MTDATA ISSUES John Gisby Hugh Davies NPL Materials

MTDATA ISSUES John Gisby, Hugh Davies NPL Materials Centre, National Physical Laboratory March 2002

DATA ISSUES MODELS KCl-Ca. Cl 2 as an example DATA ASSESSMENT Ni. O-Al 2 O 3 as an example DATA FOR OXIDE SYSTEMS MIRO project RC 132
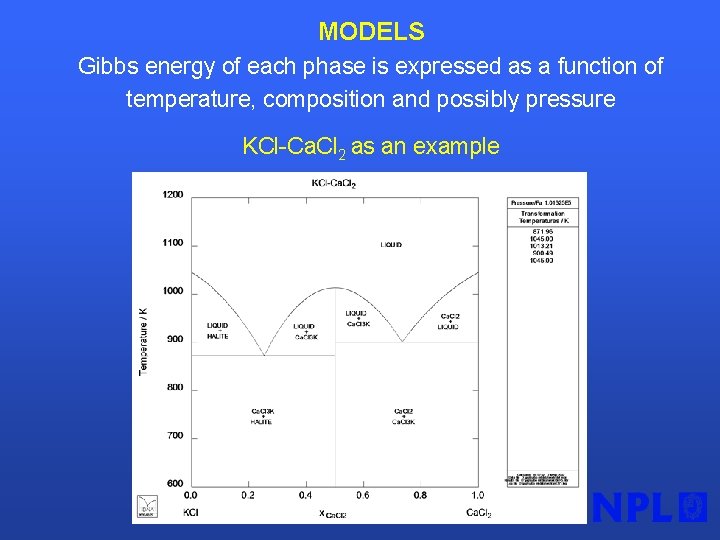
MODELS Gibbs energy of each phase is expressed as a function of temperature, composition and possibly pressure KCl-Ca. Cl 2 as an example

Stoichiometric substance or solution phase “end member” GCa. KCl 3 = A + BT + CTln. T + DT 2 + ET 3 + F/T Solution phase (Redlich-Kister model for Gex) GLIQUID = x. KCl GKCl + x. Ca. Cl 2 GCa. Cl 2 + RT x. KClln x. KCl + RT x. Ca. Cl 2 ln x. Ca. Cl 2 + Gex = (L 00+L 01 T) x. KClx. Ca. Cl 2 + (L 10+L 11 T) x. KClx. Ca. Cl 2 (x. KCl-x. Ca. Cl 2) + (L 20+L 21 T) x. KClx. Ca. Cl 2 (x. KCl-x. Ca. Cl 2)2 +…

