Mrs Wilson Staple your lab rubric to the

Mrs. Wilson

§Staple your lab rubric to the back of your Foul Water Lab. Make sure you’ve written your name and your partners’ names. §Turn in your Foul Water Labs. §Please follow the LASA honor code.

§ (it’s very easy )

§ Calculate the number of representative particles (atoms, ions, molecules, or formula units) in a sample of material if mass is given OR if # of moles are given. § Calculate the # of moles or the mass of a sample of material if the number of representative particles is given. Homework: 1. 9 Homework + Daily Quiz 1 -10 Next Class

§The SI unit used to measure the amount of substance is the mole (Latin for “pile”) §The “mole” is a counting unit, used to make counting things much easier. 1 dozen of ANYTHING = 12 things 1 mole of ANYTHING = 6. 02 x 1023 things 6. 02 x 1023 = Avogadro’s Number (symbol NA) THIS IS EQUAL TO THE NUMBER OF CARBON-12 ATOMS IN 12 G OF CARBON-12. BUT… HOW DO WE COUNT ATOMS?

This exercise is to help you understand that for chemists to count atoms, they have to weigh them. Let’s say the mass of Cheerios in one box = 500. g (don’t worry about the box or the bag) A “gaggle” of Cheerios = 100 Cheerios. Mass of one “gaggle” = 15. 0 grams How many Cheerios are in the box? Use sig figs to express your final answer. (hint: # Cheerios is a counting unit, so don’t use 100 to help you determine sig figs. )

This exercise is to help you understand that for chemists to count atoms, they have to weigh them. Let’s say the mass of all Cheerios in one box = 500. g (don’t worry about the box or the bag) A “gaggle” of Cheerios = 100 Cheerios. Mass of one “gaggle” = 15. 0 grams How many Cheerios are in the box? 500. g / 15. 0 g = 33. 3 “gaggles” x 100 = 3. 33 x 103 Cheerios

