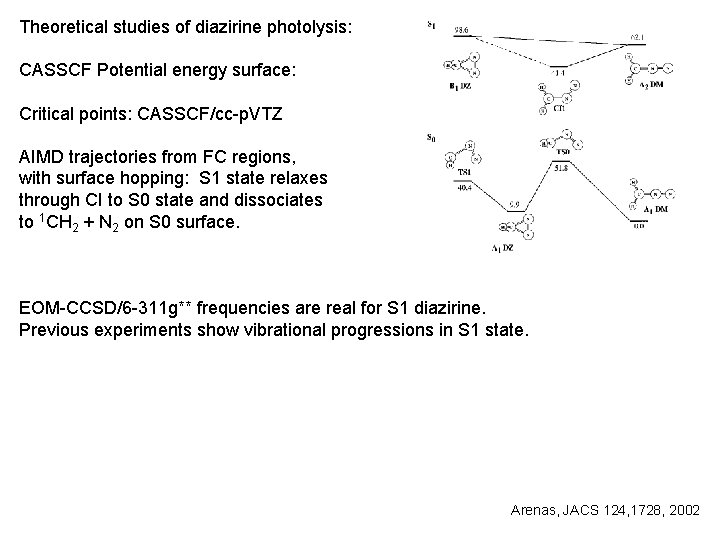
Theoretical studies of diazirine photolysis: CASSCF Potential energy surface: Critical points: CASSCF/cc-p. VTZ AIMD trajectories from FC regions, with surface hopping: S 1 state relaxes through CI to S 0 state and dissociates to 1 CH 2 + N 2 on S 0 surface. EOM-CCSD/6 -311 g** frequencies are real for S 1 diazirine. Previous experiments show vibrational progressions in S 1 state. Arenas, JACS 124, 1728, 2002

