Morphine Named after the Greek God Morpheus God

























- Slides: 79


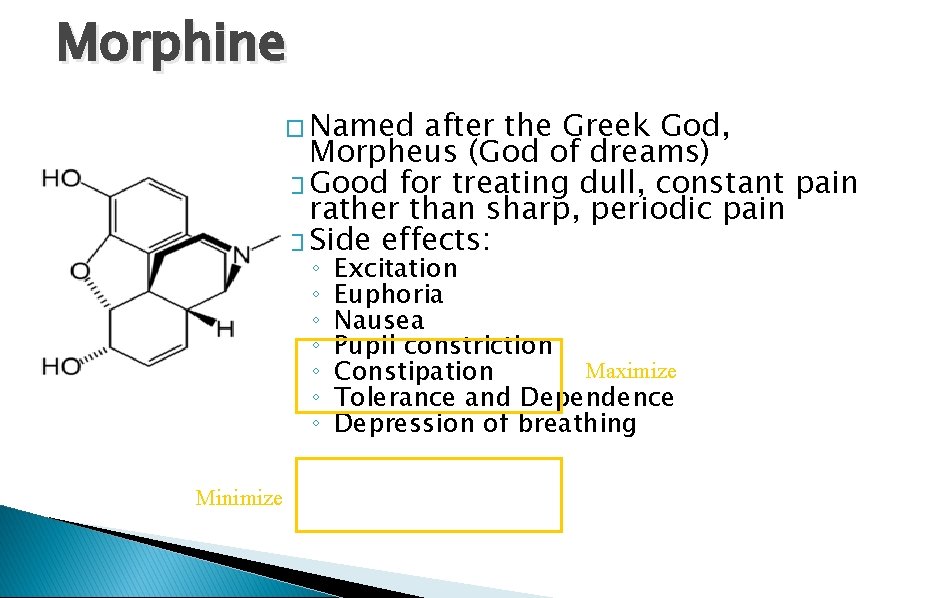
Morphine � Named after the Greek God, Morpheus (God of dreams) � Good for treating dull, constant pain rather than sharp, periodic pain � Side effects: ◦ ◦ ◦ ◦ Minimize Excitation Euphoria Nausea Pupil constriction Maximize Constipation Tolerance and Dependence Depression of breathing

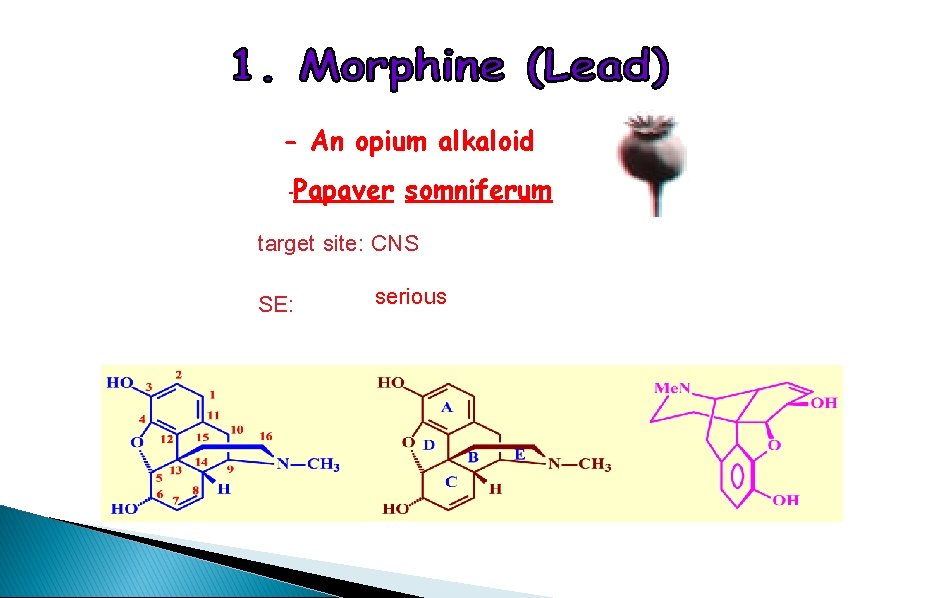
- An opium alkaloid -Papaver somniferum target site: CNS SE: serious

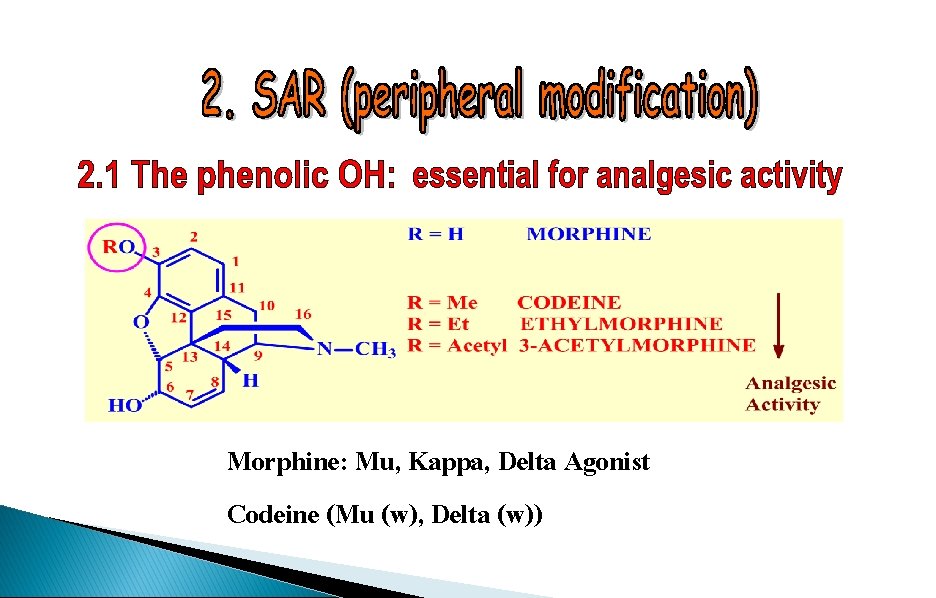
Morphine: Mu, Kappa, Delta Agonist Codeine (Mu (w), Delta (w))

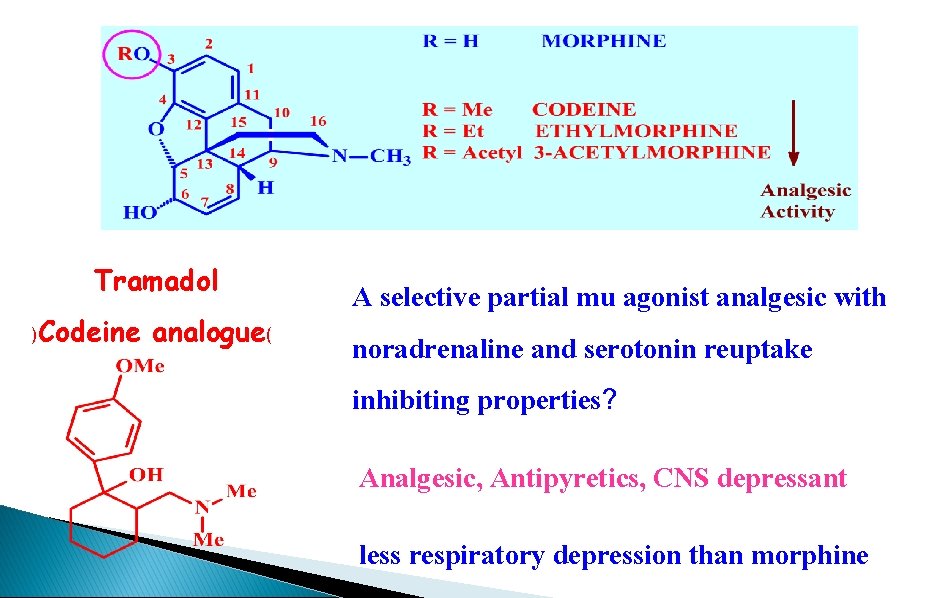
Tramadol )Codeine analogue( A selective partial mu agonist analgesic with noradrenaline and serotonin reuptake inhibiting properties? Analgesic, Antipyretics, CNS depressant less respiratory depression than morphine

