Muharram 1435 November 2013 KSUCOMCourse 341 Water Electrolytes

Muharram 1435 -November 2013 KSU-COM-Course 341 Water & Electrolytes Disorders (H 2 O/Na+/K+) Ahmad Raed Tarakji, MD, MSPH, FRCPC, FACP, FASN, FNKF Assistant Professor Nephrology Unit, Department of Medicine College of Medicine, King Saud University Consultant Internist & Nephrologist King Khalid University Hospital Atarakji@ksu. edu. sa

Objectives 1) Composition of the fluid compartments 2) Mechanisms which regulate fluid and Sodium balance 3) Disorders of water balance 4) Disorders of Sodium balance 5) Disorders of Potassium balance Nov 2013 A R Tarakji, MD 2

Homeostasis A relative constancy in the internal environment of the body, naturally maintained by adaptive responses that promote cell function and survival Nov 2013 A R Tarakji, MD 3
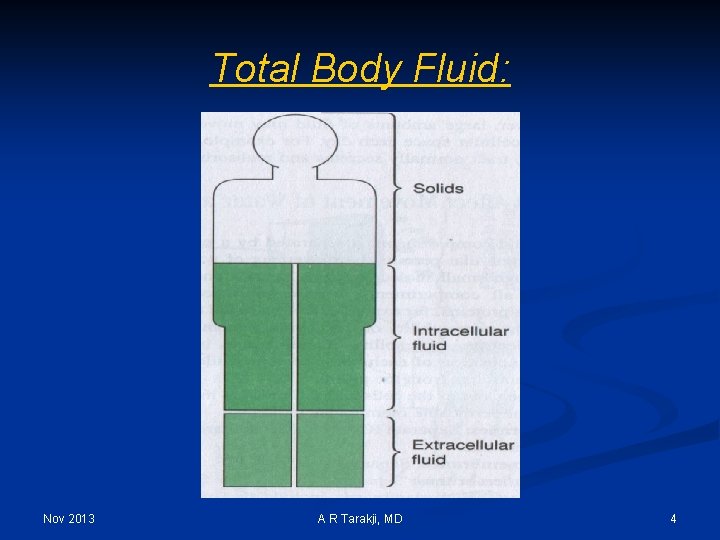
Total Body Fluid: Nov 2013 A R Tarakji, MD 4
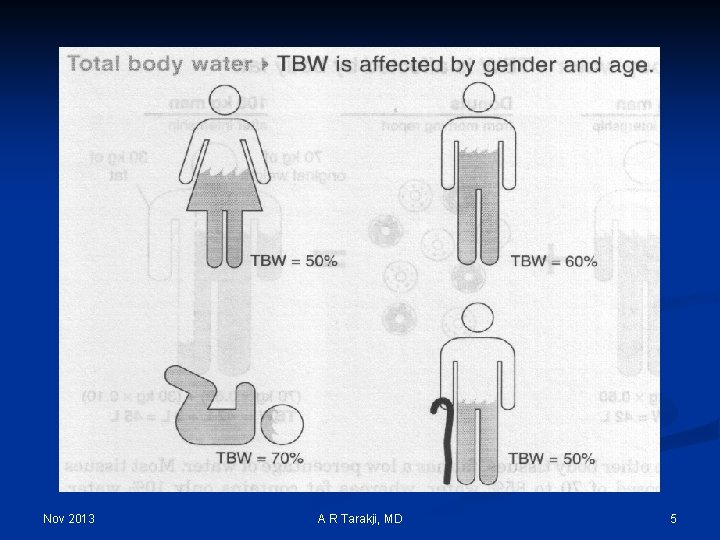
Nov 2013 A R Tarakji, MD 5

Body Fluid Compartments Total Body Water TBW (0. 6 x Body Weight) Nov 2013 Intracellular Fluid ICF Extracellular Fluid ECF (2/3 x TBW) (0. 4 x Body weight) (1/3 x TBW) (0. 2 x Body weight) Interstitial Fluid ISF Plasma IV (3/4 x ECF) (0. 75 x ECF) (1/4 x ECF) (0. 25 x ECF) A R Tarakji, MD 6
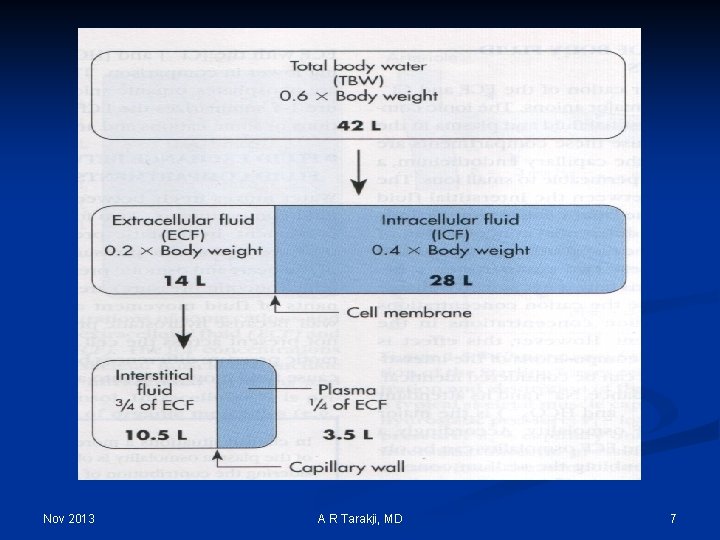
Nov 2013 A R Tarakji, MD 7

Body Fluid Compartments n Fluid compartments are separated by thin semipermeable membranes with pores to allow fluid movement and molecules of a specific size to pass while preventing larger heavier molecules from passing n The bodies fluid is composed of water and dissolved substances known as solutes (electrolytes or nonelectrolytes) n Electrolytes are substances that dissolved in solutions and dissociated into particles called ions n Cations: Positively charged ions n Anions: Negatively charged ions Nov 2013 A R Tarakji, MD 8

Definitions: q Osmosis: movement of water q Diffusion: movement of solutes q Filtration: movement of both solutes and water q Osmolality: q Osmoles in solution: m. Osm/kg water q Calc Posm = (2 x serum Na+) + blood urea + glucose q For Na+, K+ and Cl-: 1 m. Eq = 1 m. Osm q Normal osmolality of body fluids: 283 -292 m. Osm/kg water Nov 2013 A R Tarakji, MD 9
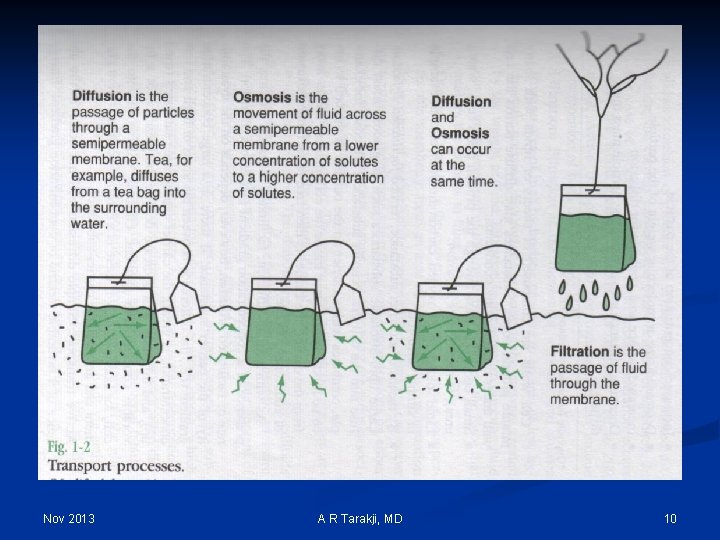
Nov 2013 A R Tarakji, MD 10