Elements have ATOMS Covalent compounds have uncharged MOLECULES (groups of ATOMS) § Covalent compounds = consist of nonmetal atoms only (found on right side of periodic table; H is also a nonmetal) Ionic compounds have FORMULA UNITS (neutral groups of IONS) § Ions = atoms or molecules that have gained or lost electrons § Ionic compounds = consist of metal ions and nonmetal ions (metals found on LEFT and MIDDLE side of periodic table)
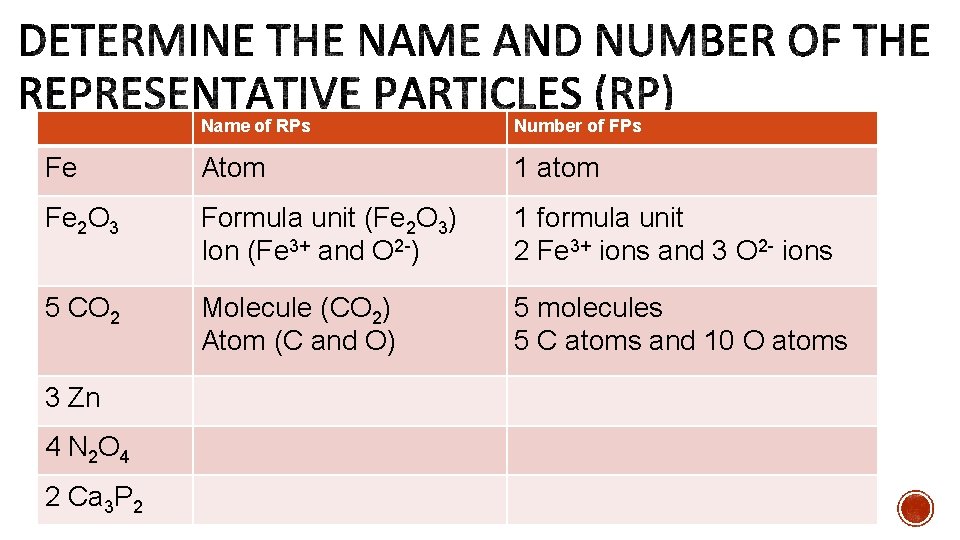
Name of RPs Number of FPs Fe Atom 1 atom Fe 2 O 3 Formula unit (Fe 2 O 3) Ion (Fe 3+ and O 2 ) 1 formula unit 2 Fe 3+ ions and 3 O 2 ions 5 CO 2 Molecule (CO 2) Atom (C and O) 5 molecules 5 C atoms and 10 O atoms 3 Zn 4 N 2 O 4 2 Ca 3 P 2
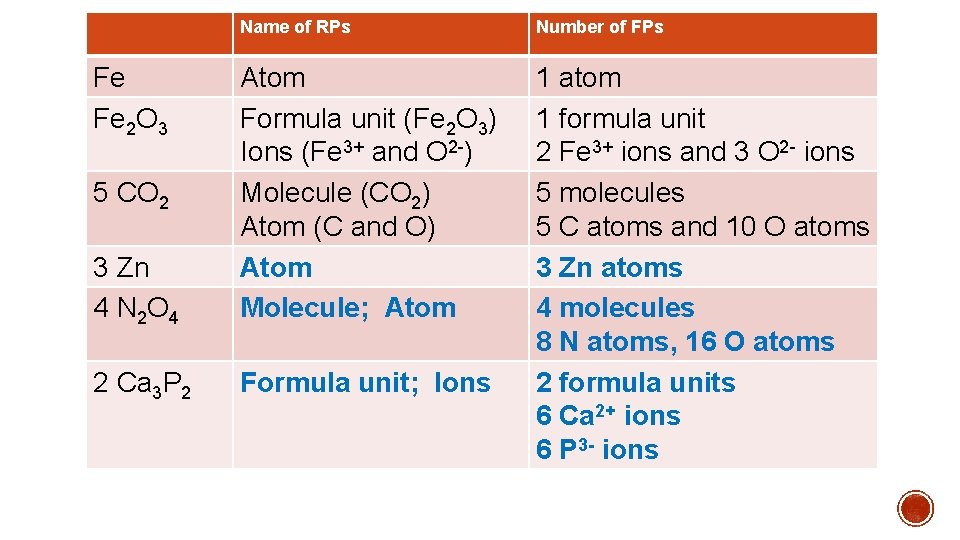
Name of RPs Number of FPs 3 Zn 4 N 2 O 4 Atom Formula unit (Fe 2 O 3) Ions (Fe 3+ and O 2 ) Molecule (CO 2) Atom (C and O) Atom Molecule; Atom 2 Ca 3 P 2 Formula unit; Ions 1 atom 1 formula unit 2 Fe 3+ ions and 3 O 2 ions 5 molecules 5 C atoms and 10 O atoms 3 Zn atoms 4 molecules 8 N atoms, 16 O atoms 2 formula units 6 Ca 2+ ions 6 P 3 - ions Fe Fe 2 O 3 5 CO 2

§ How many atoms are in 0. 150 moles of Co? 9. 03 x 1022 atoms § How many formula units are in 12. 07 moles of Na 2 O? 7. 266 x 1024 formula units

§ How many Na+ IONS are in 12. 07 moles of Na 2 O? 1. 453 x 1025 ions You Do: How many O atoms are in 0. 075 moles of Na. NO 3 ?

§ How many Na+ IONS are in 12. 07 moles of Na 2 O? 1. 453 x 1024 ions You Do: How many O atoms are in 0. 075 moles of Na. NO 3 ? 1. 4 x 1023 atoms of O

§ You Do: How many moles is 5. 07 x 1023 atoms of Zr? § You Do: How many moles is 8. 90 x 1024 molecules of CO 2?

§ You Do: How many moles is 5. 07 x 1023 atoms of Zr? 0. 842 moles of Zr § You Do: How many moles is 8. 90 x 1024 molecules of CO 2? 14. 8 moles of CO 2

§ Remember the “gaggle” of Cheerios? (1 “gaggle” = 100 Cheerios = 15. 0 g)? § Similarly, you need to know the mass of one mole of the chemical in order to convert to mass. This is also known as “molar mass. ” § You have to know this because one mole of a chemical CAN weigh differently compared to another, but they have the same NUMBER of RP’s.