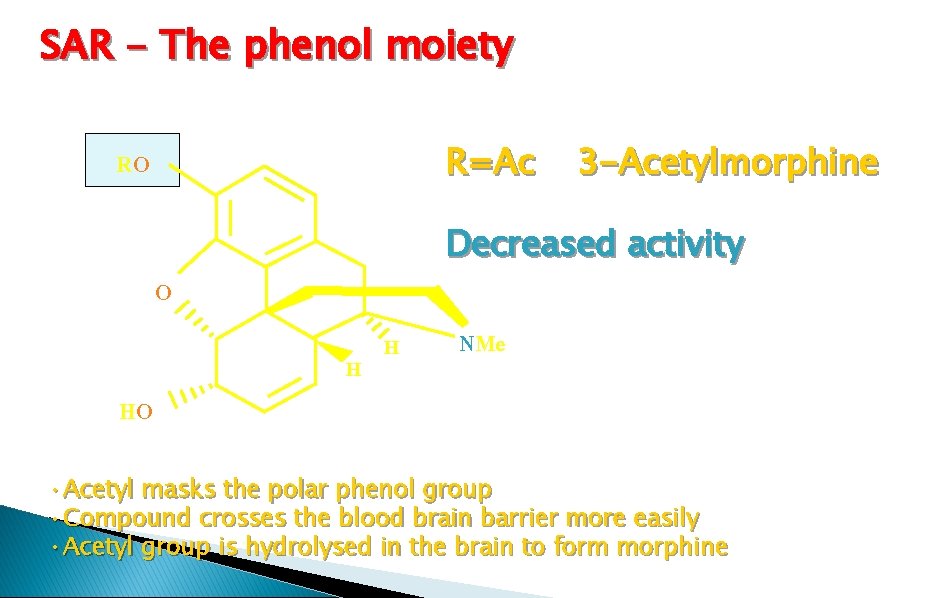
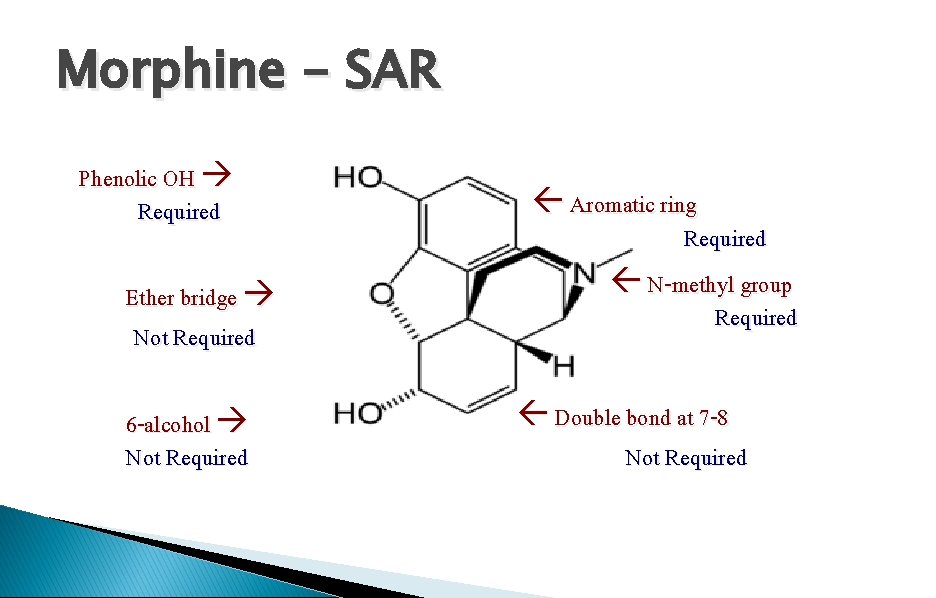
SAR - The phenol moiety Notes Codeine is metabolised in the liver to morphine. The activity observed is due to morphine. Codeine is used for mild pain and coughs Weaker analgesic but weaker side effects. Conclusion Masking phenol is bad for activity

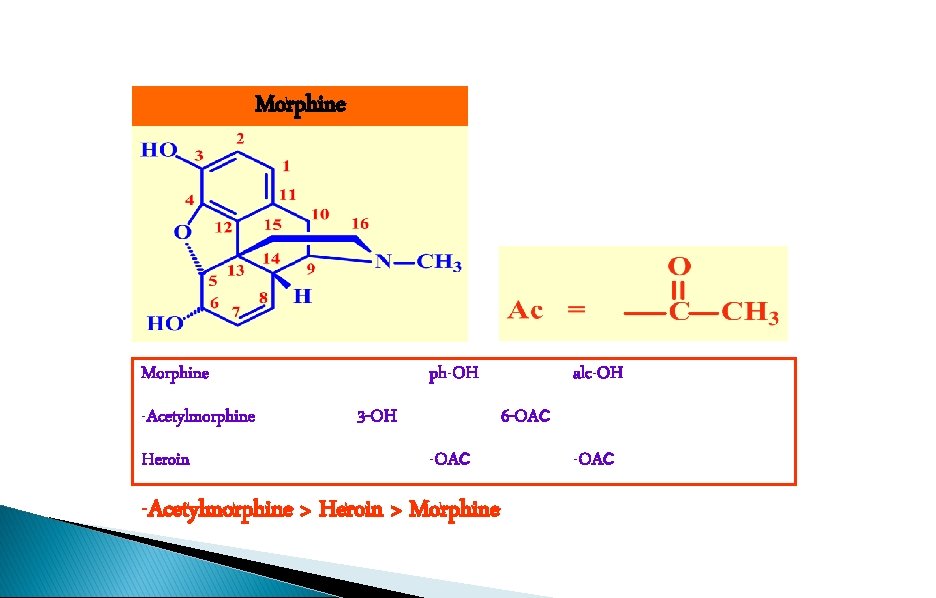
SAR - The phenol moiety R=Ac RO Decreased activity O HO 3 -Acetylmorphine H H NMe • Acetyl masks the polar phenol group • Compound crosses the blood brain barrier more easily • Acetyl group is hydrolysed in the brain to form morphine

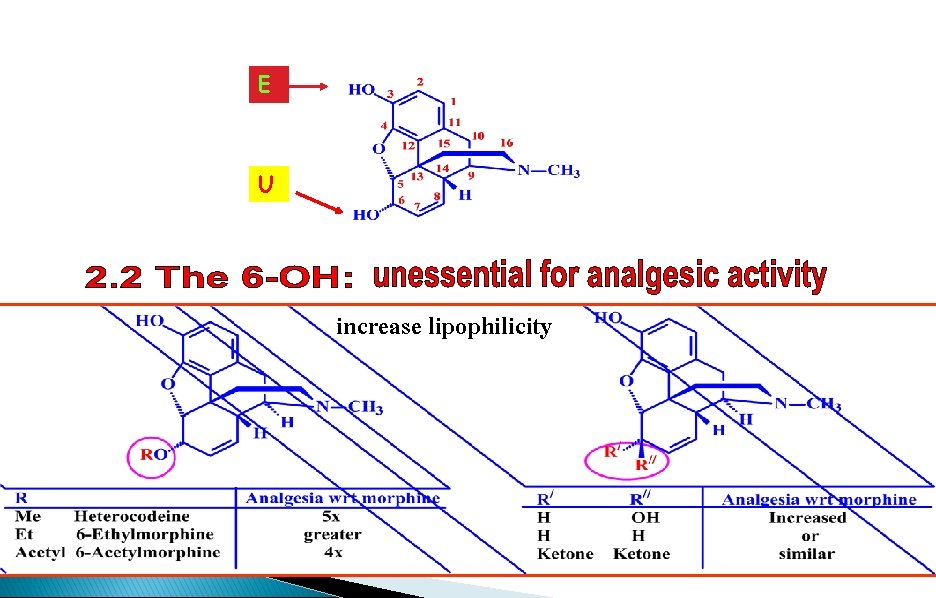
E U increase lipophilicity

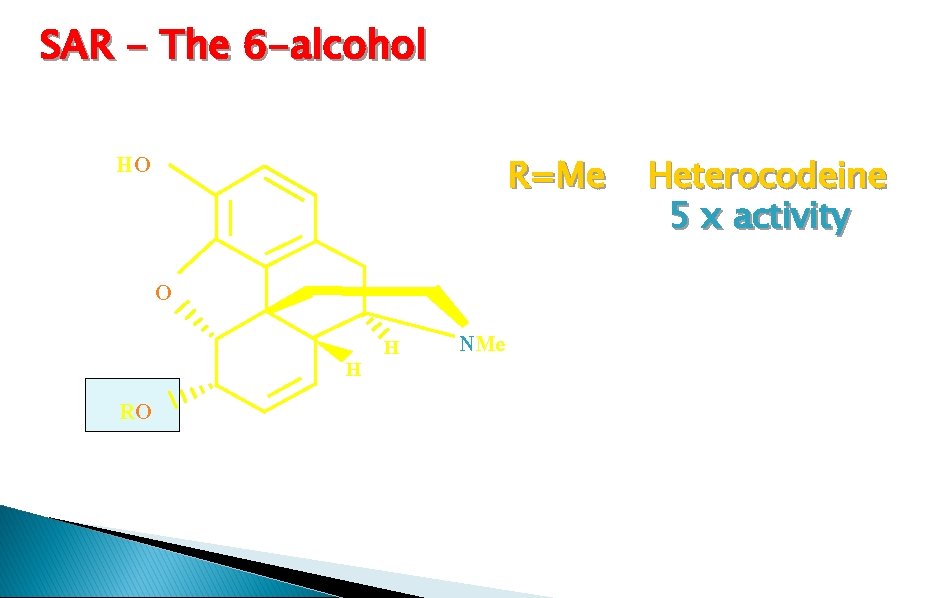
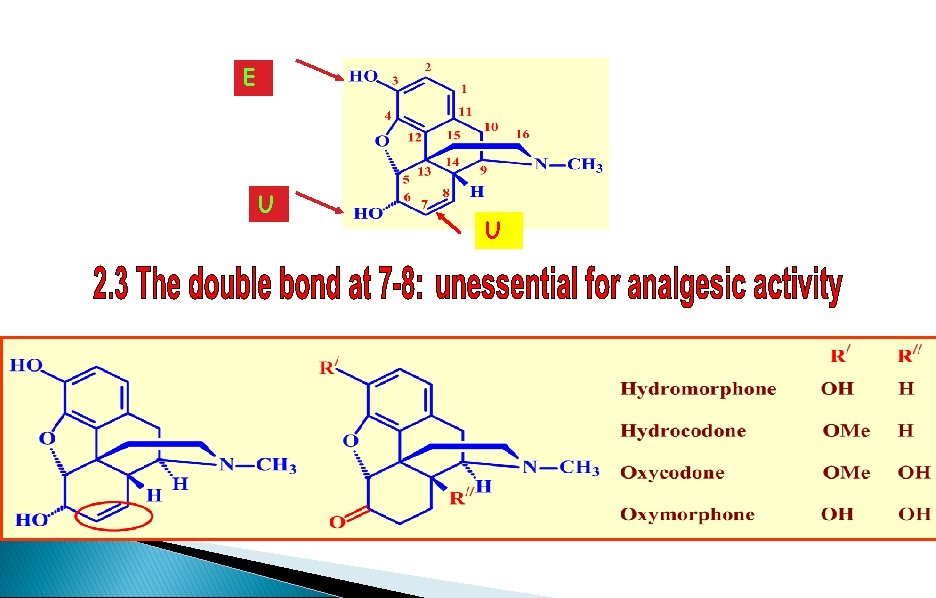
SAR - The 6 -alcohol HO R=Me O RO H H NMe Heterocodeine 5 x activity