GCa. KCl 3 = A + BT + CTln. T + DT 2 + ET 3 + F/T

Gex = (L 00+L 01 T) x. KClx. Ca. Cl 2 + (L 10+L 11 T) x. KClx. Ca. Cl 2 (x. KCl-x. Ca. Cl 2) + (L 20+L 21 T) x. KClx. Ca. Cl 2 (x. KCl-x. Ca. Cl 2)2 + …
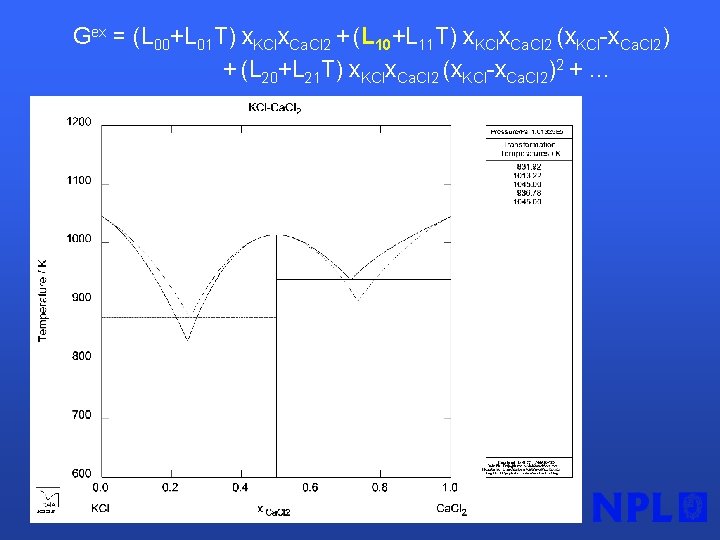
Gex = (L 00+L 01 T) x. KClx. Ca. Cl 2 + (L 10+L 11 T) x. KClx. Ca. Cl 2 (x. KCl-x. Ca. Cl 2) + (L 20+L 21 T) x. KClx. Ca. Cl 2 (x. KCl-x. Ca. Cl 2)2 + …
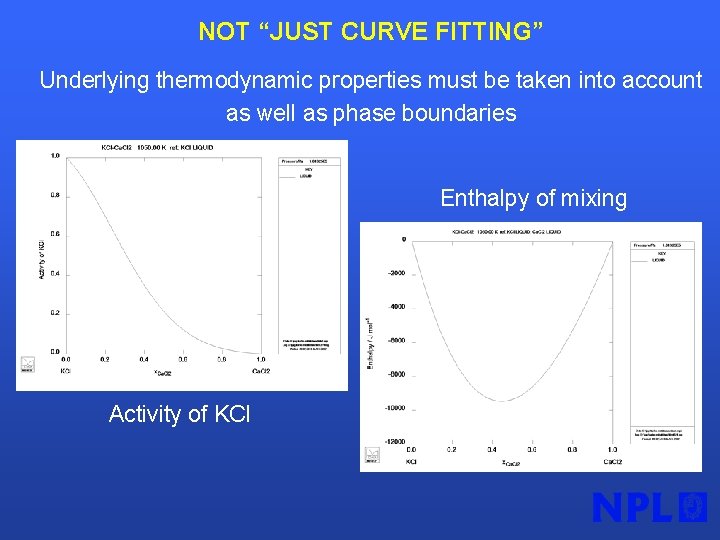
NOT “JUST CURVE FITTING” Underlying thermodynamic properties must be taken into account as well as phase boundaries Enthalpy of mixing Activity of KCl
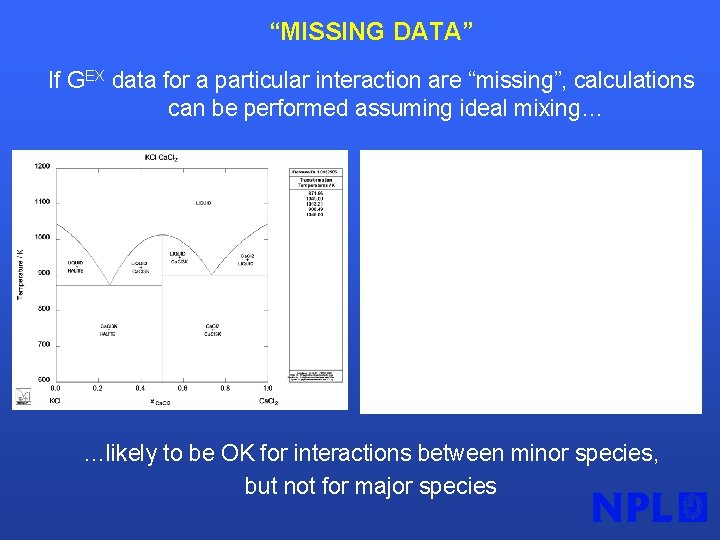
“MISSING DATA” If GEX data for a particular interaction are “missing”, calculations can be performed assuming ideal mixing… …likely to be OK for interactions between minor species, but not for major species
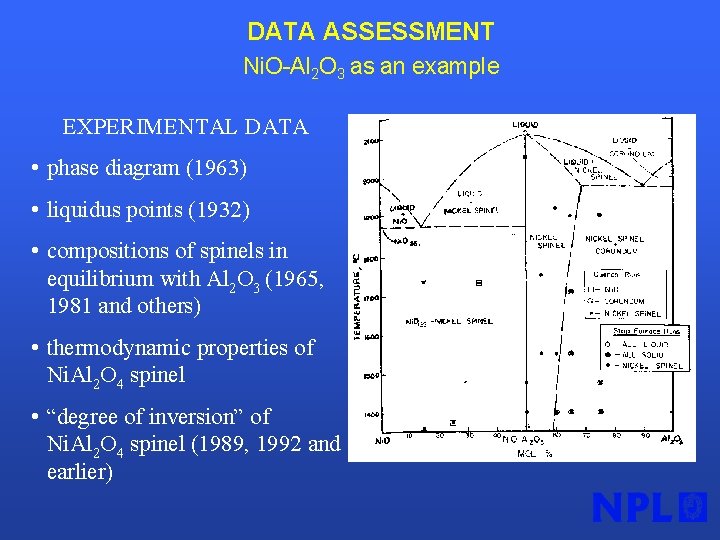
DATA ASSESSMENT Ni. O-Al 2 O 3 as an example EXPERIMENTAL DATA • phase diagram (1963) • liquidus points (1932) • compositions of spinels in equilibrium with Al 2 O 3 (1965, 1981 and others) • thermodynamic properties of Ni. Al 2 O 4 spinel • “degree of inversion” of Ni. Al 2 O 4 spinel (1989, 1992 and earlier)

SPINEL MODEL (Ni 2+, Al 3+)1 (Ni 2+, Al 3+, Va)2 (O 2 -)4 “normal” Ni. Al 2 O 4 - (Ni 2+) (Al 3+) (Va)2 (O 2 -)4 “inverse” Ni. Al 2 O 4 - (Al 3+) (Ni 2+ Al 3+) (Va 2) (O 2 -)4 “degree of inversion” is the fraction of sites on the first sublattice occupied by Al 3+