The mass of one copper atom is 63. 546 amu (atomic mass units) (1 amu = 1/12 th the mass of one carbon 12 atom) (1 amu = 1. 6605 x 10 27 kg) 1. What is the mass of one copper atom in kg? 2. Convert this mass to grams. 3. Multiply the mass of one copper atom in grams by 6. 022 x 1023. What do you notice? What is the meaning of 63. 546?
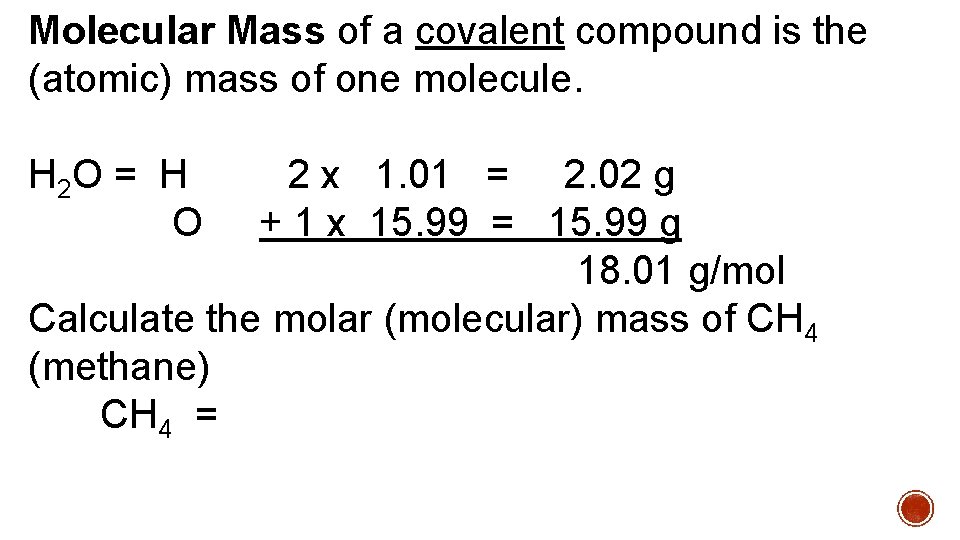
Molecular Mass of a covalent compound is the (atomic) mass of one molecule. H 2 O = H 2 x 1. 01 = 2. 02 g O + 1 x 15. 99 = 15. 99 g 18. 01 g/mol Calculate the molar (molecular) mass of CH 4 (methane) CH 4 =
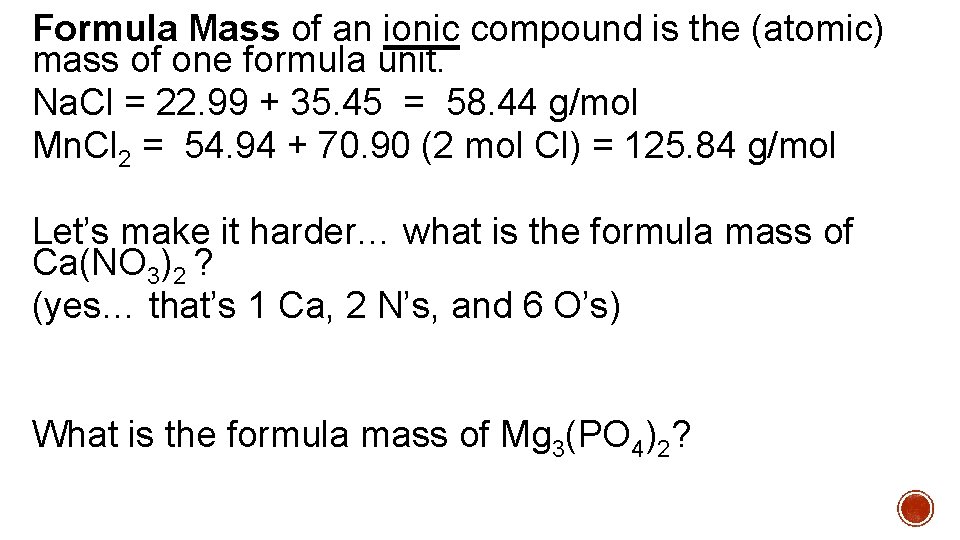
Formula Mass of an ionic compound is the (atomic) mass of one formula unit. Na. Cl = 22. 99 + 35. 45 = 58. 44 g/mol Mn. Cl 2 = 54. 94 + 70. 90 (2 mol Cl) = 125. 84 g/mol Let’s make it harder… what is the formula mass of Ca(NO 3)2 ? (yes… that’s 1 Ca, 2 N’s, and 6 O’s) What is the formula mass of Mg 3(PO 4)2?