Nov 2013 A R Tarakji, MD 11

Body Fluid Compartments n n ECF and ICF are in osmotic equilibrium ICFosm = ECFosm = Posm IV ICF ISF K+ 140 Na+ 140 Nov 2013 A R Tarakji, MD 12
Nov 2013 A R Tarakji, MD 13

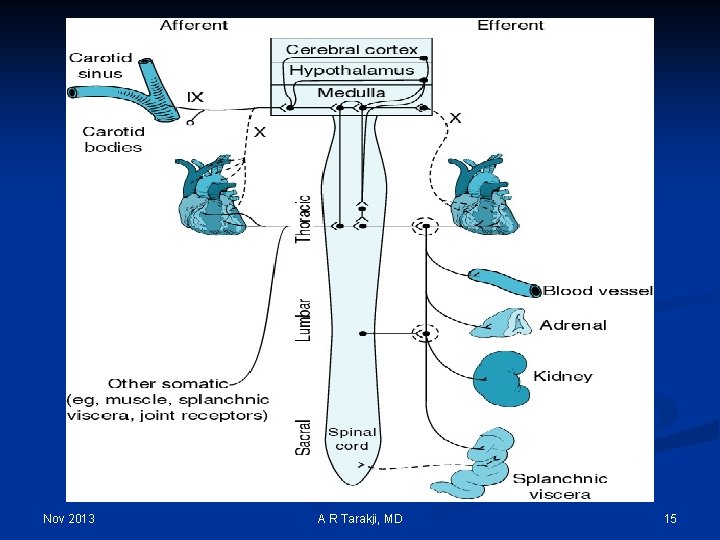
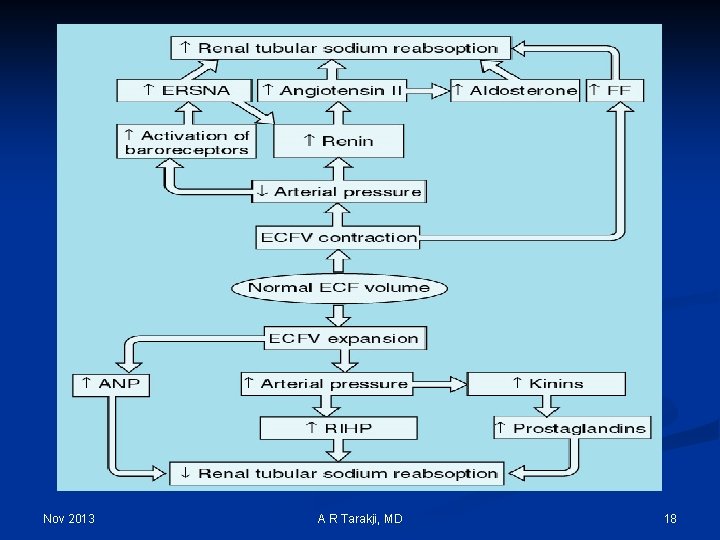
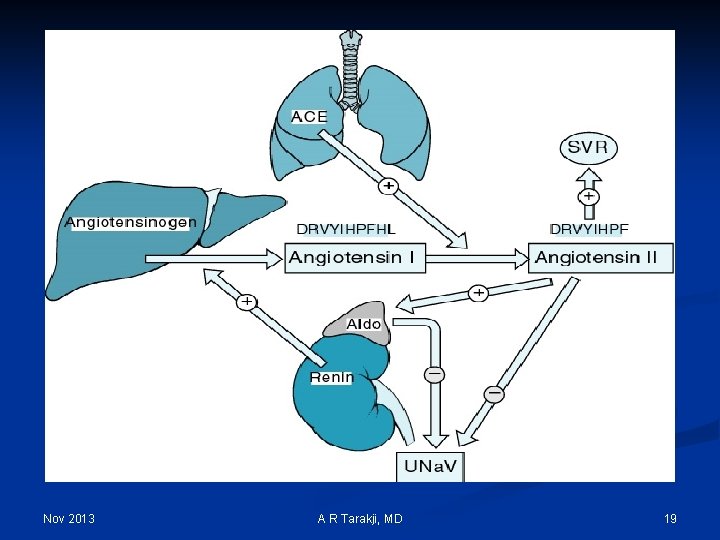
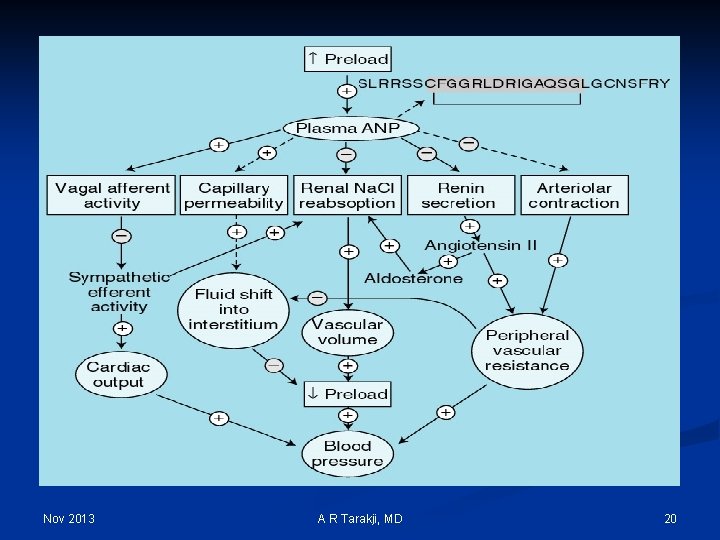
Regulation Mechanisms of Fluid and Electrolytes: n Regulation of osmolality and volume is achieved through thirst and the osmoreceptor-antidiuretic hormone system (vasopressin) n The regulation of volume also occurs through neurological and renal mechanisms n The stretch receptors (baroreceptors) n The Renin-Angiotension-Aldosterone System Nov 2013 A R Tarakji, MD 14
Nov 2013 A R Tarakji, MD 15
Nov 2013 A R Tarakji, MD 16
Nov 2013 A R Tarakji, MD 17
Nov 2013 A R Tarakji, MD 18
Nov 2013 A R Tarakji, MD 19
Nov 2013 A R Tarakji, MD 20
Nov 2013 A R Tarakji, MD 21
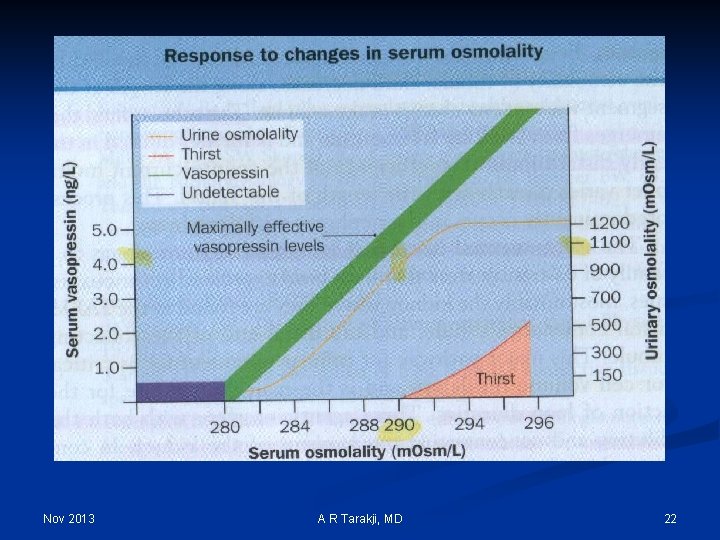
Nov 2013 A R Tarakji, MD 22