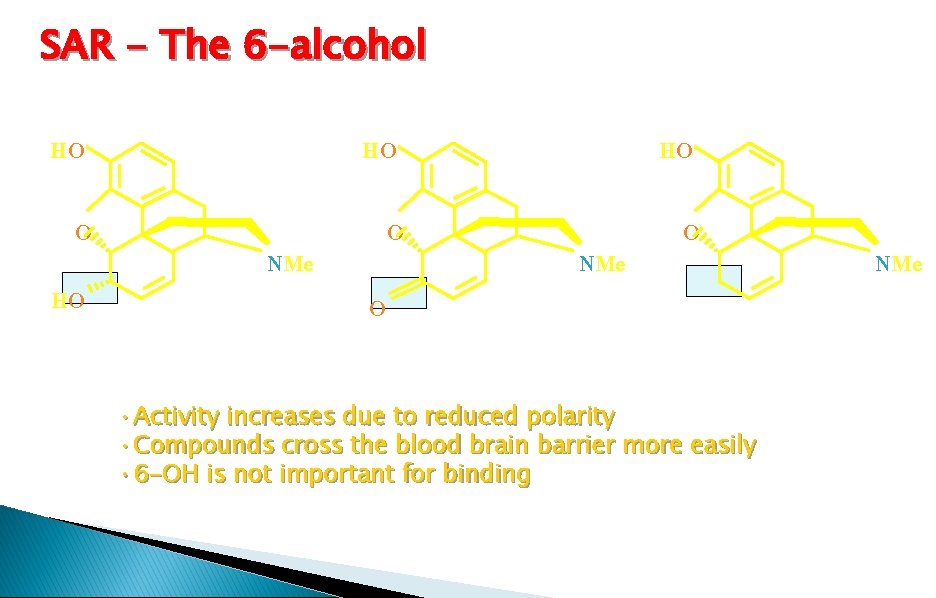
SAR - The 6 -alcohol HO HO HO NMe • Activity increases due to reduced polarity • Compounds cross the blood brain barrier more easily • 6 -OH is not important for binding NMe

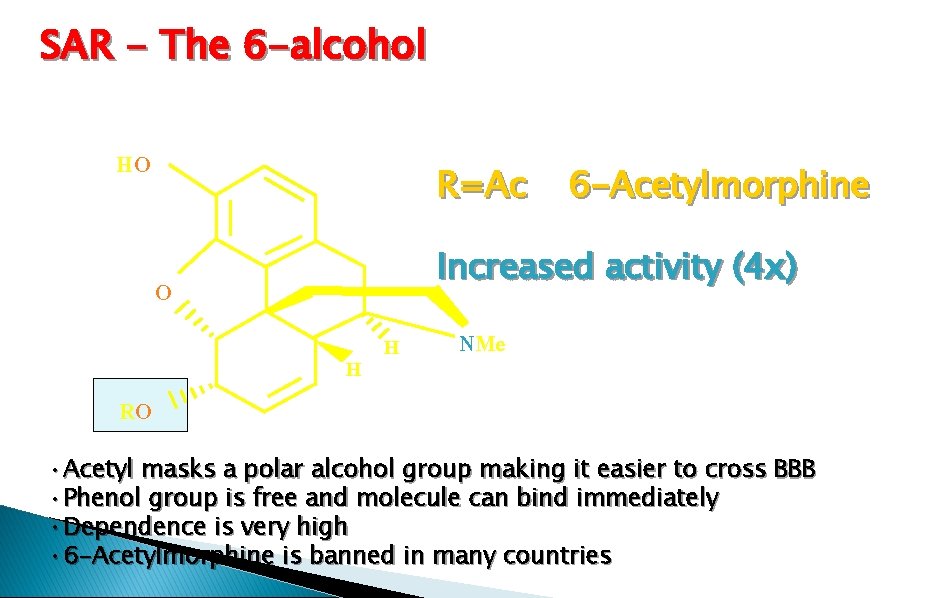
SAR - The 6 -alcohol HO R=Ac Increased activity (4 x) O RO 6 -Acetylmorphine H H NMe • Acetyl masks a polar alcohol group making it easier to cross BBB • Phenol group is free and molecule can bind immediately • Dependence is very high • 6 -Acetylmorphine is banned in many countries

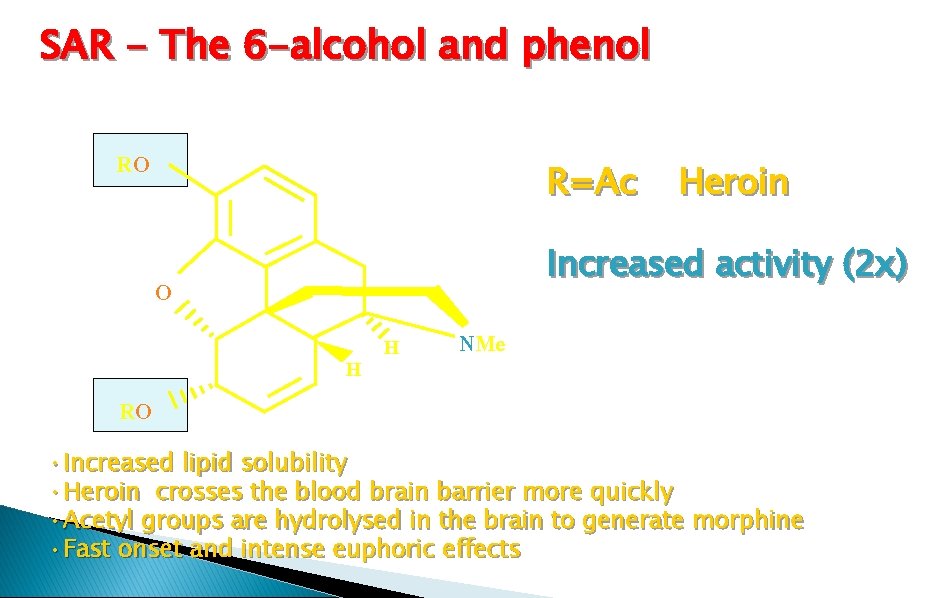
SAR - The 6 -alcohol and phenol RO R=Ac Increased activity (2 x) O RO Heroin H H NMe • Increased lipid solubility • Heroin crosses the blood brain barrier more quickly • Acetyl groups are hydrolysed in the brain to generate morphine • Fast onset and intense euphoric effects

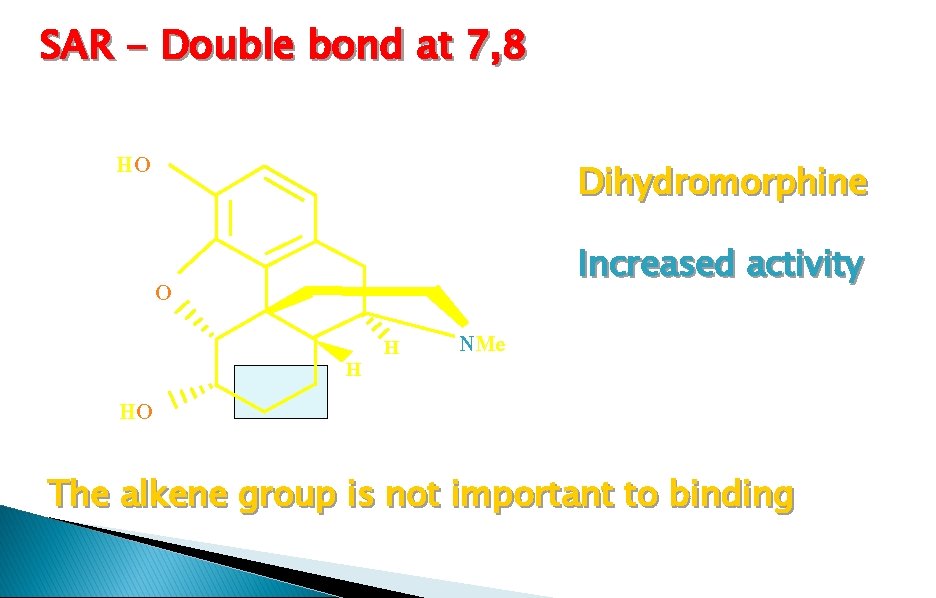
SAR - Double bond at 7, 8 HO Dihydromorphine Increased activity O HO H H NMe The alkene group is not important to binding

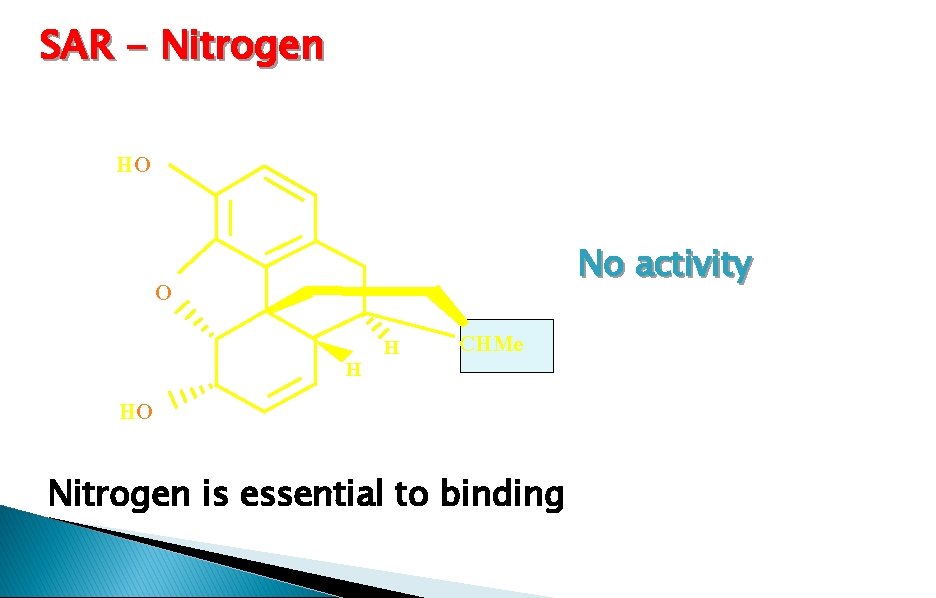
SAR - Nitrogen HO No activity O HO H H CHMe Nitrogen is essential to binding