INITIAL PHASE DIAGRAM AND DEGREE OF INVERSION

CHANGING ENTHALPY / ENTROPY DATA FOR TWO SPINEL UNARIES TO MODEL DEGREE OF INVERSION, GIBBS ENERGY OF FORMATION AT 298. 15 K, AND MELTING POINT OF Ni. Al 2 O 4 SPINEL

NEW PHASE DIAGRAM AND DEGREE OF INVERSION PLOT

NOW INTRODUCE INTERACTION PARAMETERS IN SPINEL PHASE TO MODEL EQUILIBRIA WITH Al 2 O 3 (CORUNDUM)

NOW INTRODUCE INTERACTION PARAMETERS IN LIQUID PHASE TO MODEL LIQUIDUS
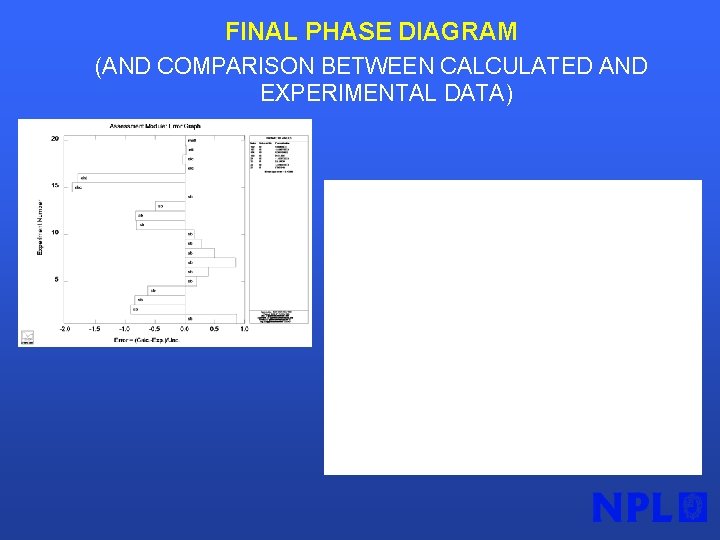
FINAL PHASE DIAGRAM (AND COMPARISON BETWEEN CALCULATED AND EXPERIMENTAL DATA)

MIRO (Mineral Industry Research Organisation) Projects RC 54, RC 81 and RC 132 To develop a high quality, comprehensive thermodynamic database for oxide and sulphur containing systems of industrial interest for use in predictive phase equilibrium calculations Facilitates calculations directly relevant to industrial processes for temperature and composition ranges outside the scope of hard copy phase diagram compilations § Systems and priorities determined by the sponsors § Database validated against plant / process data by sponsors

SPONSORS (current sponsors in white) Johnson Matthey Rio Tinto Corus Federal Mogul Amplats Sheffield University Britannia Zinc AEA Technology Outokumpu Oy Pilkington BNFL Metal. Europ Shell DTI

CURRENT ELEMENTS Al, B, Ba, Cr, Cu, Fe, K, La, Li, Mg, Mn, Na, Ni, O, Pb, S, Si, Sr, U, Zn, Zr CURRENT SYSTEMS Na 2 O-K 2 O-Ca. O-Fe-O-Mg. O-Al 2 O 3 -Si. O 2 -S Na 2 O-Ca. O-Fe-O-Mg. O-Al 2 O 3 -B 2 O 3 -Si. O 2 Li 2 O-Na 2 O-B 2 O 3 -Si. O 2 Ca. O-Cu-O-Fe-Si. O 2 (-Mg. O-Al 2 O 3) Ca. O-Fe-O-Mn. O-Mg. O-Al 2 O 3 -Si. O 2 Ca. O-Fe-O-Pb. O-Zn. O-Al 2 O 3 -Si. O 2 Ca. O-Mg. O-Al 2 O 3 -Si. O 2 -Zr. O 2 Ca. O-Cr-O-Fe-Mg. O-Al 2 O 3 -Si. O 2 Ca. O-Fe-O-Mg. O-Ni. O-Al 2 O 3 -Si. O 2 Cu-Fe-Ni-S-O (mattes / alloys) Some Ba. O, La 2 O 3, Sr. O, UO 2 containing binary systems

AREAS BEING CONSIDERED FOR FUTURE WORK CONTINUE TO EXTEND COVERAGE OF CURRENT OXIDES plus § addition of Co, Mn, Pb to matte / metal phase § addition of Co. O, Ti. O 2, V 2 O 3, rare earth oxides to oxide phases § molten salt - oxide systems (including addition of Ca. F 2 to oxide phases) § inclusion of P, As § inclusion of H 2 O (at high and low temperatures) § modelling volume changes in oxide phases

APPLICATIONS § § § Preventing iron formation in zinc blast furnace Analysing possible consequences of nuclear reactor accident Modelling inclusion formation in steels Analysing problems associated with glass making Optimising recovery of Cu, Ni, precious metals Investigating refractory wear Improving understanding of cement manufacturing process Modelling of corrosion Pollution control Process modelling (links with other commercial software) Analysing why borax additions to steelmaking slags reduce dust generation after solidification
- Slides: 22