Movement of materials Movement of particles Passive transport


























- Slides: 29

Movement of materials

Movement of particles • Passive transport - Does NOT need energy (diffusion, facilitated diffusion, osmosis)

Movement of particles • Passive transport - Does NOT need energy (diffusion, facilitated diffusion, osmosis) • Diffusion – random movement of particles in liquids and gases going from a high concentration to an area of low concentration.

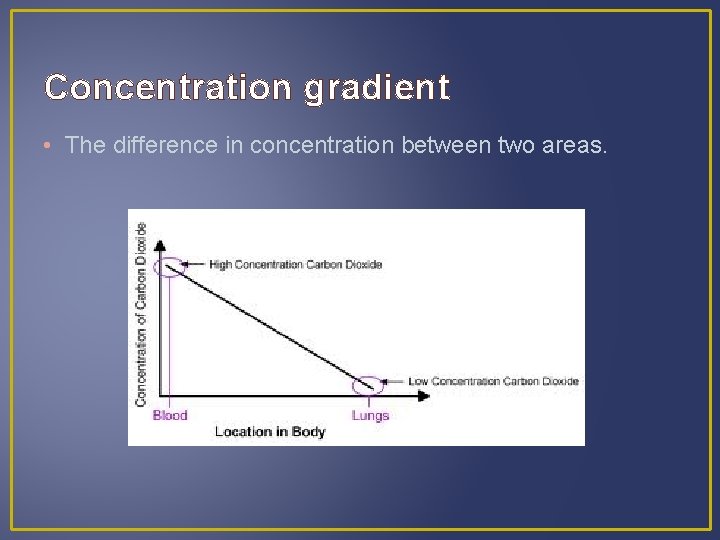
Concentration gradient • The difference in concentration between two areas.

Concentration gradient • The difference in concentration between two areas. • The higher the concentration gradient, the faster the rate of diffusion.

Concentration gradient • The difference in concentration between two areas. • The higher the concentration gradient, the faster the rate of diffusion. • Other factors affecting diffusion: - Size – small particles diffuse faster

Concentration gradient • The difference in concentration between two areas. • The higher the concentration gradient, the faster the rate of diffusion. • Other factors affecting diffusion: - Size – small particles diffuse faster (Small molecules can slip by the polar heads of the phospholipids and through the membrane to the other side. Oxygen gas, carbon dioxide and water can move in this manner. Very large molecules like proteins cannot diffuse across the membrane at all. )

Concentration gradient • The difference in concentration between two areas. • The higher the concentration gradient, the faster the rate of diffusion. • Other factors affecting diffusion: - Size – small particles diffuse faster - Temperature – higher temp = faster diffusion (In general, increases in temperature cause all molecules to move faster. Diffusion is a passive movement of molecules so quicker molecule movement translates into quicker diffusion. )

Concentration gradient • The difference in concentration between two areas. • The higher the concentration gradient, the faster the rate of diffusion. • Other factors affecting diffusion: - Size – small particles diffuse faster - Temperature – higher temp = faster diffusion - State – gas particles diffuse faster than liquid.

Facilitated diffusion • This is when a molecule diffuses faster than is possible just by the concentration gradient – facilitated = aided/helped/to make easier.

Facilitated diffusion • This is when a molecule diffuses faster than is possible just by the concentration gradient – facilitated = aided/helped/to make easier. • Transport proteins in the membrane provides channels for this process.

Facilitated diffusion • This is when a molecule diffuses faster than is possible just by the concentration gradient – facilitated = aided/helped/to make easier. • Transport proteins in the membrane provides channels for this process. • Carrier proteins are specific – only carry one type of molecule.

Facilitated diffusion • This is when a molecule diffuses faster than is possible just by the concentration gradient – facilitated = aided/helped/to make easier. • Transport proteins in the membrane provides channels for this process. • Carrier proteins are specific – only carry one type of molecule. • EG – Cytochrome (a protein) can transport oxygen 1. 8 times faster across the membrane.

Osmosis • The movement of water across a semi-permeable membrane from an area of high water potential, to an area of low water potential until water potentials are equal. • http: //www. youtube. com/watch? v=AYNwynwa. ALo

Osmosis • The movement of water across a semi-permeable membrane from an area of high water potential, to an area of low water potential until water potentials are equal. • http: //www. youtube. com/watch? v=AYNwynwa. ALo • High water potential means there are few solute particles dissolved in it – eg. Fresh water from the tap.

Osmosis • The movement of water across a semi-permeable membrane from an area of high water potential, to an area of low water potential until water potentials are equal. • http: //www. youtube. com/watch? v=AYNwynwa. ALo • High water potential means there are few solute particles dissolved in it – eg. Fresh water from the tap. • Sea water has many particles of salt dissolved in it, therefore the water potential is low.


