Multiphase System PTT 108 MATERIAL AND ENERGY BALANCE

Multiphase System PTT 108 MATERIAL AND ENERGY BALANCE Noor Amirah Abdul Halim
Introduction �Multiphase systems – mainly involved in separation process. Example: ◦ Distillation : vapor-liquid separation �Driving force of separation : Vapor pressure
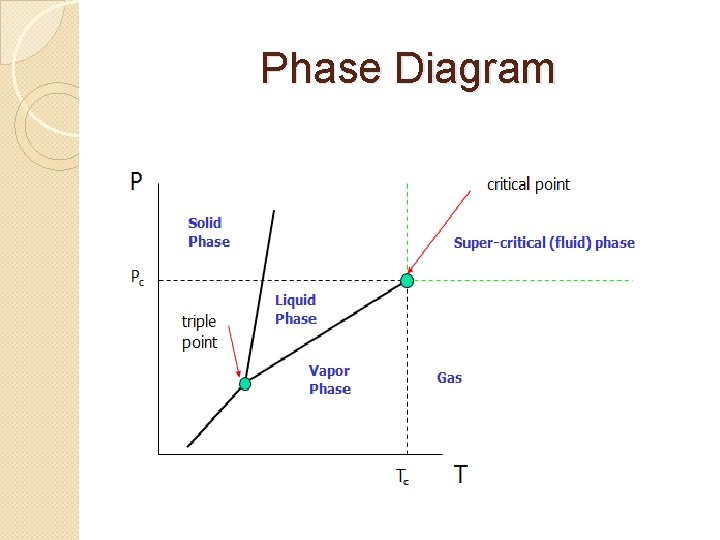
Phase Diagram
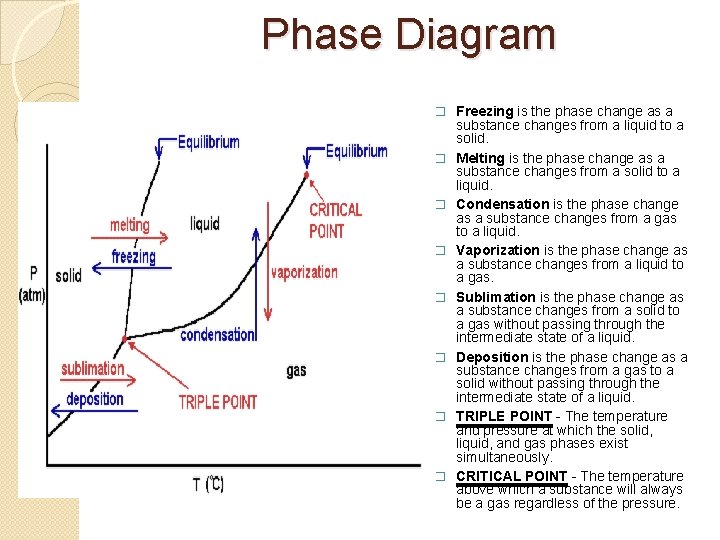
Phase Diagram � Freezing is the phase change as a � � � � substance changes from a liquid to a solid. Melting is the phase change as a substance changes from a solid to a liquid. Condensation is the phase change as a substance changes from a gas to a liquid. Vaporization is the phase change as a substance changes from a liquid to a gas. Sublimation is the phase change as a substance changes from a solid to a gas without passing through the intermediate state of a liquid. Deposition is the phase change as a substance changes from a gas to a solid without passing through the intermediate state of a liquid. TRIPLE POINT - The temperature and pressure at which the solid, liquid, and gas phases exist simultaneously. CRITICAL POINT - The temperature above which a substance will always be a gas regardless of the pressure.