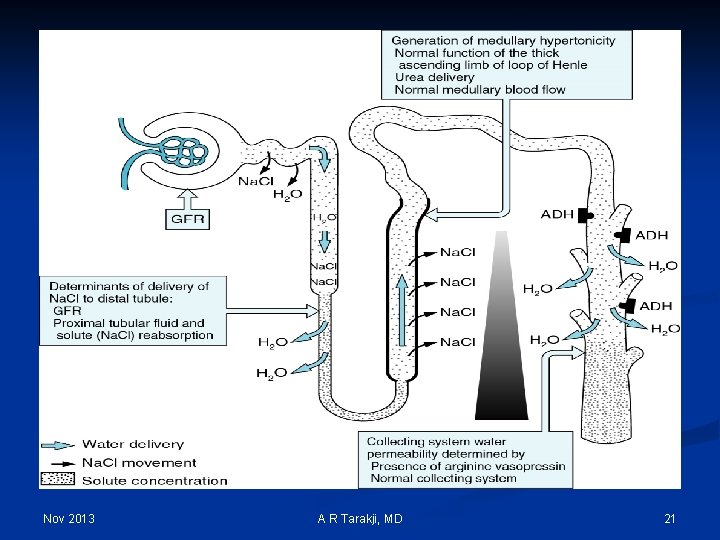
Urine Output & Daily Solute Load Nov 2013 A R Tarakji, MD 23
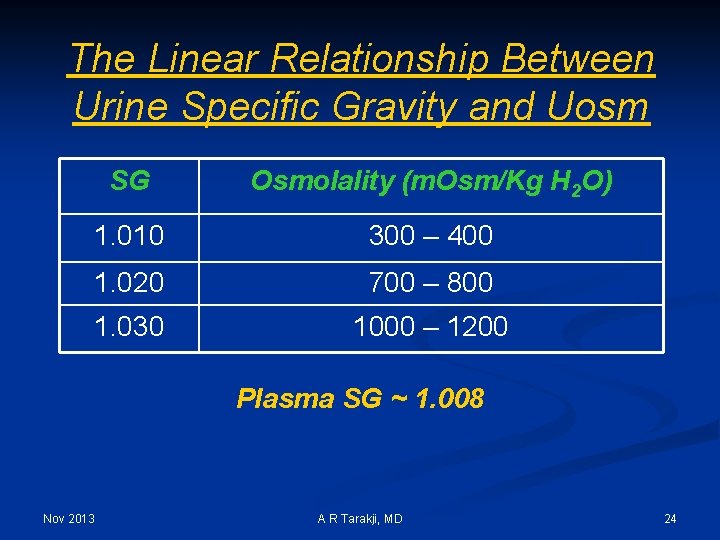
The Linear Relationship Between Urine Specific Gravity and Uosm SG Osmolality (m. Osm/Kg H 2 O) 1. 010 300 – 400 1. 020 700 – 800 1. 030 1000 – 1200 Plasma SG ~ 1. 008 Nov 2013 A R Tarakji, MD 24
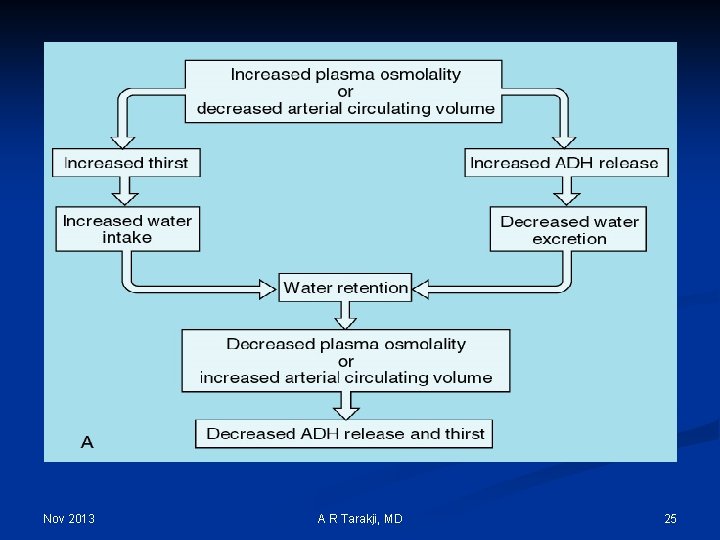
Nov 2013 A R Tarakji, MD 25
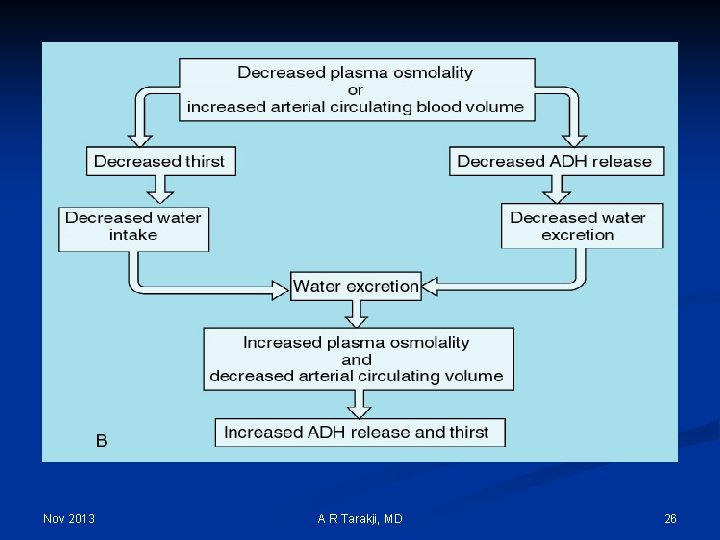
Nov 2013 A R Tarakji, MD 26
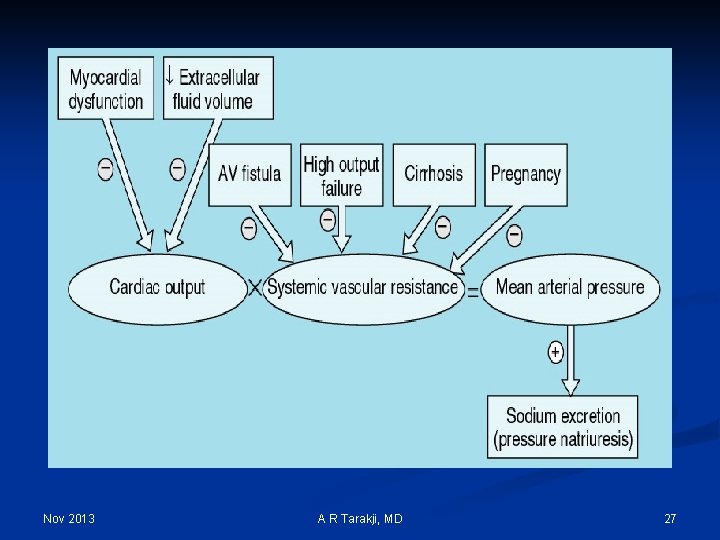
Nov 2013 A R Tarakji, MD 27
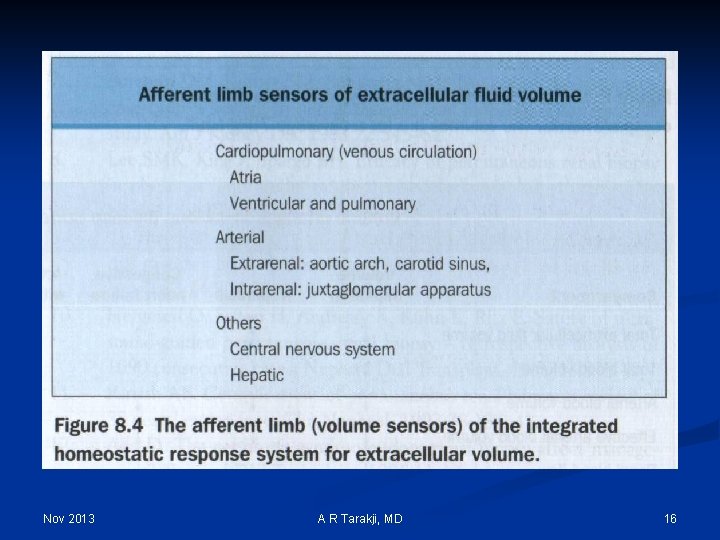
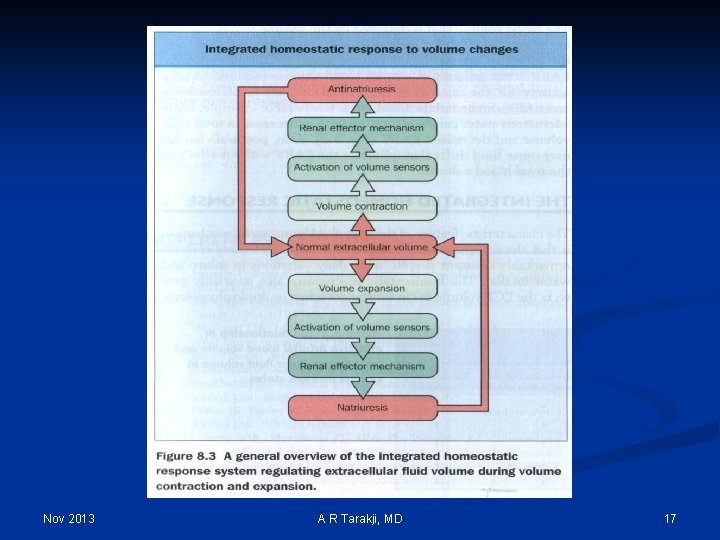
Effective Arterial Blood Volume (EABV): n Although the absolute volume of the intravascular space is an important component of circulatory “fullness”, the adequacy of the circulation (more commonly called the effective arterial blood volume or EABV) also is determined by cardiac output and systemic vascular resistance Nov 2013 A R Tarakji, MD 28
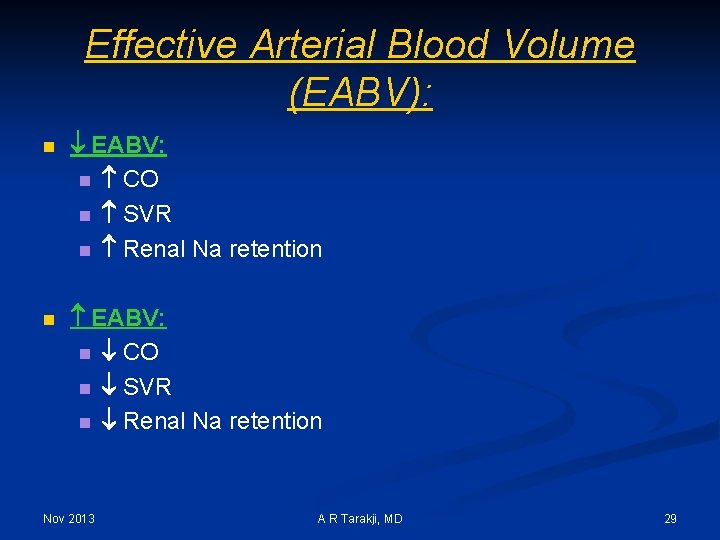
Effective Arterial Blood Volume (EABV): n EABV: n CO n SVR n Renal Na retention Nov 2013 A R Tarakji, MD 29
Effective Arterial Blood Volume (EABV): n EABV is the amount of arterial blood volume required to adequately ‘fill’ the capacity of the arterial circulation n ECF volume and EABV can be independent of each other n Edematous states: increase in total ECF volume and decreased EABV n Postural changes may cause shifts that influence the EABV without affecting the total blood volume Nov 2013 A R Tarakji, MD 30