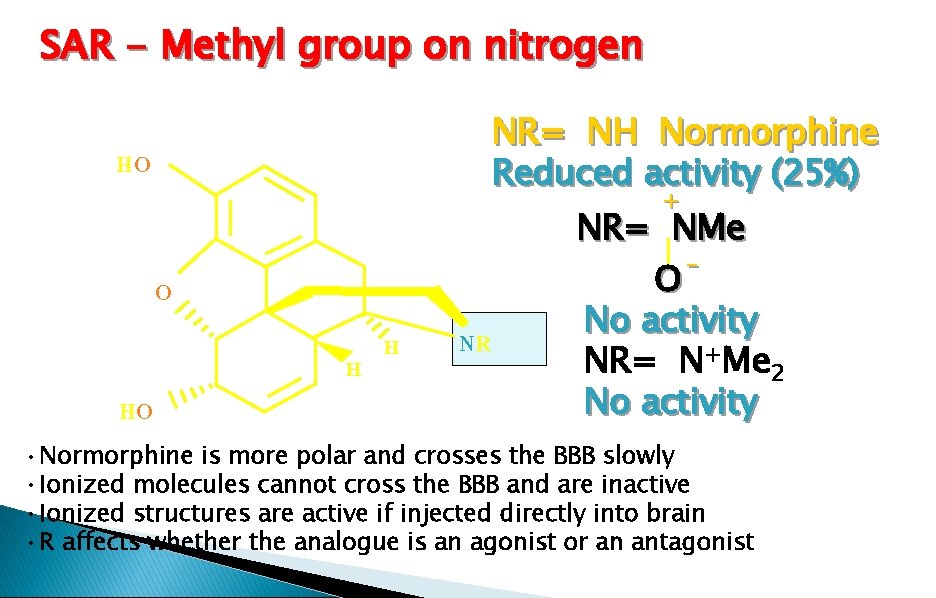
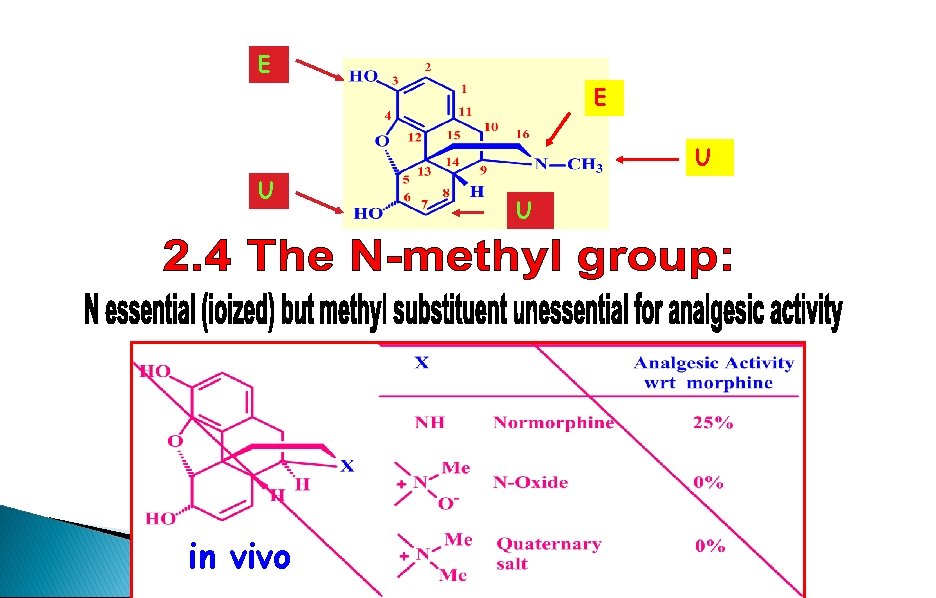
SAR - Methyl group on nitrogen HO NR= NH Normorphine Reduced activity (25%) HO NR= NMe O No activity NR= N+Me 2 No activity + O H H NR • Normorphine is more polar and crosses the BBB slowly • Ionized molecules cannot cross the BBB and are inactive • Ionized structures are active if injected directly into brain • R affects whether the analogue is an agonist or an antagonist

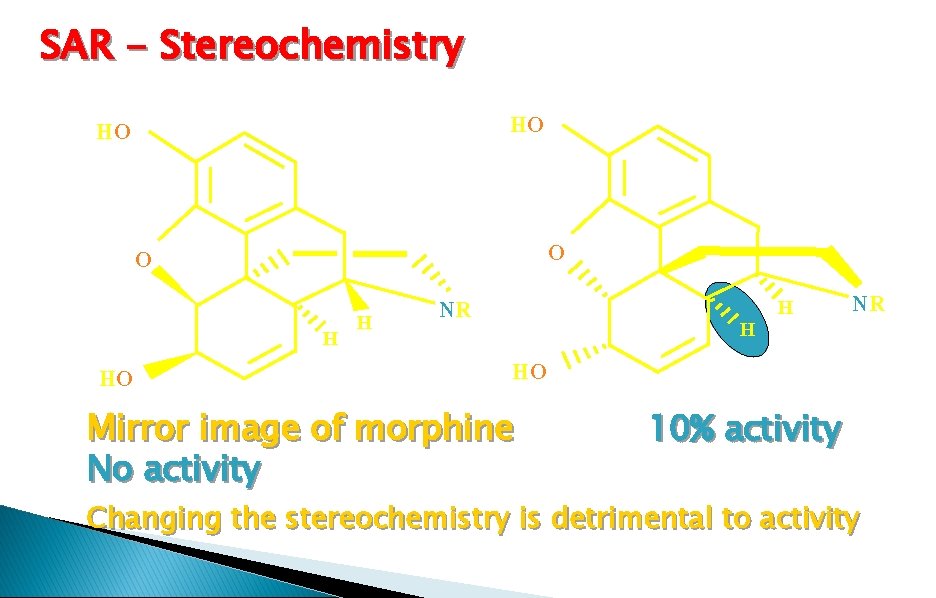
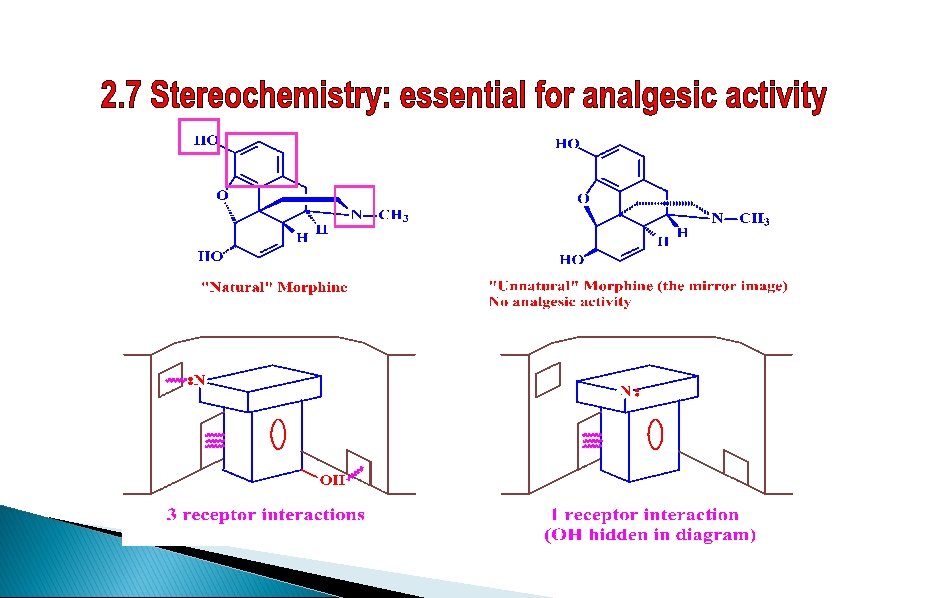
SAR - Stereochemistry HO HO O O HO H H NR HO Mirror image of morphine No activity H H NR 10% activity Changing the stereochemistry is detrimental to activity

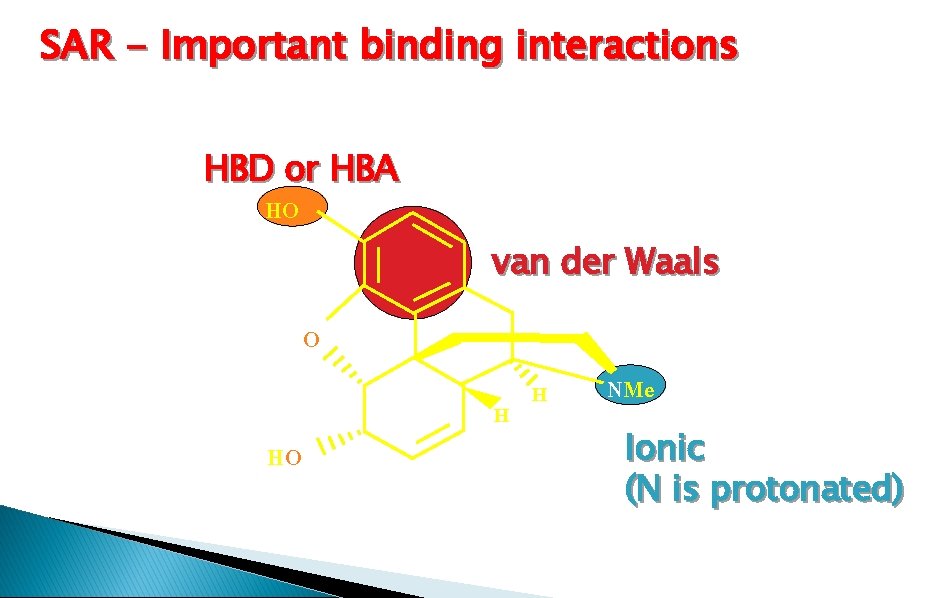
SAR - Important binding interactions HBD or HBA HO van der Waals O HO H H NMe Ionic (N is protonated)

