MULTI DRUG RESISTANT TUBERCULOSIS CHAIRPERSON DR MAYUR S

MULTI DRUG RESISTANT TUBERCULOSIS CHAIRPERSON : DR MAYUR S S PRESENTER : DR JYOTSNA R

INTRODUCTION �Tuberculosis is an ancient disease that has caused inestimable suffering and claimed millions of lives over the centuries �Discovery of effective anti TB drugs in 1940 – a medical milestone �Misuse of these “miracle” drugs has resulted in new public problem – Drug Resistant TB

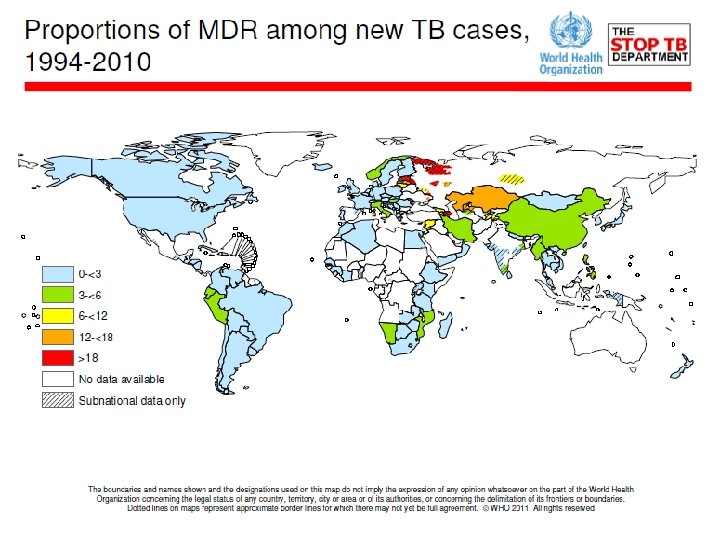
�Emergence of drug resistance tuberculosis and particularly MDR TB – public health problem �An obstacle to effective TB control program �First WHO endorsed DOTS Plus program – 2000 � 27 countries with high burden of MDR TB – responsible for 85% of global burden

�China and India account for 50% of estimated global burden of MDR TB �Population based survey –Gujarat and Maharashtra estimated Ø 3% among New cases Ø 12 -17% among Retreatment cases

Ø MDR TB cases estimated in 2010 q 21000 among new TB cases q 43000 among retreatment cases q Confirmed cases of MDR TB- 2967 Ø Magnitude of problem remains to be inadequately determined because of inadequate culture and DST facilities.





DRUG RESISTANCE �Definition: Ø Capacity of an organism and its progeny to remain viable or multiply in the presence of a concentration of a drug that would normally destroy them or inhibit their growth

CLASSIFICATION �TYPES : Ø Primary resistance Ø Secondary / Acquired resistance

�DEPENDING ON DRUG SENSITIVITY TESTS : Ø Mono resistance Ø Poly resistance Ø Multi drug resistance Ø Extensively drug resistance

FACTORS RELATED TO MDR TB �MDR-TB is a man-made phenomenon �poor treatment, poor drugs and poor adherence lead to the development of MDR-TB. �Most powerful predictor of presence of MDRTB is a history of previous treatment �In management most common error is adding single drug to failing regimen

CAUSES Ø Microbial Ø Clinical Ø Programmatic

Contd…. Ø PROGRAMS OR PROVIDERS : Inadequate regimens �Absence or inappropriate guidelines �Non compliance with guidelines �Inadequate training of health staff �Non monitoring of treatment �Poorly organized or funded TB control programs

Ø DRUGS : Inadequate supply or quality �Non availability of drugs �Poor quality �Poor storage conditions �Wrong dosages or combination

