Mr Quinn Ms Tom February 11 2014 Aim

Mr. Quinn & Ms. Tom February 11, 2014 Aim: How do we determine the limiting reagent in a chemical reaction? Do Now: balance the following chemical equation. ___ YCl 3 + ___ Nb. Br 5 __ YBr 3 + ___ Nb. Cl 5

The Steps 1. Balance the equation 2. Solve for the amount of product formed for each reactant • You will get different numbers 3. Whichever reactant formed a smaller amount of product is your limiting reagent.

Example 1: Carbon and Fluorine • Graphite (C) burns in fluorine according to the following unbalanced equation. If 16 g of carbon are burned with 48 g of fluorine, what is the limiting reagent? 1. Balance the equation (list the elements, tally the elements, solve) C+ F 2 CF 4

Example 1: Carbon and Fluorine 1 C + 2 F 2 1 CF 4 2. Find the molar mass of both reactants and one product Graphite (C) Element Number Mass C 1 12 Fluorine (F 2) Total Sum of everything: Number Mass C F Sum of everything: Number F 2 Mass Sum of everything: CF 4 Element Total

Example 1: Carbon and Fluorine 3. Use dimensional analysis to predict the product formation from both reactants. Starting Mass (From problem) Mole ratio from balanced equation (Al: Al. F 3) Reciprocal of Molar Mass Reactant 1 (C) X Starting Mass (From problem) X X Mole ratio from balanced equation (F 2: Al. F 3) Reciprocal of Molar Mass Reactant 2 (F 2) X Molar mass of product (Al. F 3) X Result (in g of Al. F 3) =

Example 2: Aluminum and Fluorine • Aluminum (Al) burns in fluorine according to the following unbalanced equation. If 16 g of aluminum are burned with 57 g of fluorine, what is the limiting reagent? 1. Balance the equation (list the elements, tally the elements, solve) Al + F 2 Al. F 3

Example 1: Aluminum and Fluorine 2 Al + 3 F 2 2 Al. F 3 2. Find the molar mass of both reactants and one product Aluminum (Al) Element Number Al 1 Mass Fluorine (F 2) Total Number Mass F Sum of everything: Al. F 3 Element Number Mass Sum of everything: Total
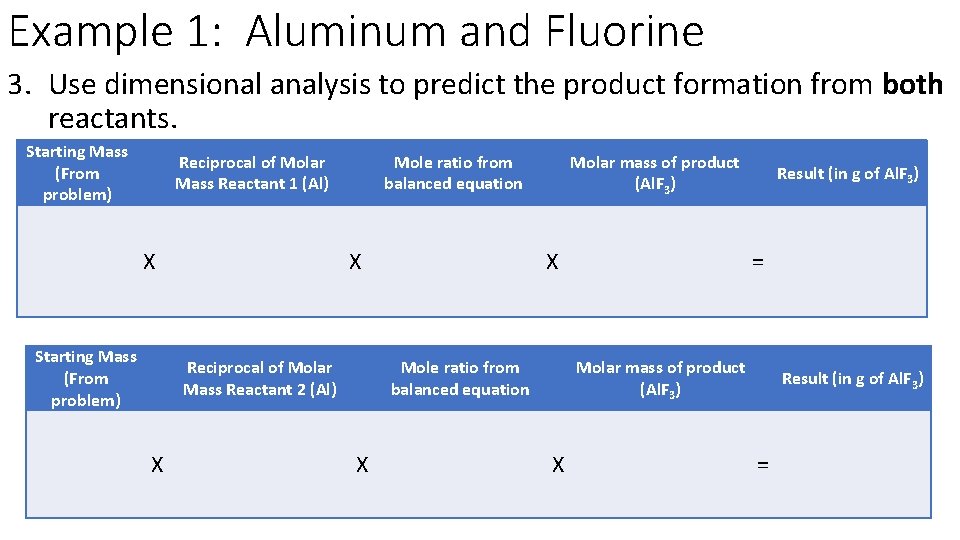
Example 1: Aluminum and Fluorine 3. Use dimensional analysis to predict the product formation from both reactants. Starting Mass (From problem) Reciprocal of Molar Mass Reactant 1 (Al) X Starting Mass (From problem) Mole ratio from balanced equation X Reciprocal of Molar Mass Reactant 2 (Al) X Molar mass of product (Al. F 3) X Mole ratio from balanced equation X Result (in g of Al. F 3) = Molar mass of product (Al. F 3) X Result (in g of Al. F 3) =

Example 1: Sulfur and Fluorine • Sulfur (S 8) burns in fluorine according to the following unbalanced equation. If 128 g of sulfur are burned with 144 g of fluorine, what is the limiting reagent? 1. Balance the equation (list the elements, tally the elements, solve) S 8 + F 2 SF 2

Example 1: Carbon and Fluorine 1 S 8 + 8 F 2 8 SF 2 2. Find the molar mass of both reactants and one product

Example 1: Carbon and Fluorine 3. Use dimensional analysis to predict the product formation from both reactants. Starting Mass (From problem) Reciprocal of Molar Mass Reactant 1 X Starting Mass (From problem) Mole ratio from balanced equation X Reciprocal of Molar Mass Reactant 2 X Molar mass of product X Mole ratio from balanced equation X Result = Molar mass of product X Result =

Your Turn!
- Slides: 12