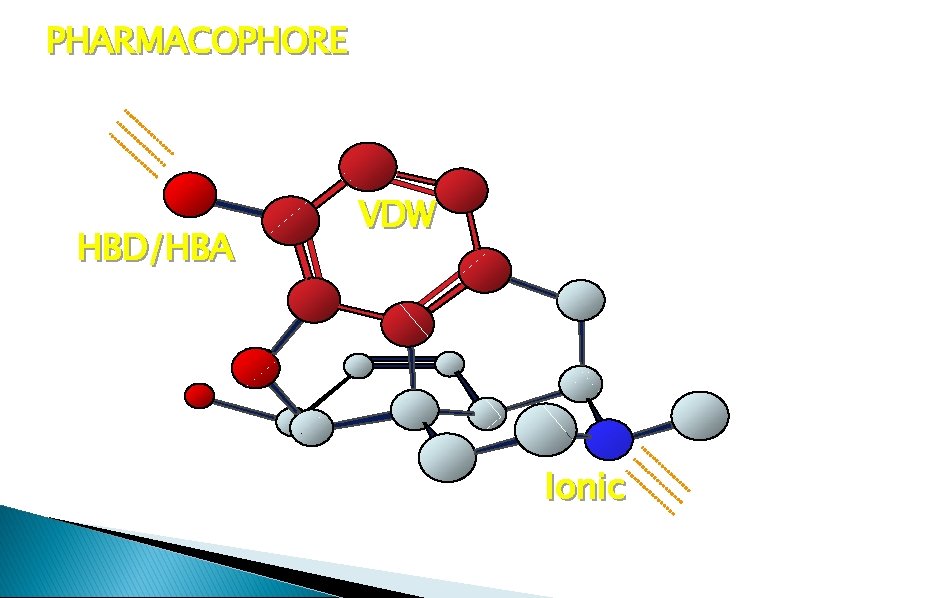
PHARMACOPHORE HBD/HBA VDW Ionic

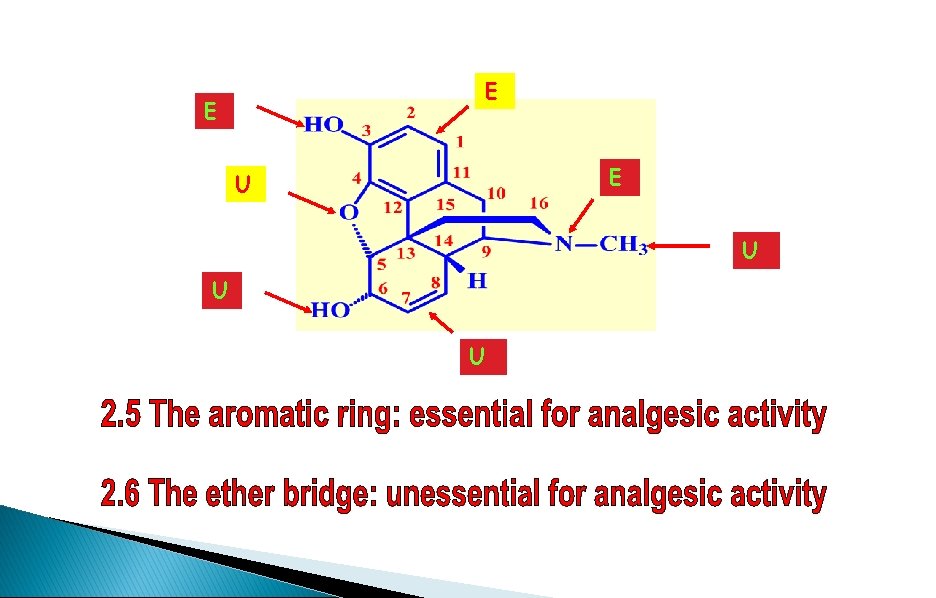
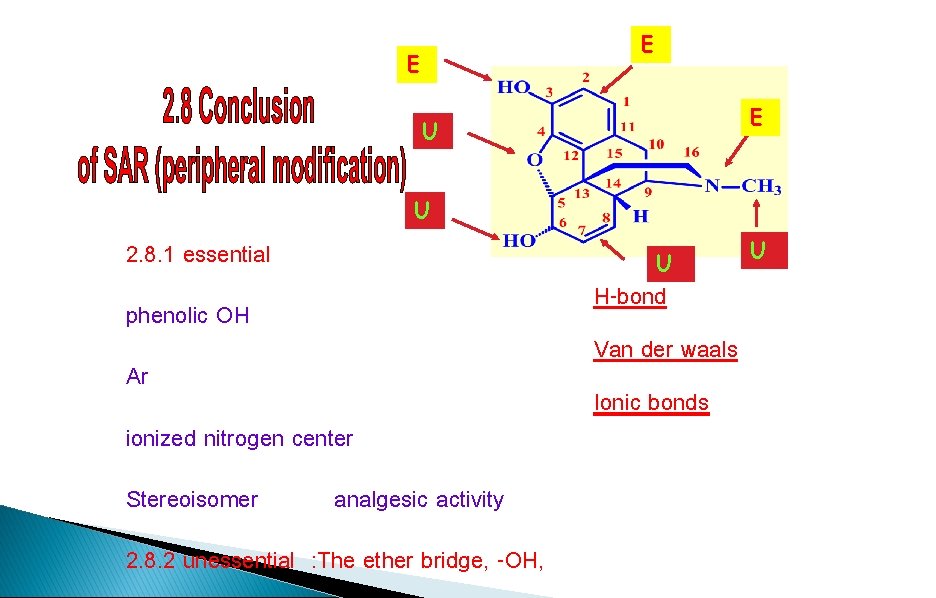
Morphine - SAR Phenolic OH Required Ether bridge Not Required 6 -alcohol Not Required Aromatic ring Required N-methyl group Required Double bond at 7 -8 Not Required

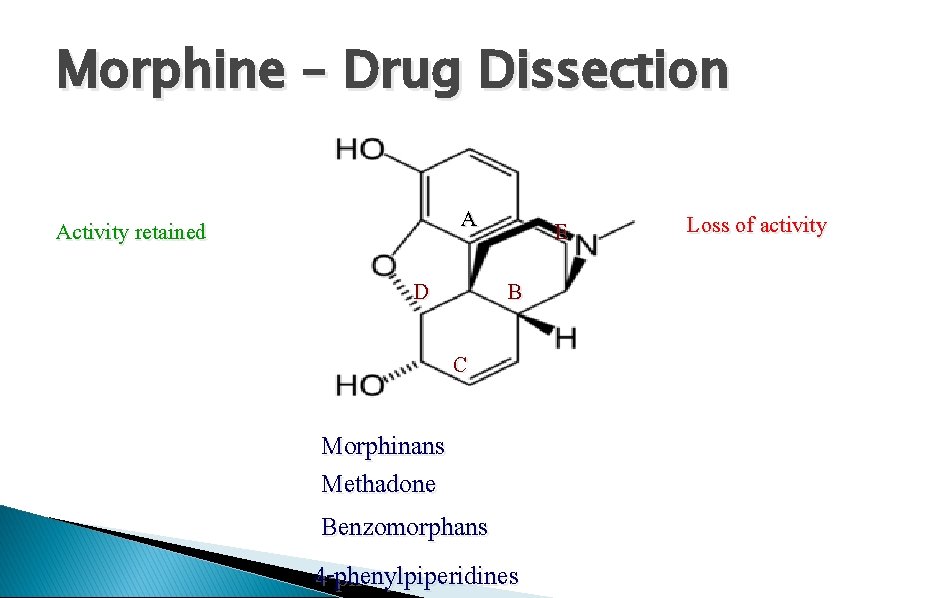
Morphine – Drug Dissection A Activity retained D E B C Morphinans Methadone Benzomorphans 4 -phenylpiperidines Loss of activity

Morphine -Acetylmorphine Heroin ph-OH 3 -OH alc-OH 6 -OAc -Acetylmorphine > Heroin > Morphine -OAc

E U U

E E U U in vivo U

E E E U U


E E E U U 2. 8. 1 essential U H-bond phenolic OH Van der waals Ar ionized nitrogen center Stereoisomer analgesic activity 2. 8. 2 unessential : The ether bridge, -OH, Ionic bonds U

morphine codeine 6 - acetylmorphine heroin hydromorphone hydrocodone oxymorphone oxycodone

