Mr Quinn Ms Tom February 10 2014 Aim

Mr. Quinn & Ms. Tom February 10, 2014 Aim: How do we determine the limiting reagent in a chemical reaction? Do Now: balance the following chemical equations. ___ CH 4 + ___ O 2 __ CO 2 + ___ H 2 O ___ Ca. O + ___ Pb. CO 3 __ Ca. CO 3 + ___ Pb. O

The Steps 1. Balance the equation 2. Solve for the amount of product formed for each reactant • You will get different numbers 3. Whichever reactant formed a smaller amount of product is your limiting reagent.

Example 1: A Tabletop Volcano Paper Mache volcanos bubble up because of the reaction between C 2 H 4 O 2 and Na. HCO 3, according to the following balanced equation. If 10 g of C 2 H 4 O 2 and 10 g of Na. HCO 3 are combined, which is the limiting reagent? C 2 H 4 O 2 + Na. HCO 3 Na. C 2 H 3 O 2 + H 2 CO 3
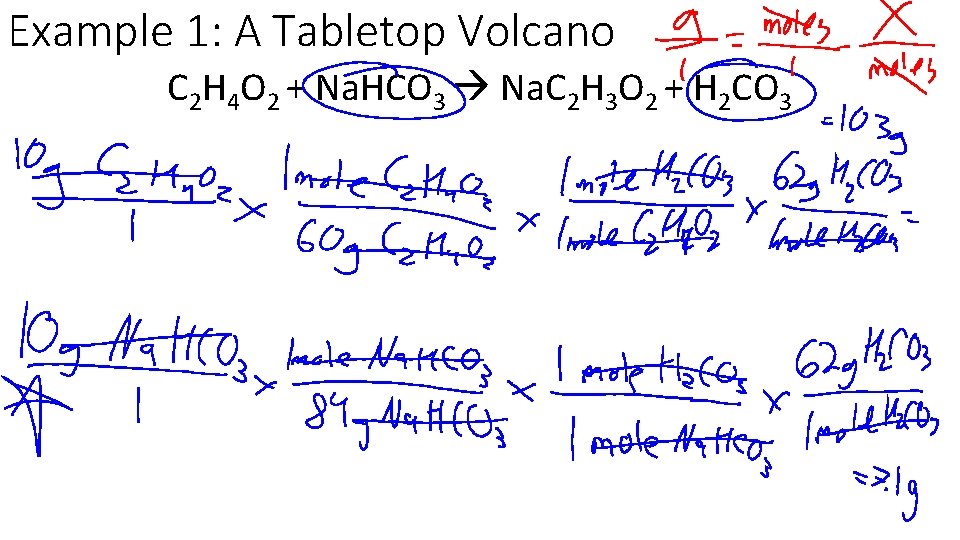
Example 1: A Tabletop Volcano C 2 H 4 O 2 + Na. HCO 3 Na. C 2 H 3 O 2 + H 2 CO 3
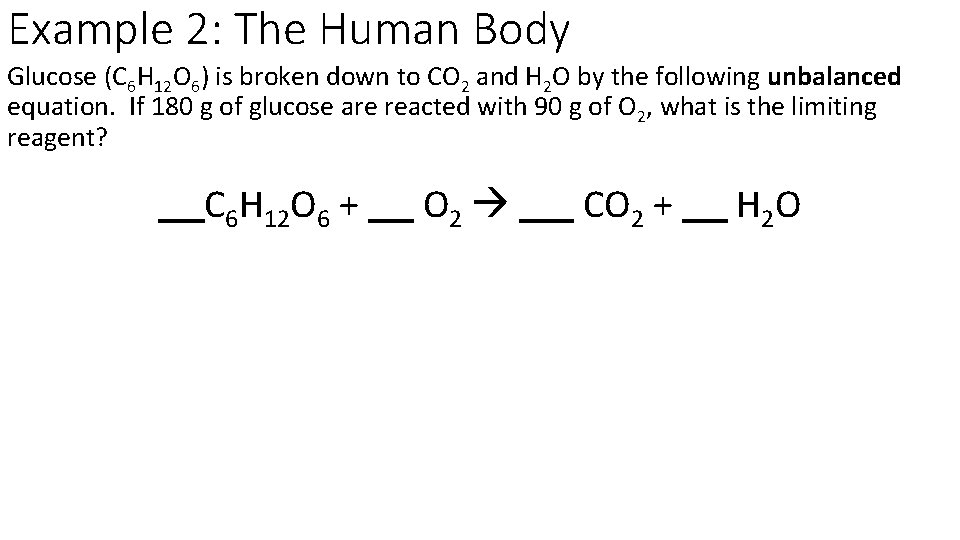
Example 2: The Human Body Glucose (C 6 H 12 O 6) is broken down to CO 2 and H 2 O by the following unbalanced equation. If 180 g of glucose are reacted with 90 g of O 2, what is the limiting reagent? C 6 H 12 O 6 + O 2 CO 2 + H 2 O
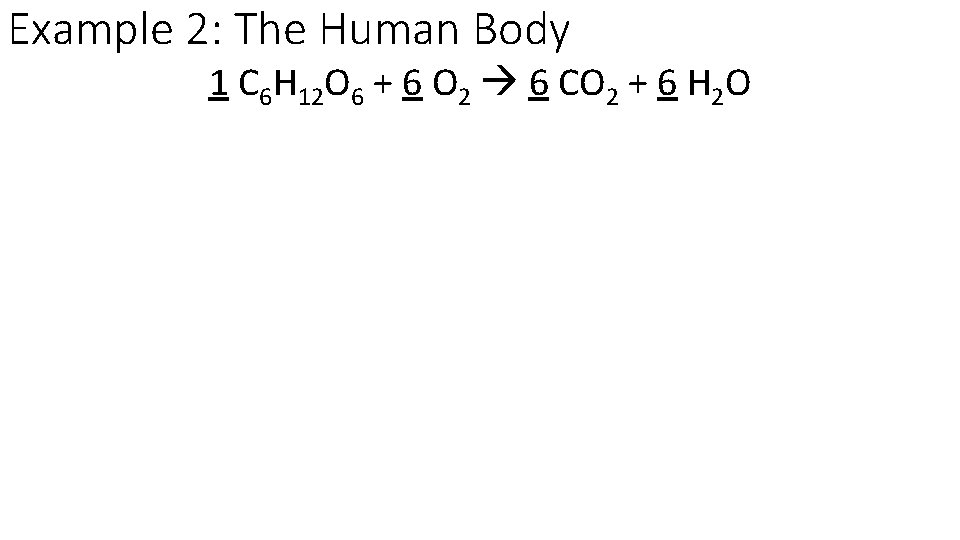
Example 2: The Human Body 1 C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O

Example 3: Architecture Zinc protects iron from rusting by being oxidized according to the following unbalanced equation. If 120 g of zinc encounter 120 g of O 2, which is the limiting reagent? Zn + O 2 Zn. O

Example 3: Architecture 2 Zn + 1 O 2 2 Zn. O

Your Turn!
- Slides: 9