Ø PATIENTS : Inadequate drug intake �Poor adherence �Lack of information �Non availability of free drugs �Adverse drug reactions �Social and economic barriers �Malabsorption �Substance abuse disorders
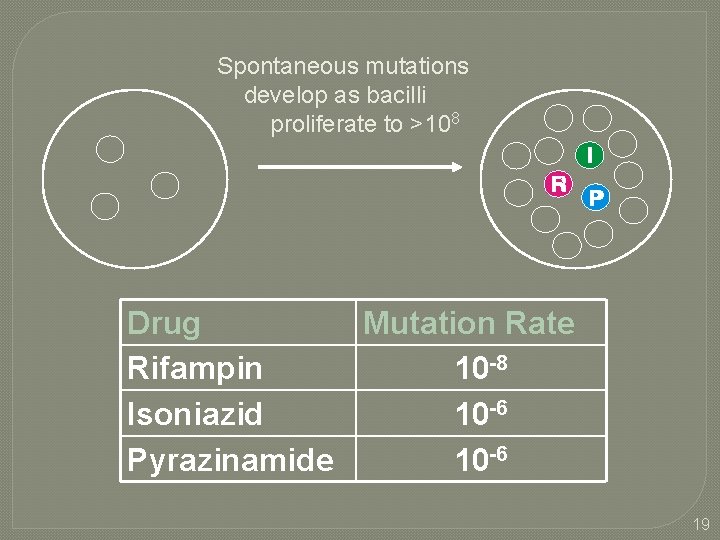
Spontaneous mutations develop as bacilli proliferate to >108 Drug Mutation Rate Rifampin 10 -8 Isoniazid 10 -6 Pyrazinamide 10 -6 19
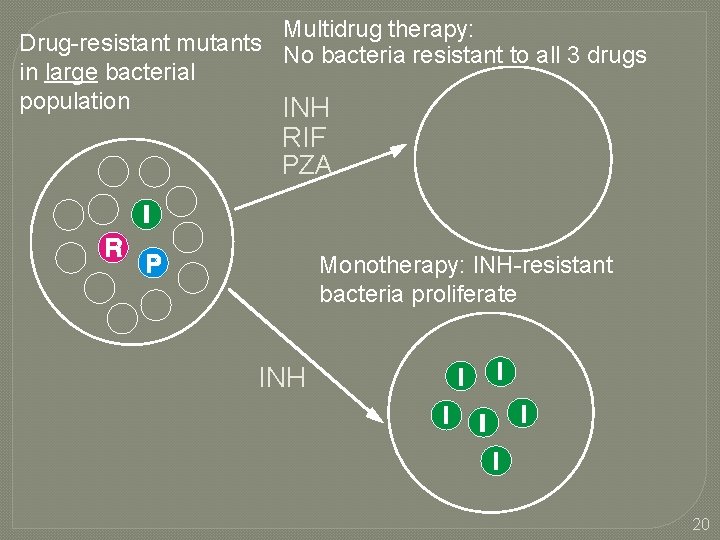
Multidrug therapy: Drug-resistant mutants No bacteria resistant to all 3 drugs in large bacterial population INH RIF PZA Monotherapy: INH-resistant bacteria proliferate INH 20
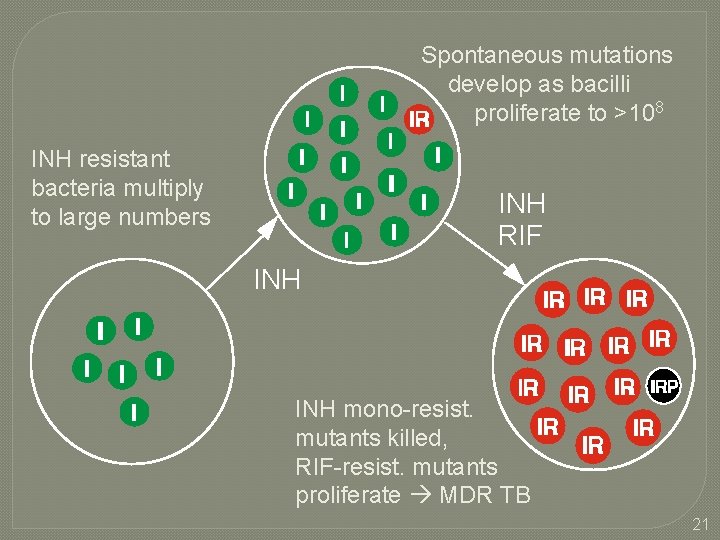
Spontaneous mutations develop as bacilli proliferate to >108 INH resistant bacteria multiply to large numbers INH RIF INH mono-resist. mutants killed, RIF-resist. mutants proliferate MDR TB 21
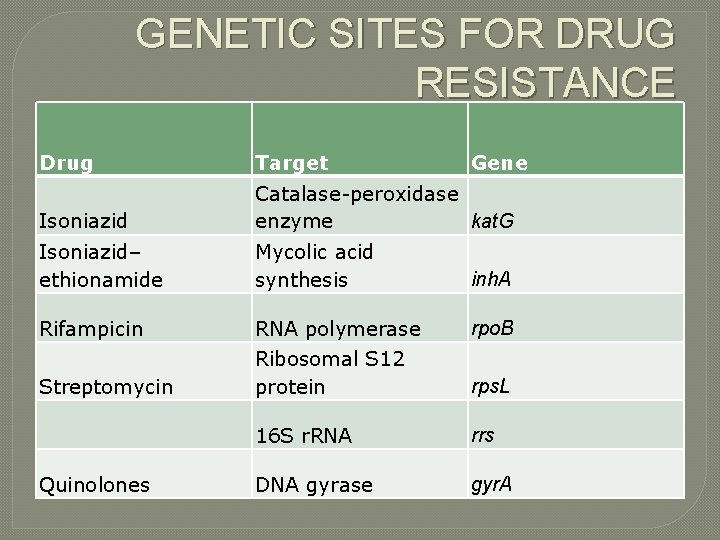
GENETIC SITES FOR DRUG RESISTANCE Drug Target Isoniazid Catalase-peroxidase kat. G enzyme Isoniazid– ethionamide Mycolic acid synthesis inh. A Rifampicin RNA polymerase rpo. B Streptomycin Ribosomal S 12 protein rps. L 16 S r. RNA rrs DNA gyrase gyr. A Quinolones Gene