- drug extension - simplification - rigidification

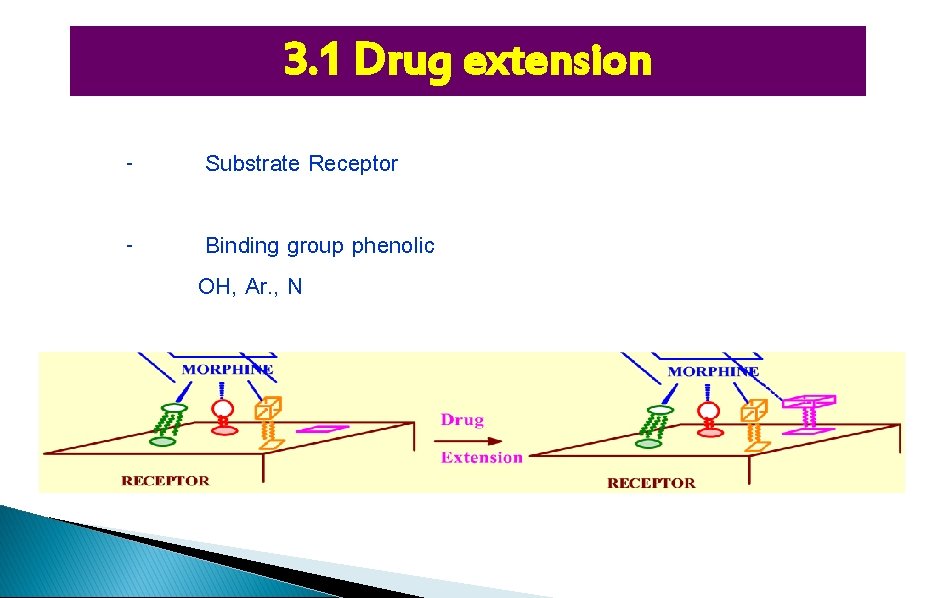
3. 1 Drug extension - Substrate Receptor - Binding group phenolic OH, Ar. , N

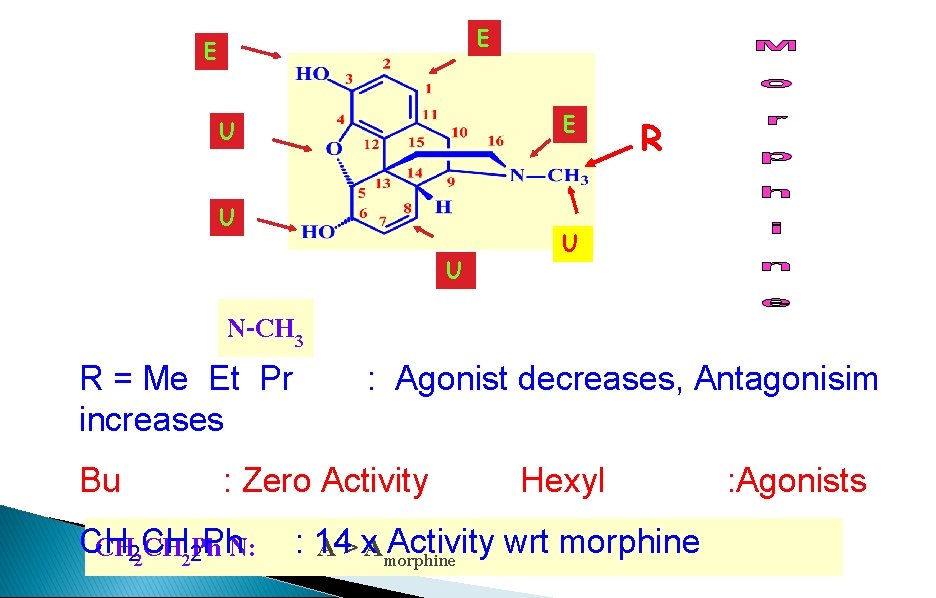
E E E U U U R U N-CH 3 R = Me Et Pr increases Bu : Agonist decreases, Antagonisim : Zero Activity CH CH 22 Ph. N: Hexyl : 14 Activity wrt morphine A > x. Amorphine : Agonists

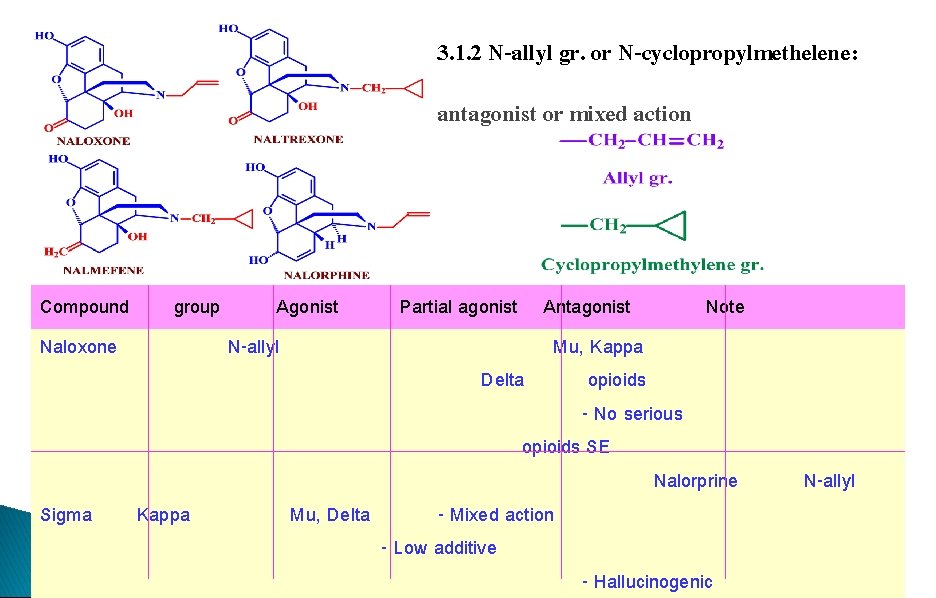
3. 1. 2 N-allyl gr. or N-cyclopropylmethelene: antagonist or mixed action Compound Naloxone Sigma group Kappa Agonist N-allyl Partial agonist Antagonist Note Mu, Kappa Delta opioids - No serious opioids SE Nalorprine Mu, Delta - Mixed action - Low additive - Hallucinogenic N-allyl

Naloxone Naltrexon e Nalmefen e

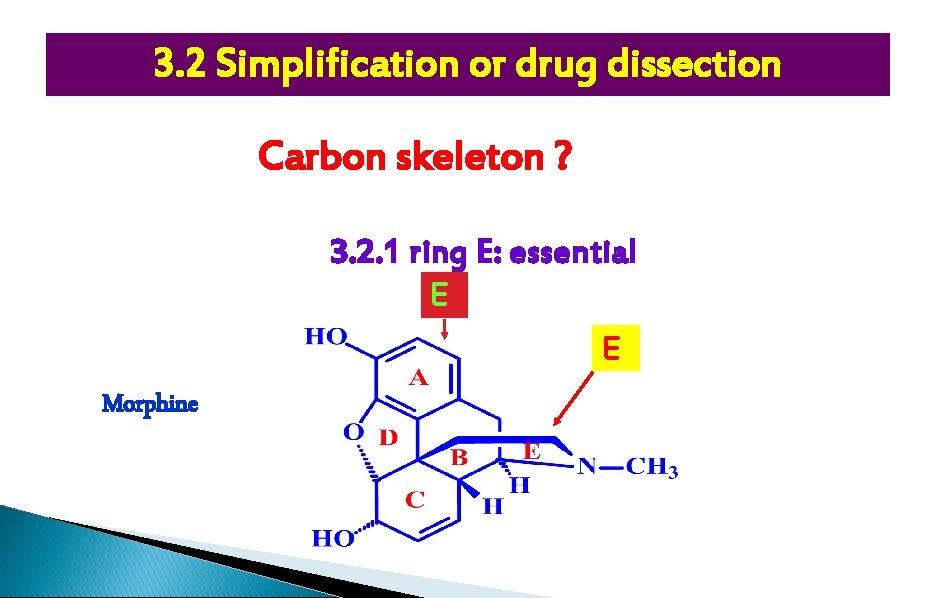
3. 2 Simplification or drug dissection Carbon skeleton ? 3. 2. 1 ring E: essential E E Morphine

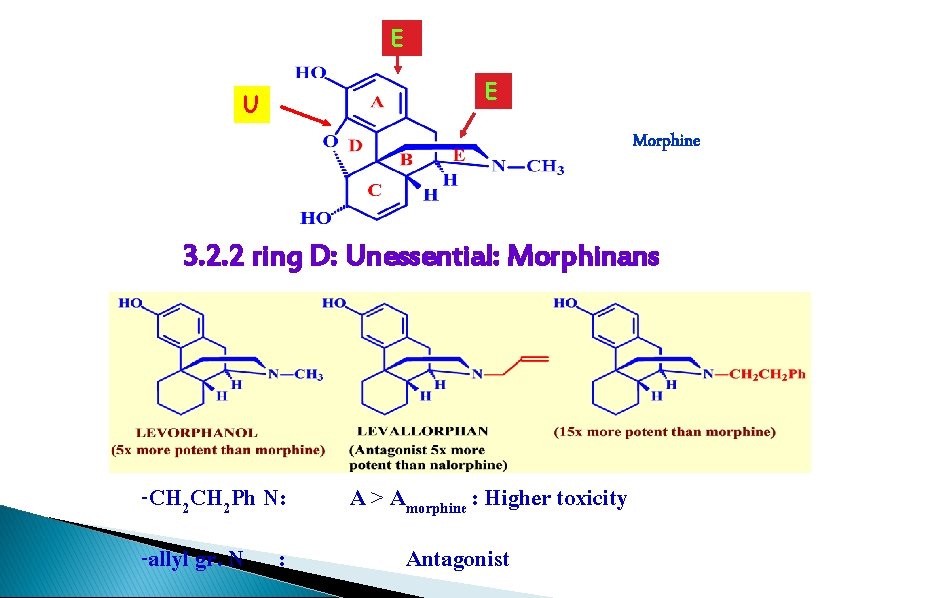
E U E Morphine 3. 2. 2 ring D: Unessential: Morphinans -CH 2 Ph N: -allyl gr. N : A > Amorphine : Higher toxicity Antagonist

Morphine Analogues - Morphinans Levorphanol � How it’s related: � Activity: � Advantage: � Side effects: � Marketed as Levo-Dromoran® ◦ Ether bridge removed ◦ 5 x that of morphine ◦ It can be taken orally ◦ Lasts longer ◦ Easier to synthesize ◦ High toxicity, comparable dependence

1. ring D is not essential 2. more active, longer acting but more SE than morphine: Levorphanol: orally active 3. Morphinans and Morphine: reacting with the same receptors in the same way.