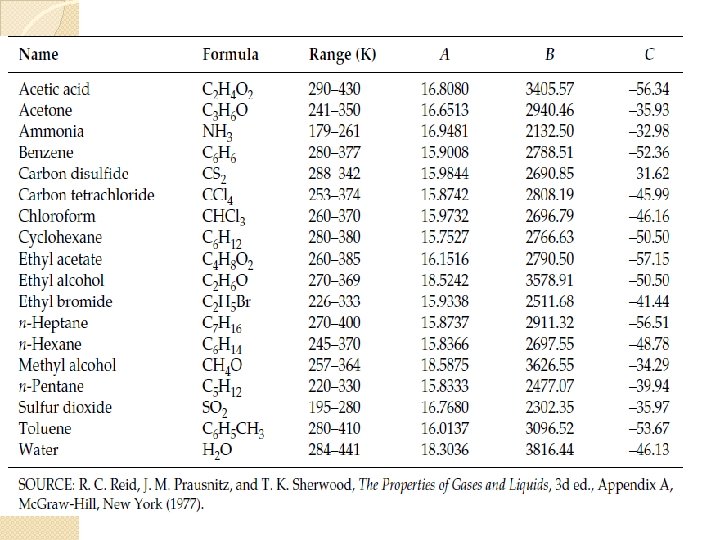
Vapor Pressure � The pressure of vapor when it is in equilibrium with the liquid /solid phase. For a point (T, P) on the vapor-liquid equilibrium curve, P is the vapor pressure of the liquid. For a point (T, P) on the solid-vapor equilibrium curve, P is the vapor pressure of the solid. � Source of vapor pressure data : ◦ Experimental Data from Literature �Perry’s Chemical Handbook �Journals ◦ Equations and Coefficients : Antoine Equation �Perry’s Chemical Handbook �Data Books, Databases ◦ Cox chart ◦ Estimation from Clauisus – Clapeyron Equation

Antoine Equation

Vapor-Liquid Equilibrium (VLE) � Vapor-liquid equilibrium (VLE) curve : the locus of points for which liquid and vapor can coexist � Composition of component between vapor and liquid phase can be determine by phase equilibrium thermodynamics simplified by : ◦ Raoult’s Law �Valid for almost pure component. Similar components ◦ Henry’s Law �Valid for almost dilute component.

VLE- Raoult’s Law � Given a liquid solution with one or more volatile components A, the partial pressure (p. A) in a gas that is in equilibrium with the liquid is assumed to be given by Where: P = y. A = x. A = p. A * = The total pressure, The mole fraction of A in the gas phase, The mole fraction of A in the liquid phase, The vapor pressure of pure liquid A (a function of temperature)

VLE- Henry’s Law � Henry’s Law is applies to vapor-liquid equilibrium when the volatile species A is dilute in the liquid solution. � Given a liquid solution with a volatile component A, the partial pressure of A in a gas mixture in equilibrium with the liquid is given by Where: P = y. A = x. A = HA = The total pressure, The mole fraction of A in the gas phase, The mole fraction of A in the liquid phase, Henry’s law constant of species A (a function of temperature) ( source: perry’s chemical handbook)
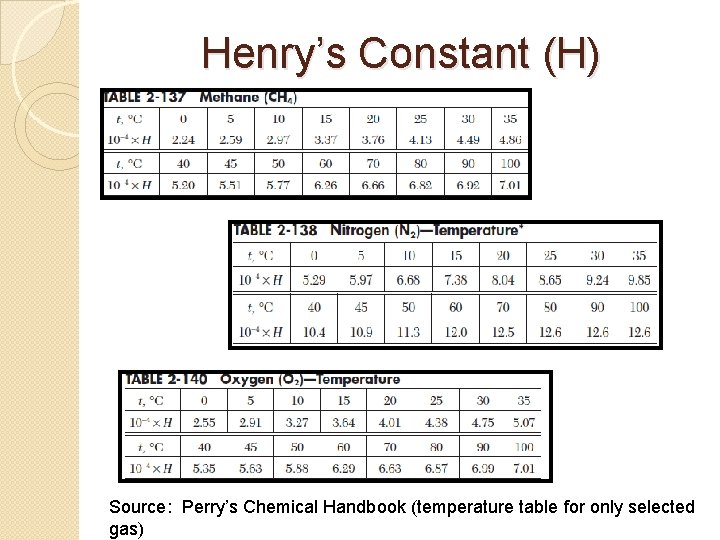
Henry’s Constant (H) Source: Perry’s Chemical Handbook (temperature table for only selected gas)

Example 1 (VLE)

VLE for Ideal Solution- Mixture of components �Ideal solution- is a mixture that follows Raoult's law, which states the vapor pressure above a solution is directly proportional to the mole fraction of the solvent. �Bubble point - is the point at which the first drop of a liquid mixture begins to vaporize. �Dew point - is the point at which the first drop of a gaseous mixture begins to condense.