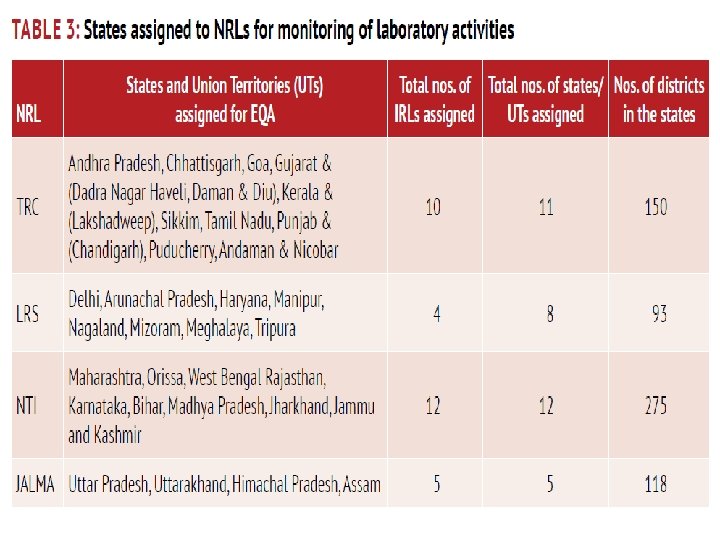
ORGANIZATION OF LABORATORY NETWORK RNTCP has 3 tier laboratory network system 1. Designated Microscopy Centres (DMC) 2. Intermediate Reference Laboratory (IRL) 3. National Reference Laboratory (NRL) �

IRL � 27 IRL in India �Training of personnel under DOTS Plus sites �External Quality Assessment (EQA) of sputum smear microscopy network in the districts and DMCs �Culture and Drug Susceptibility Testing (DST) for first line drugs for M tuberculosis
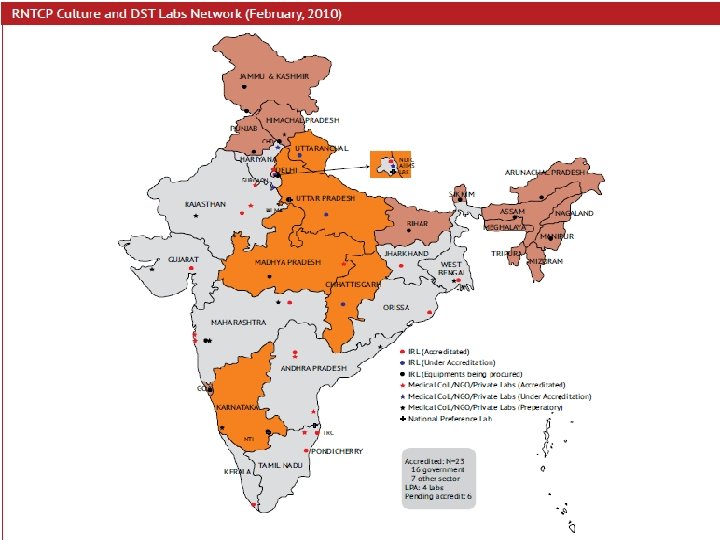

NRL � 4 NRL in India �Trainings of IRL staff �EQA of sputum smear microscopy network in the states alloted �Culture and DST for both first and second line drugs

SUPRANATIONAL REFERENCE LABORATORY NETWORK �Comprises 28 laboratories world wide �Acts as a global mechanism to ensure the quality of laboratory data through a system of proficiency tests


MDR TB SUSPECT �MDR TB Suspect : ü Any patient who fails Cat I treatment regimen ü Any Cat II patient who remains smear positive at the 4 th month of treatment or later ü Contacts of MDR TB cases

�DIAGNOSIS ü PURELY ü QUALITY OF MDR TB A LABORATORY DIAGNOSIS ASSURED ACCREDITED LABORATORIES MANDATORY FOR DIAGNOSIS

CASE FINDING STRATEGY CAT- I FAILURE RE-REGISTER AS FAILURE START ON CAT II REGIMEN
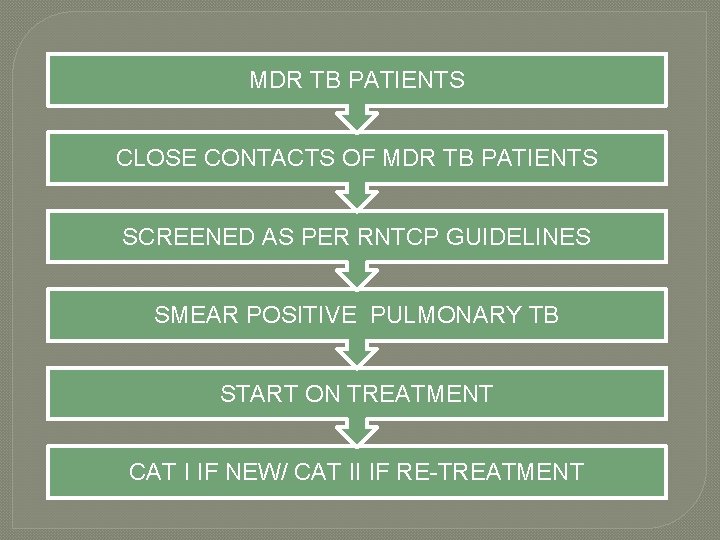
MDR TB PATIENTS CLOSE CONTACTS OF MDR TB PATIENTS SCREENED AS PER RNTCP GUIDELINES SMEAR POSITIVE PULMONARY TB START ON TREATMENT CAT I IF NEW/ CAT II IF RE-TREATMENT
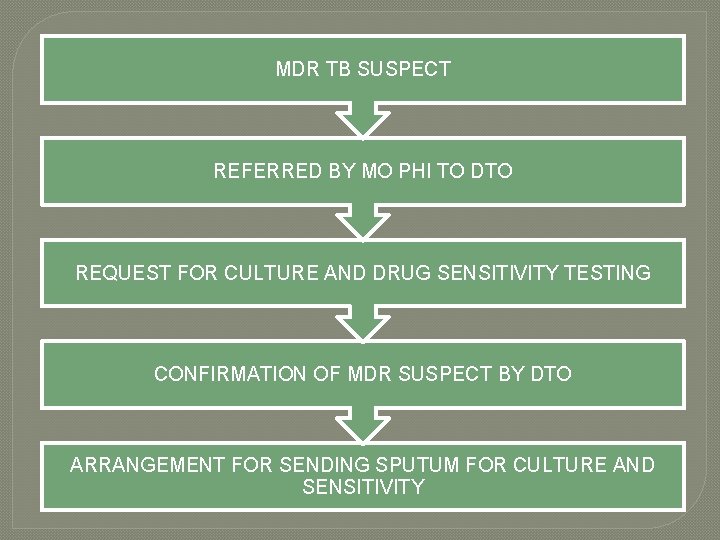
MDR TB SUSPECT REFERRED BY MO PHI TO DTO REQUEST FOR CULTURE AND DRUG SENSITIVITY TESTING CONFIRMATION OF MDR SUSPECT BY DTO ARRANGEMENT FOR SENDING SPUTUM FOR CULTURE AND SENSITIVITY