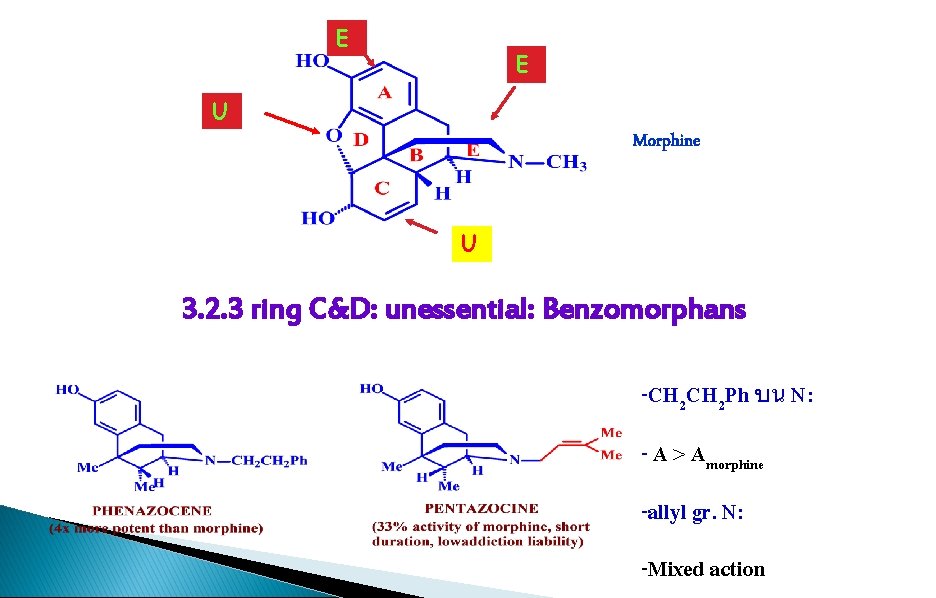
E E U Morphine U 3. 2. 3 ring C&D: unessential: Benzomorphans -CH 2 Ph บน N: - A > Amorphine -allyl gr. N: -Mixed action

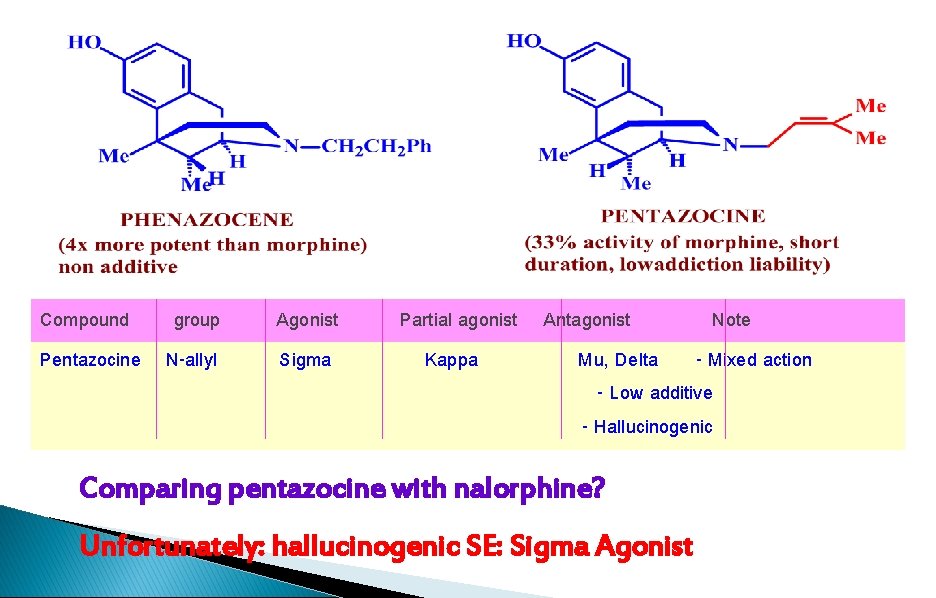
Compound group Pentazocine N-allyl Agonist Sigma Partial agonist Antagonist Note Kappa Mu, Delta - Mixed action - Low additive - Hallucinogenic Comparing pentazocine with nalorphine? Unfortunately: hallucinogenic SE: Sigma Agonist

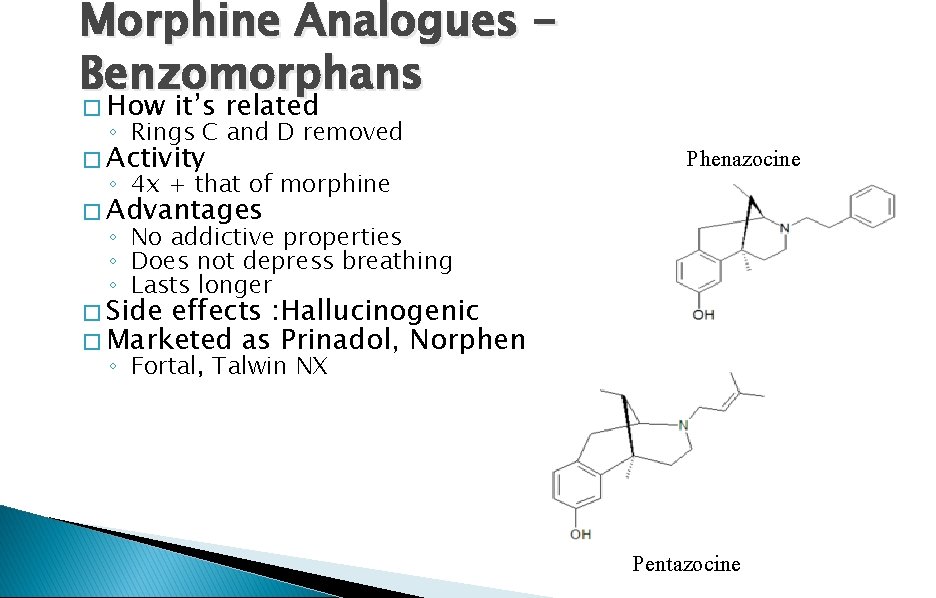
Morphine Analogues Benzomorphans � How it’s related ◦ Rings C and D removed � Activity ◦ 4 x + that of morphine Phenazocine � Advantages ◦ No addictive properties ◦ Does not depress breathing ◦ Lasts longer � Side effects : Hallucinogenic � Marketed as Prinadol, Norphen ◦ Fortal, Talwin NX Pentazocine

1. ring C, D are not essential 2. analgesia and addiction are not necessarily coexistent 3. reasonable analgesic activity, less addictive liability, less tolerance 4. simpler to synthesize 5. phenazocene, pentazocine 6. hallucinogenic effect

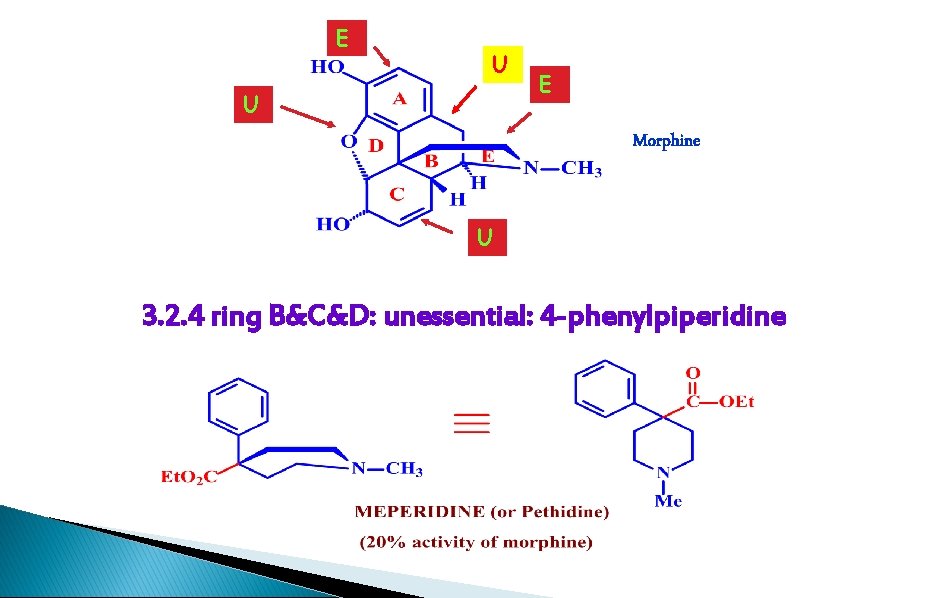
E U U E Morphine U 3. 2. 4 ring B&C&D: unessential: 4 -phenylpiperidine

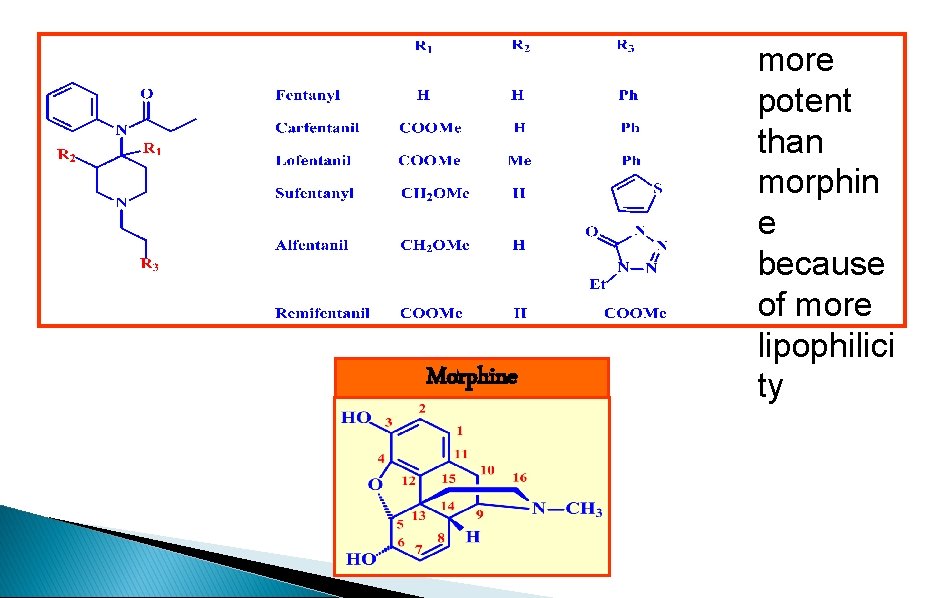
Morphine more potent than morphin e because of more lipophilici ty