Osmosis • A dilute (weak) solution is hypotonic

Osmosis • A dilute (weak) solution is hypotonic • A concentrated (strong) solution is hypertonic

osmosis • A dilute (weak) solution is hypotonic • A concentrated (strong) solution is hypertonic • Two solutions with the same concentration are isotonic.

Osmosis • • A dilute (weak) solution is hypotonic A concentrated (strong) solution is hypertonic Two solutions with the same concentration are isotonic. Diffusion and osmosis mean that particles move in both directions across the membrane, however the net direction is towards the area of lower concentration.

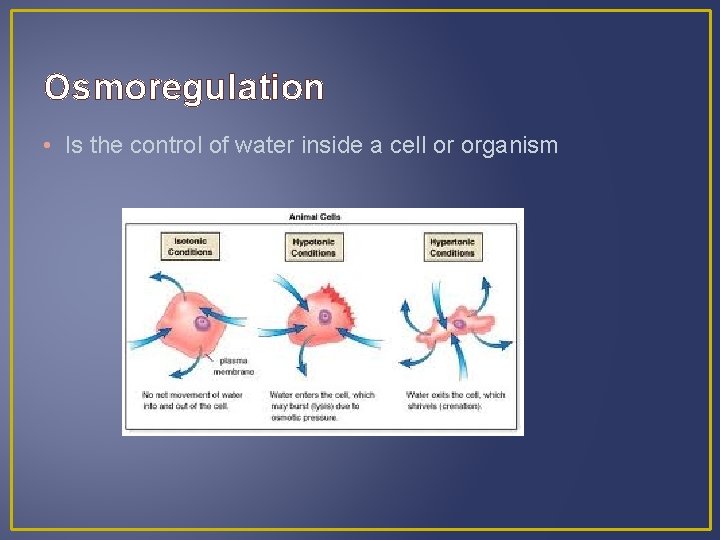
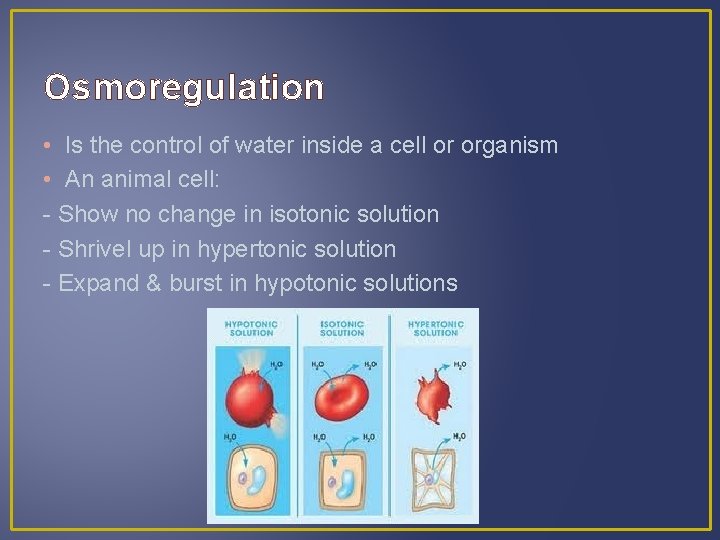
Osmoregulation • Is the control of water inside a cell or organism

Osmoregulation • Is the control of water inside a cell or organism • An animal cell: - Show no change in isotonic solution

Osmoregulation • Is the control of water inside a cell or organism • An animal cell: - Show no change in isotonic solution - Shrivel up in hypertonic solution

Osmoregulation • Is the control of water inside a cell or organism • An animal cell: - Show no change in isotonic solution - Shrivel up in hypertonic solution - Expand & burst in hypotonic solutions

Osmoregulation in Plant cells • Show no change in isotonic solution

Osmoregulation in Plant cells • Show no change in isotonic solution • Become plasmolysed in hypertonic solutions – where water drains from the vacuole and the membrane pulls away from the cell wall = the cells become flaccid and the plant wilts.

Osmoregulation in Plant cells • Show no change in isotonic solution • Become plasmolysed in hypertonic solutions – where water drains from the vacuole and the membrane pulls away from the cell wall = the cells become flaccid and the plant wilts. • Become firm in hypotonic solution – turgid. The cell wall stops the cell bursting and the plant remains upright.

Osmoregulation in Plant cells • Show no change in isotonic solution • Become plasmolysed in hypertonic solutions – where water drains from the vacuole and the membrane pulls away from the cell wall = the cells become flaccid and the plant wilts. • Become firm in hypotonic solution – turgid. The cell wall stops the cell bursting and the plant remains upright. *** Read page 113 and complete pages 114 and 115 in Biozone