VLE for Ideal Solution- Mixture of components � Bubble point temperature ( Tbp) Temperature at which the first drop of a liquid mixture begins to vaporize at constant pressure � Dew point temperature ( Tdp) Temperature at which the first drop of a gaseous mixture begins to condense at constant pressure � Bubble point pressure ( Pbp) Pressure at which the first drop of a liquid mixture begins to vaporize at constant temperature � Dew point pressure ( Pdp) Pressure at which the first drop of a gaseous mixture begins to condense at constant temperature
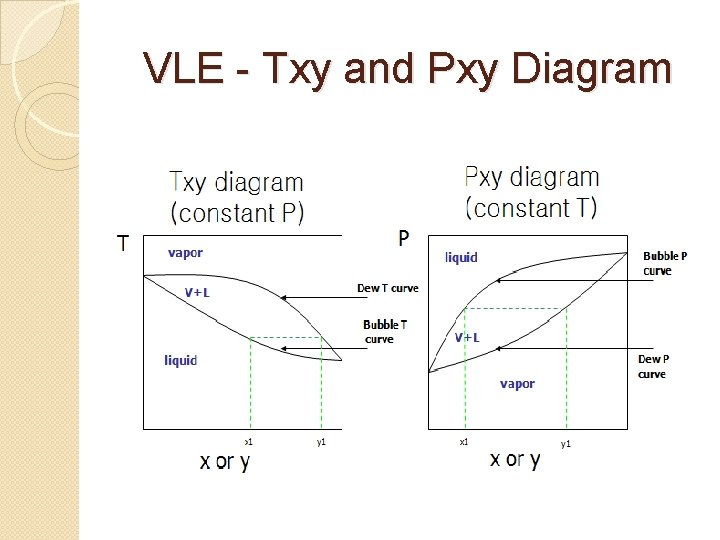
VLE - Txy and Pxy Diagram

VLE for Ideal Solution- Mixture of components

Bubble Point Pressure (Pbp)

Dew Point Pressure (Pdp)
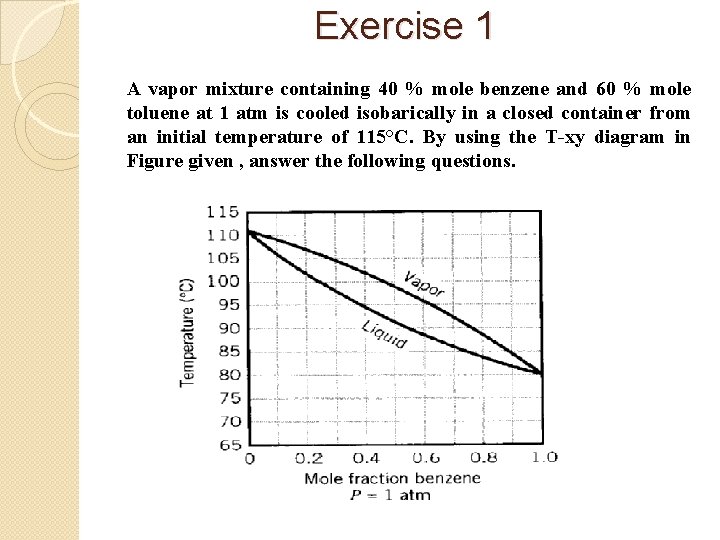
Exercise 1 A vapor mixture containing 40 % mole benzene and 60 % mole toluene at 1 atm is cooled isobarically in a closed container from an initial temperature of 115°C. By using the T-xy diagram in Figure given , answer the following questions.

(a) Estimate the dew point temperature (Tdp) and the equilibrium liquid composition of benzene (x. B) and toluene (x. T). (b) Estimate the mole fraction of benzene in the vapor (y. B) and liquid (x. B) if the mixture is steadily cooled to 90°C (a) If the mixture is in liquid phase, estimate the bubble point temperature (Tbp) and the equilibrium vapor composition of benzene (y. B) and toluene (y. T) (b) Estimate the final temperature if the liquid mixture is steadily vaporized until the remaining liquid contains 20% benzene.

Exercise 2 A vapor mixture consisting of 65 mole % benzene, 35 mole % toluene is compressed isothermally at 80°C until condensation occurs. (a) Estimate the pressure where the condensation begin? (b) Calculate the composition of the initial condensates

Chapter ended…
- Slides: 22