DIAGNOSIS OF MDR TB 1. 2. 3. Ø Ø Ø Sputum Smear microscopy Culture Drug Susceptibility Testing Absolute Concentration method Resistance Ratio method Proportionate method – method recommended for DOTS Plus sites in India

� Two SPUTUM COLLECTION FOR CULTURE morning samples : early morning and spot � Collected at DMC and sent or collected at DTC � From DTC samples transported to the laboratory for culture and DST � If delay in transportation > 72 hrs, samples transported in 1% Cetyl Pyridinium Chloride (CPC) & 2% Na. Cl � MDR TB suspect continues to take treatment till results are available

�Samples sent to accredited laboratory for culture and DST �Decontamination and homogenisation of samples done �Specimens inoculated and incubated �Cultures examined after 48 -72 hrs for gross contamination �Cultures examined weekly, upto 8 wks

�Typical colonies of M tuberculosis are rough, crumbly, waxy, buff coloured �Colonies with doubtful morphology are confirmed with ZN staining �The positive cultures are subjected for drug sensitivity testing by proportionate method

PROPORTIONATE METHOD Ø Ratio of the number of colonies growing on drug containing media to the number of colonies growing on drug free media – proportion of drug resistant bacilli present in the bacterial population
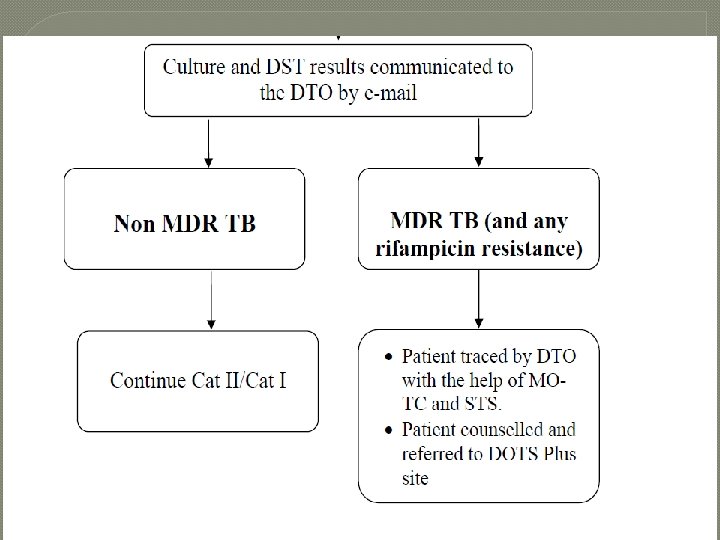

� CONFIRMED MDR TB CASE : Ø A MDR TB suspect Ø Sputum culture positive Ø TB is due to Mycobacterium tuberculosis Ø Resistant to Isoniazid and Rifampicin, Ø With or without resistance to other anti TB drugs

References �Revised national Tuberculosis Programme DOTS-Plus guidelines – 2010. �Guidelines for surveillance of drug resistance in tuberculosis, 4 th ed – World Health Organisation (WHO). �Management of MDR-TB – A field guide – WHO 2009.

CONTD… �Text book of Tuberculosis, A Mohan and SK Sharma 2004 �Park’s Textbook of Preventive and Social Medicine, 21 st ed - 2011


THANK YOU
- Slides: 45