Effective Arterial Blood Volume (EABV): Nov 2013 A R Tarakji, MD 31

Clinical features of Hypovolaemia & Hypervolaemia Hypovolemia Symptoms Thirst Dizziness on standing Weakness Signs Low JVP Postural hypotension Tachycardia Dry mouth Reduced skin turgor Reduced urine output Weight loss Confusion, stupor Nov 2013 A R Tarakji, MD Hypervolemia Ankle swelling Abdominal swelling Breathlessness Raised JVP Peripheral oedema Pulmonary crepitations Pleural effusion Ascites Hypertension (sometimes) Weight gain 32
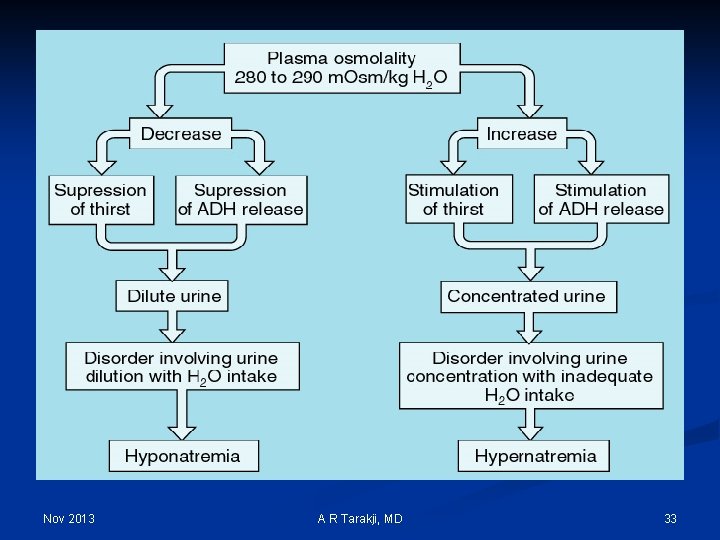
Nov 2013 A R Tarakji, MD 33

Sodium and Water: n n n ECF volume= absolute amounts of Sodium and water Plasma Na+ = ratio between the amounts of Sodium and water (Concentration) Hyponatremia = Water Excess Hypernatremia = Water Deficit Hypervolemia (Edema) = Sodium Excess Hypovolemia (Dehydration) = Sodium Deficit Nov 2013 A R Tarakji, MD 34