How do you determine the amount of moles in a compound? 1. If you have 9. 0 grams of water, you have ____moles of water. Water - H 2 O = 18. 01 g/mol 9. 0 g H 2 O x 1 mole of H 2 O = 0. 50 mol of H 2 O 18. 01 g H 2 O 2. If you have 13. 0 grams of water, you have ____moles of water. 0. 722 moles of water

If you weigh out 5. 00 g of formaldehyde (formula CH 2 O), how many moles is that? 0. 166 moles What about 60. 0 g of formaldehyde? 2. 00 moles

Counting Moles 2 mol C 6 H 12 O 6 contains: § 2 mole of C 6 H 12 O 6 molecules § 2 x 6 = 12 moles of C atoms § 2 x 12 = 24 moles of H atoms § 2 x 6 = 12 moles of O atoms § 48 moles of atoms in total Complete the rest of “Counting Atoms” for BF 3 and Ca(NO 3 )2.

How many sodium ions are in 50. 0 g of Na 2 S? 7. 71 x 1023 sodium ions
You do: What is the mass of 7. 04 x 1023 molecules of PF 5? 147 g of PF 5

§I’ll run around and check your work. Always use dimensional analysis and respect the sig fig rules.
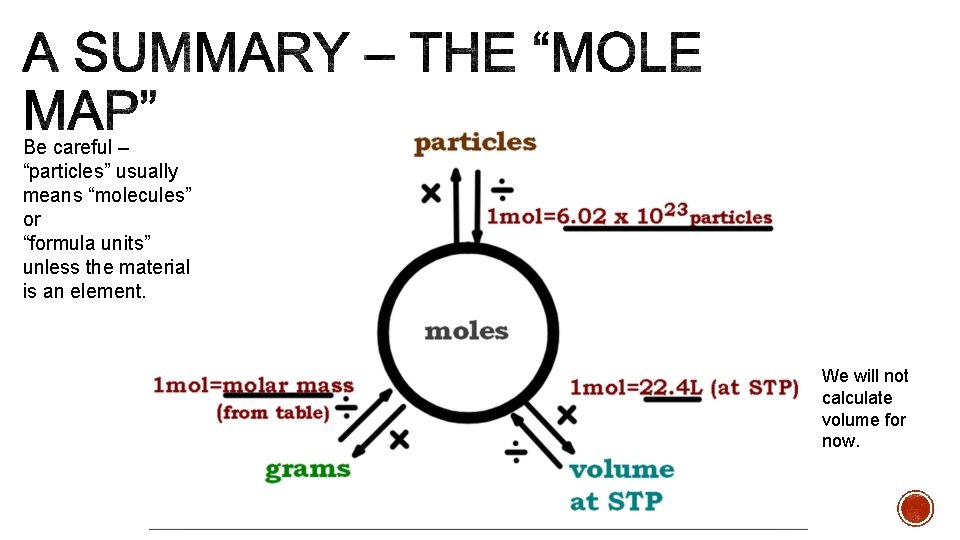
Be careful – “particles” usually means “molecules” or “formula units” unless the material is an element. We will not calculate volume for now.

§Get a person in your group to get one whiteboard person in your group and a bag of erasers and dry erase markers. §I want to see the dimensional analysis and the answer (with sig figs) for each question.

§How many atoms of potassium are in 2 moles of K 2 O? ____________

§How many atoms of potassium are in 2 moles of K 2 O? ____________ § 2 x 1024 potassium atoms

§How many molecules of water make up 5 MOLES? ____________

§How many molecules of water make up 5 MOLES? ____________ § 3 x 1024 molecules

§How many moles are 6. 022 x 1023 atoms of sodium? ____________

§How many moles are 6. 022 x 1023 atoms of sodium? ____________ § 1. 000 moles

§How many grams are in 2. 3 x 10 4 moles of calcium phosphate, Ca 3(PO 3)2?

§How many grams are in 2. 3 x 10 4 moles of calcium phosphate, Ca 3(PO 3)2? § 0. 12 grams
§How many oxygen atoms are in 3. 4 x 10 7 grams of silicon dioxide, Si. O 2?
§How many oxygen atoms are in 3. 4 x 10 7 grams of silicon dioxide, Si. O 2? § 6. 8 x 1015 oxygen atoms

https: //www. youtube. com/watch? v=Pv. T 51 M 0 ek 5 c “A Mole is a Unit”

§What did you learn today about the mole and mole conversions?
- Slides: 39