Sodium and Water: Hyponatremia (Water Excess) Hypovolemia (Dehydration) (Sodium Deficit) Hemorrhagic Shock Diarrhea in Children with good oral water and Seniors intake Hypervolemia Advanced (Edema) Congestive Heart (Sodium Excess) Failure Nov 2013 Hypernatremia (Water Deficit) A R Tarakji, MD Hemodialysis Patient after 3% Saline infusion 35

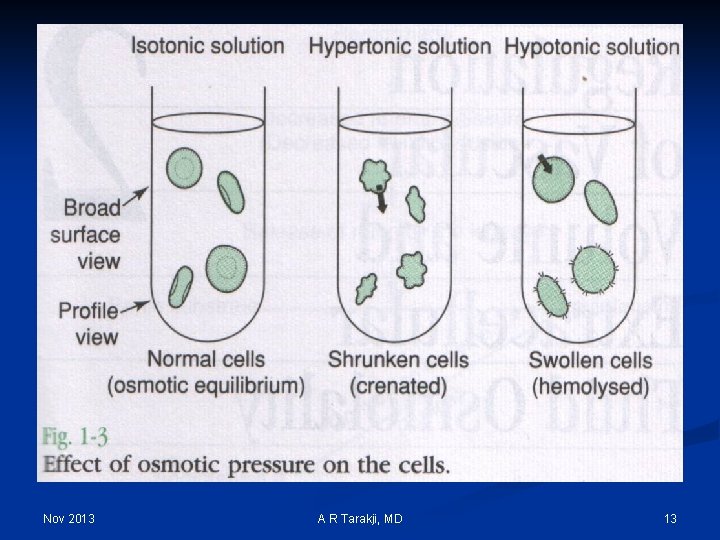
Tonicity Ø To compare the osmolality of a solution to that of another solution (body fluid compartments) Ø Used to compare the osmolality of intravenous solutions to that of the serum: Ø ISOTONIC Ø HYPERTONIC Nov 2013 A R Tarakji, MD 36
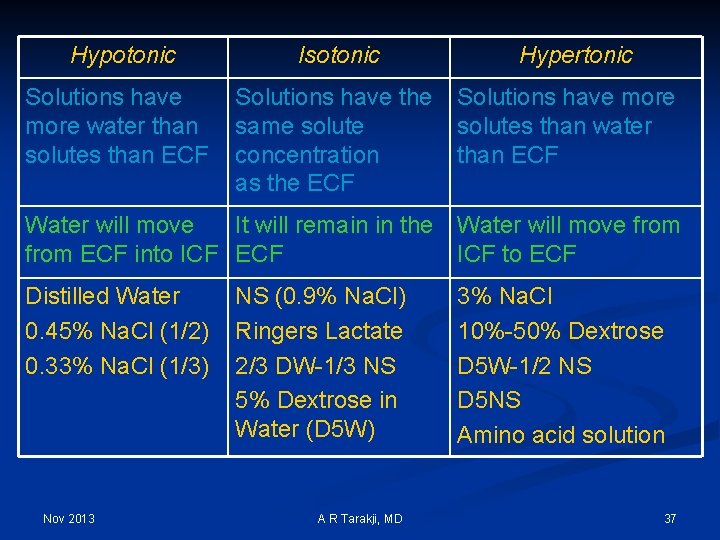
Hypotonic Isotonic Solutions have more water than solutes than ECF Solutions have the same solute concentration as the ECF Hypertonic Solutions have more solutes than water than ECF Water will move It will remain in the Water will move from ECF into ICF ECF ICF to ECF Distilled Water 0. 45% Na. Cl (1/2) 0. 33% Na. Cl (1/3) Nov 2013 NS (0. 9% Na. Cl) Ringers Lactate 2/3 DW-1/3 NS 5% Dextrose in Water (D 5 W) A R Tarakji, MD 3% Na. Cl 10%-50% Dextrose D 5 W-1/2 NS D 5 NS Amino acid solution 37

Intravenous Solutions n Crystalloids vs Colloids n Crystalloids are intravenous solutions that contain solutes that readily cross the capillary membrane n Dextrose and electrolyte solutions n Colloids are intravenous solutions that DO NOT readily cross the capillary membrane n Blood, albumin, plasma Nov 2013 A R Tarakji, MD 38
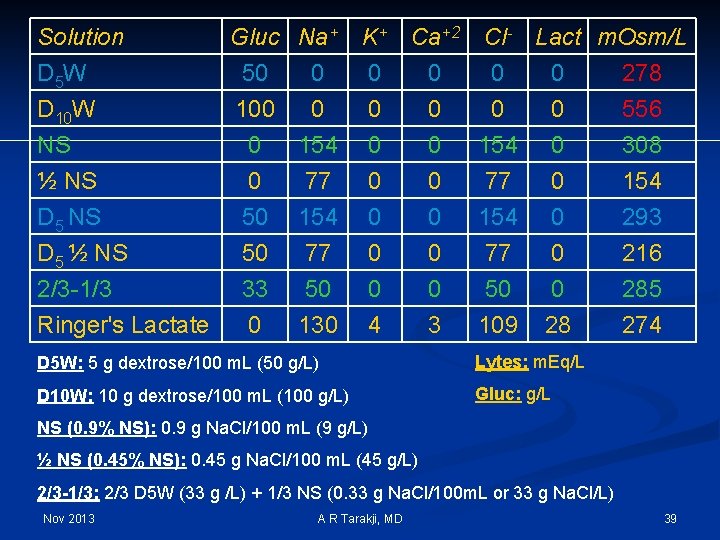
Solution D 5 W D 10 W NS ½ NS D 5 ½ NS 2/3 -1/3 Ringer's Lactate Gluc Na+ 50 0 100 0 0 154 0 50 50 33 0 K+ Ca+2 Cl- Lact m. Osm/L 0 0 278 0 0 556 0 0 154 0 308 77 154 77 50 130 0 0 4 0 0 3 77 154 77 50 109 0 0 28 D 5 W: 5 g dextrose/100 m. L (50 g/L) Lytes: m. Eq/L D 10 W: 10 g dextrose/100 m. L (100 g/L) Gluc: g/L 154 293 216 285 274 NS (0. 9% NS): 0. 9 g Na. Cl/100 m. L (9 g/L) ½ NS (0. 45% NS): 0. 45 g Na. Cl/100 m. L (45 g/L) 2/3 -1/3: 2/3 D 5 W (33 g /L) + 1/3 NS (0. 33 g Na. Cl/100 m. L or 33 g Na. Cl/L) Nov 2013 A R Tarakji, MD 39
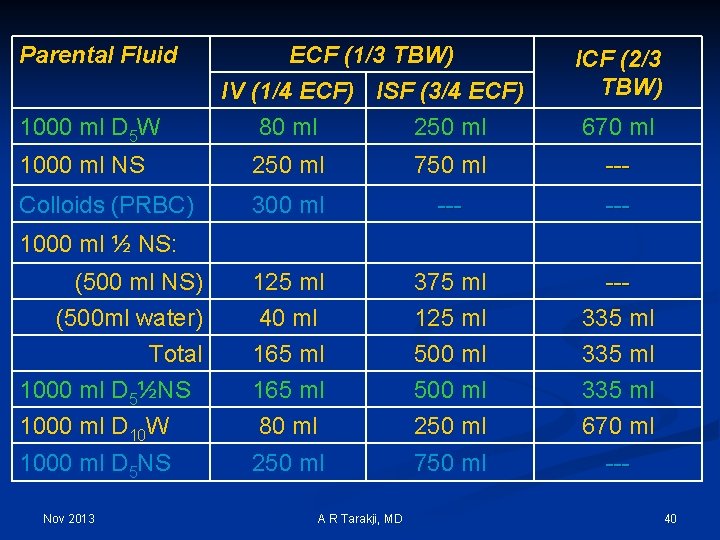
Parental Fluid 1000 ml D 5 W ECF (1/3 TBW) IV (1/4 ECF) ISF (3/4 ECF) 80 ml 250 ml ICF (2/3 TBW) 670 ml 1000 ml NS 250 ml 750 ml --- Colloids (PRBC) 300 ml --- (500 ml NS) (500 ml water) Total 1000 ml D 5½NS 1000 ml D 10 W 125 ml 40 ml 165 ml 80 ml 375 ml 125 ml 500 ml 250 ml --335 ml 670 ml 1000 ml D 5 NS 250 ml 750 ml --- 1000 ml ½ NS: Nov 2013 A R Tarakji, MD 40
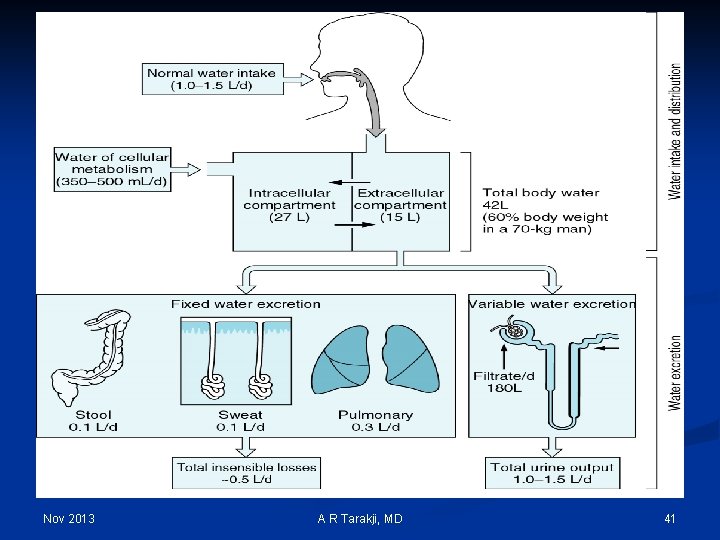
Nov 2013 A R Tarakji, MD 41

Hyponatremia Nov 2013 A R Tarakji, MD 42

Hyponatremia 1. Normotonic or Isotonic Hyponatremia 1. Factitious Hyponatremia 2. Pseudohyponatremia 3. Results from laboratory artifact due to high concentrations of proteins or lipids 2. Hypertonic Hyponatremia 1. Translocational Hyponatremia 2. Results from non-Na osmoles in serum (often glucose or mannitol) drawing Na-free H 2 O from cells 3. [Na+] declines by 1. 6 m. Eq/L for each 100 mg/d. L [5. 6 mmol/L] increase in serum glucose Nov 2013 A R Tarakji, MD 43
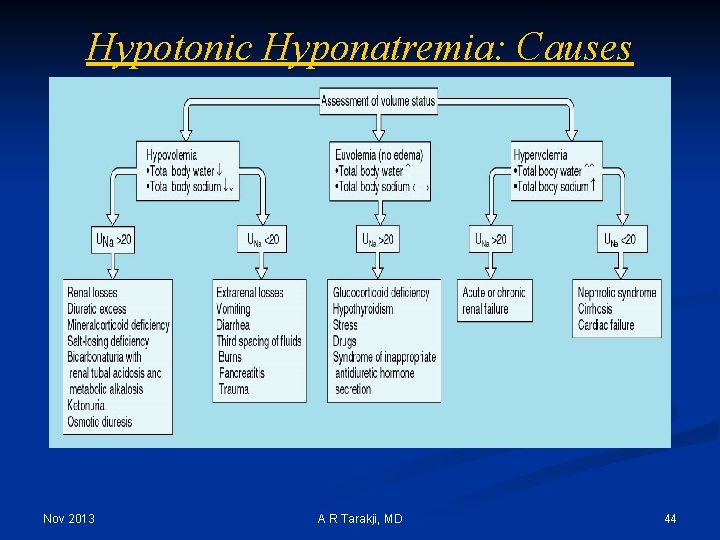
Hypotonic Hyponatremia: Causes Nov 2013 A R Tarakji, MD 44

SIADH • H: Hypoosmolar Hyponatremia (Posm <275 m. Osm/Kg H 2 O) • I: Inappropriate urine concentration (Uosm >100 m. Osm/Kg H 2 O) • V: Euvolemia, No diuretic use • E: Endocrine = normal Thyroid, adrenal and renal function • Hypouricemia (<238 mcmol/L) and low Urea (<3. 5 mmol/L) Nov 2013 A R Tarakji, MD 45
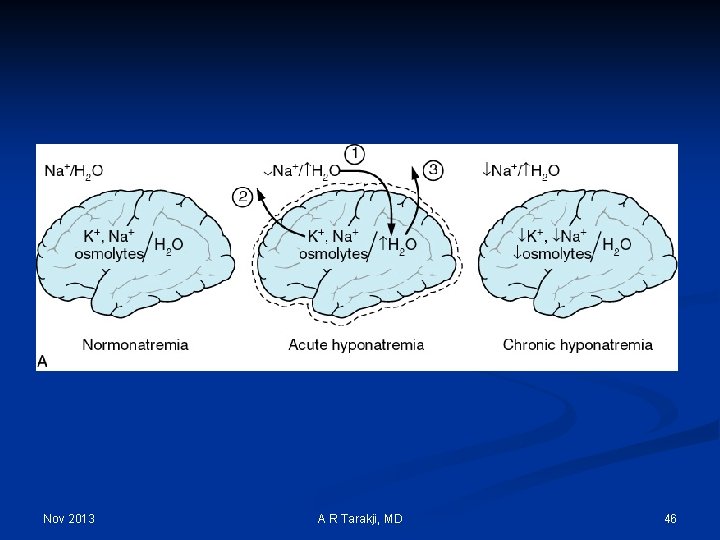
Nov 2013 A R Tarakji, MD 46
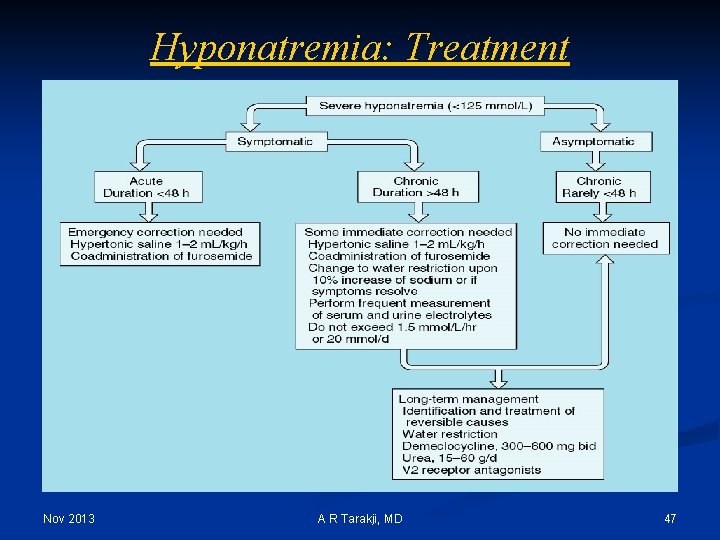
Hyponatremia: Treatment Nov 2013 A R Tarakji, MD 47

Hypernatremia Nov 2013 A R Tarakji, MD 48
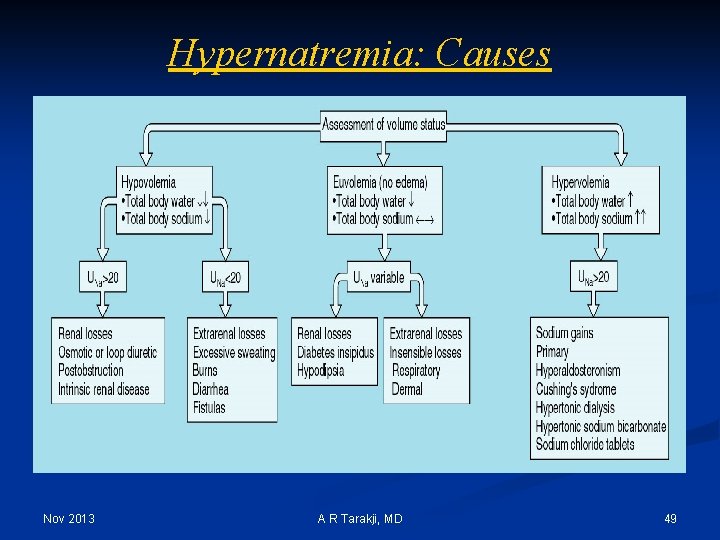
Hypernatremia: Causes Nov 2013 A R Tarakji, MD 49
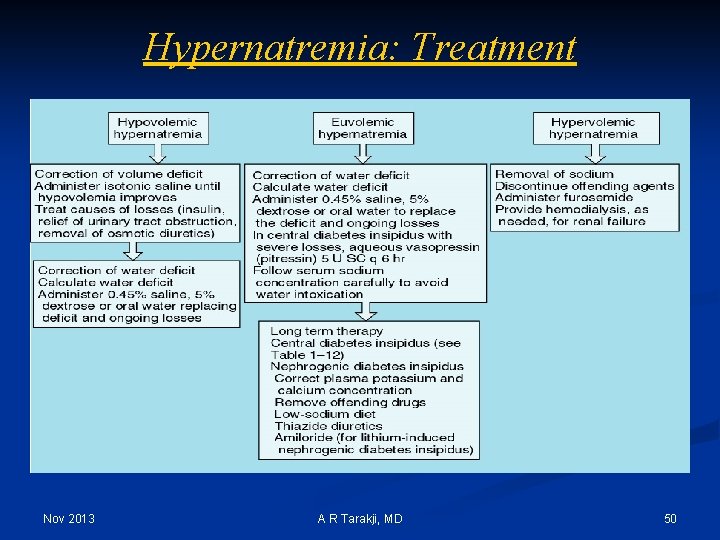
Hypernatremia: Treatment Nov 2013 A R Tarakji, MD 50

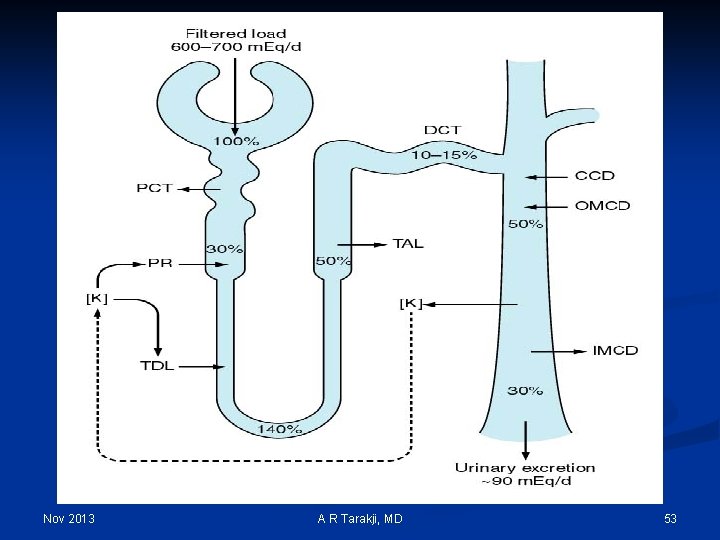
Potassium n n 98% of the total potassium is in ICF n Serum potassium is 3. 5 – 5. 5 mmol/L n Extracellular concentration is not an accurate reflection of the total body potassium Function: n Conduction of impulses n Acid base balance n Protein synthesis and carbohydrate metabolism Potassium secretion is under multiple controls: n Sodium load delivered to the kidneys n Acid base status n Potassium intake n Aldosterone levels Kidneys do not conserve potassium effectively Nov 2013 A R Tarakji, MD 51
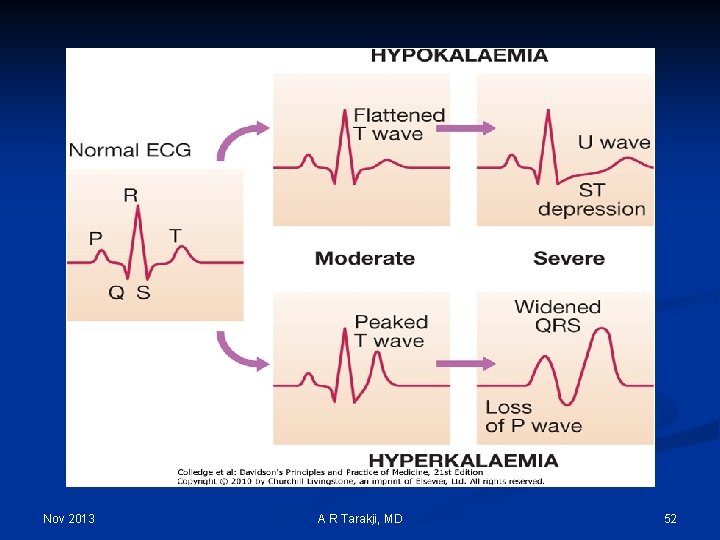
Nov 2013 A R Tarakji, MD 52
Nov 2013 A R Tarakji, MD 53

Hypokalemia Nov 2013 A R Tarakji, MD 54
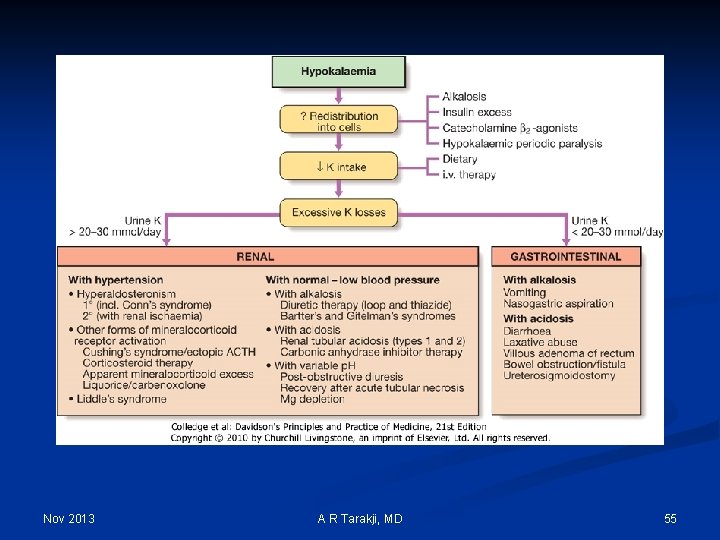
Nov 2013 A R Tarakji, MD 55

Hypokalemia: Clinical manifestations 1. Cardiovascular: Arrhythmias, Digitalis toxicity 2. Neuromuscular: 1. Smooth muscle: Ileus 2. Skeletal muscle: Weakness, Paralysis, Rhabdomyolysis 3. Endocrine: Glucose intolerance 4. Renal/electrolyte: 1. Vasopressin resistance 2. Increased ammonia production 3. Metabolic alkalosis 5. Structural changes: Renal cysts, Interstitial changes Nov 2013 A R Tarakji, MD 56
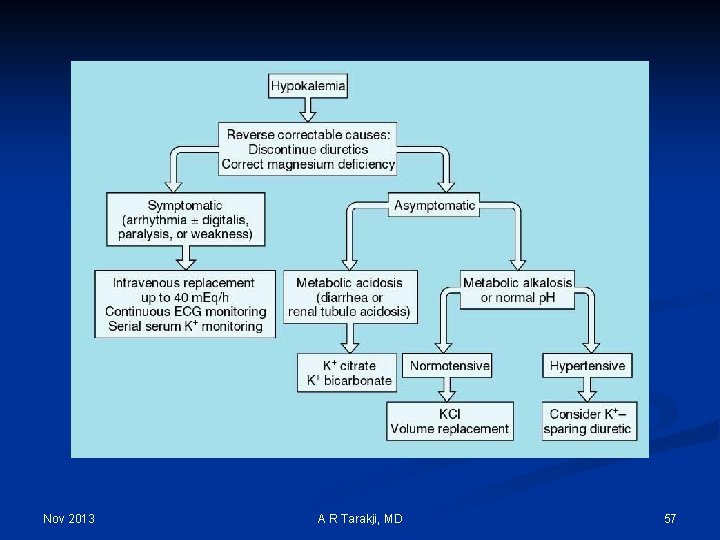
Nov 2013 A R Tarakji, MD 57
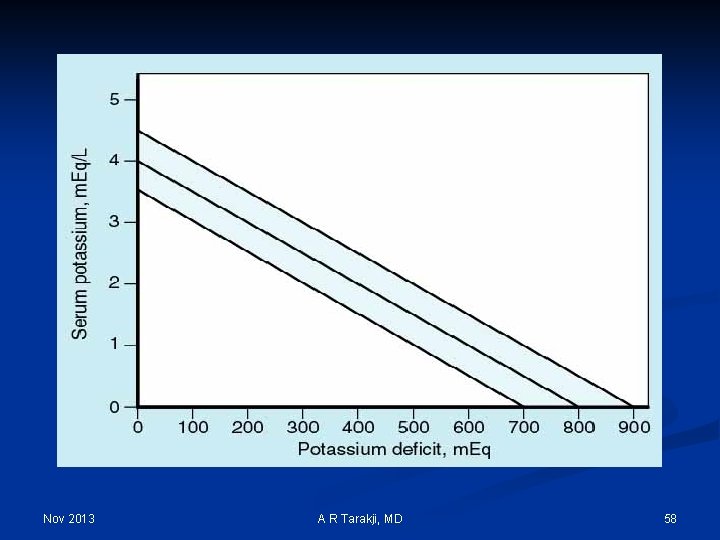
Nov 2013 A R Tarakji, MD 58

Hyperkalemia Nov 2013 A R Tarakji, MD 59
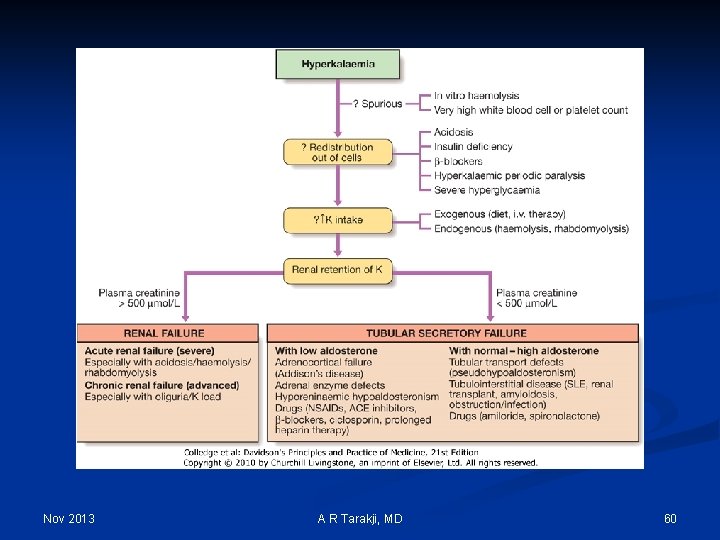
Nov 2013 A R Tarakji, MD 60

Hyperkalemia: Clinical manifestations 1. Cardiovascular: 1. T-wave abnormalities 2. Lengthened segments 3. Brady-arrhythmias 2. Neuromuscular: 1. Ileus 2. Paresthesias 3. Weakness 4. Paralysis 3. Renal/electrolyte: 1. Decreased ammonia production 2. Metabolic acidosis Nov 2013 A R Tarakji, MD 61
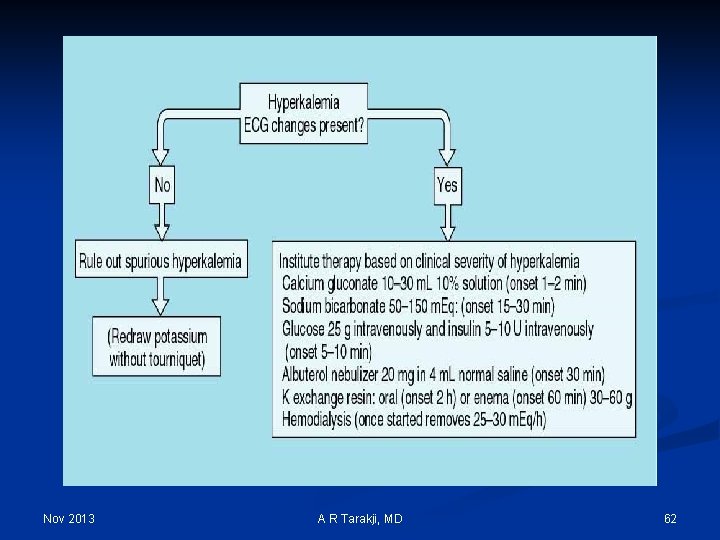
Nov 2013 A R Tarakji, MD 62

Questions? ? Nov 2013 A R Tarakji, MD 63
- Slides: 63