Morphine Analogues – 4 phenylpiperidines Fentanyl � How it’s related: ◦ Rings B, C, D removed � Activity: Fentanyl ◦ 100 x that of morphine � Advantages: ◦ ◦ Cross BBB efficiently Really easy to make Rapid onset, short duration Can be administered any way (IV, oral, transdermal, buccal)

Morphine Analogues – 4 phenylpiperidines � Used for: ◦ Anesthesia ◦ Chronic pain management � Side effects: ◦ Sudden respiratory depression ◦ More addictive than heroin ◦ Less euphoria, more sedation � Marketed as: ◦ Sufenta (used in ♥ surgery) ◦ Carfentanil (used in vet practice) ◦ “Percopop”, Oxy. Contin, “magic” (heroin/cocaine)


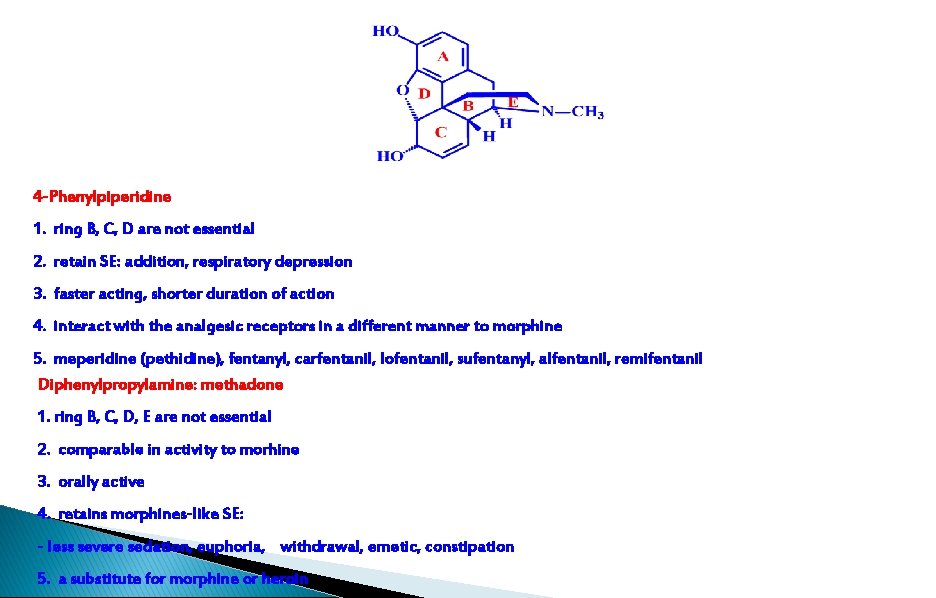
1. ring B, C, D are not essential 2. retain SE: addition, respiratory depression 3. faster acting, shorter duration of action 4. interact with the analgesic receptors in a different manner to morphine 5. meperidine (pethidine), fentanyl, carfentanil, lofentanil, sufentanyl, alfentanil, remifentanil

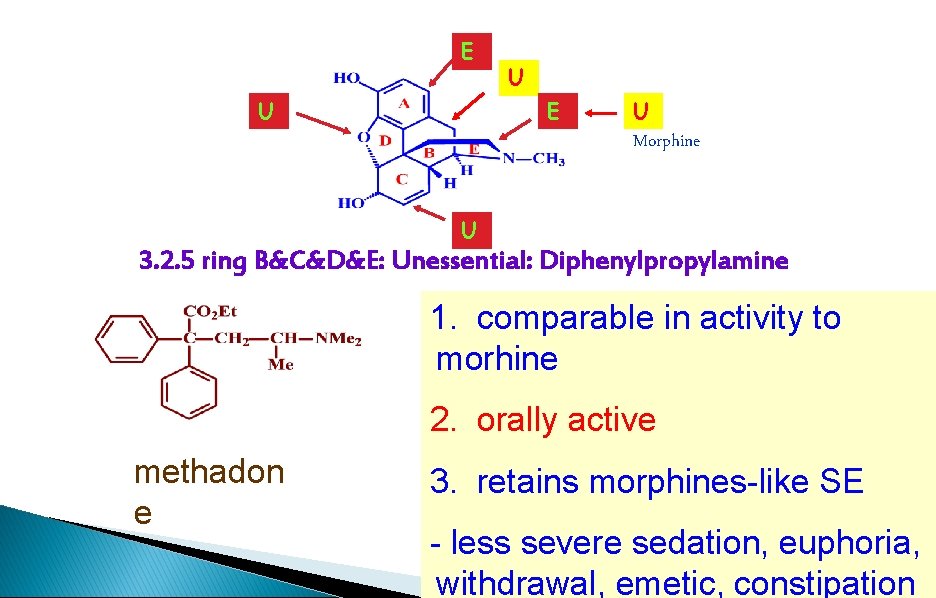
E U U E U Morphine U 3. 2. 5 ring B&C&D&E: Unessential: Diphenylpropylamine 1. comparable in activity to morhine 2. orally active methadon e 3. retains morphines-like SE - less severe sedation, euphoria, withdrawal, emetic, constipation

Morphine Analogues - Methadone � How its related: ◦ Rings B, C, D, E opened � Activity ◦ < Morphine � Used to: ◦ With addicts off heroine or morphine � Advantages: ◦ Can be given orally ◦ Less severe side effects � Marketed as Dolophine®, Amidone®, Methadose®

Morphine analogues - Naltrexone � How it’s related: ◦ Cyclopropylmethylene added to morphine � Activity: ◦ None? ! � Morphine antagonists � Used to treat: ◦ Morphine overdose ◦ Heroin addicts post-rehab Naltrexone � Advantages: ◦ No side effects � Marketed Vivitrol as: Revia, Depade, Nalorphine

Morphinans: 1. ring D is not essential 2. more active, loger acting but more SE than morphine: Levorphanol: orally active 3. Morphinans and Morphine: reacting with the same receptors in the same way. Benzomorphans: 1. ring C, D are not essential 2. analgesia and addiction are not necessarily coexistent 3. reasonable analgesic activity, less addictive liability, less tolerance simpler to synthesize 4. phenazocene, pentazocine 5. hallucinogenic effect

4 -Phenylpiperidine 1. ring B, C, D are not essential 2. retain SE: addition, respiratory depression 3. faster acting, shorter duration of action 4. interact with the analgesic receptors in a different manner to morphine 5. meperidine (pethidine), fentanyl, carfentanil, lofentanil, sufentanyl, alfentanil, remifentanil Diphenylpropylamine: methadone 1. ring B, C, D, E are not essential 2. comparable in activity to morhine 3. orally active 4. retains morphines-like SE: - less severe sedation, euphoria, withdrawal, emetic, constipation 5. a substitute for morphine or heroin

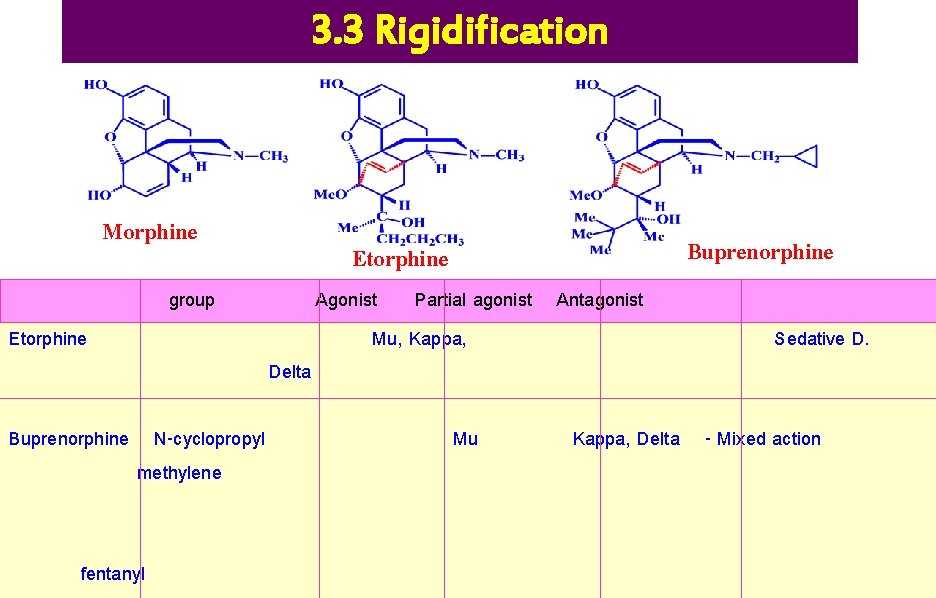
3. 3 Rigidification Morphine group Etorphine Buprenorphine N-cyclopropyl methylene fentanyl Buprenorphine Etorphine Delta Agonist Partial agonist Antagonist Mu, Kappa, Mu Sedative D. Kappa, Delta - Mixed action

Opioids receptors. 1. Mu: Mu 1: Mu 2: . 2. Kappa: , Sedation -Morphine -Nalorprine -Pentazocine -Buprenorphine 3. Delta: + ? Non-opioids receptors Sigma: + Psychotomimetic: Hallucinogenic effect

Opioid Receptors � Receptor-binding motif: ◦ Phenol OH ◦ Aromatic ring ◦ Amine group

An ideal analgesic agent? Orally active Mu: antagonist Kappa: agonist Sigma: No activity

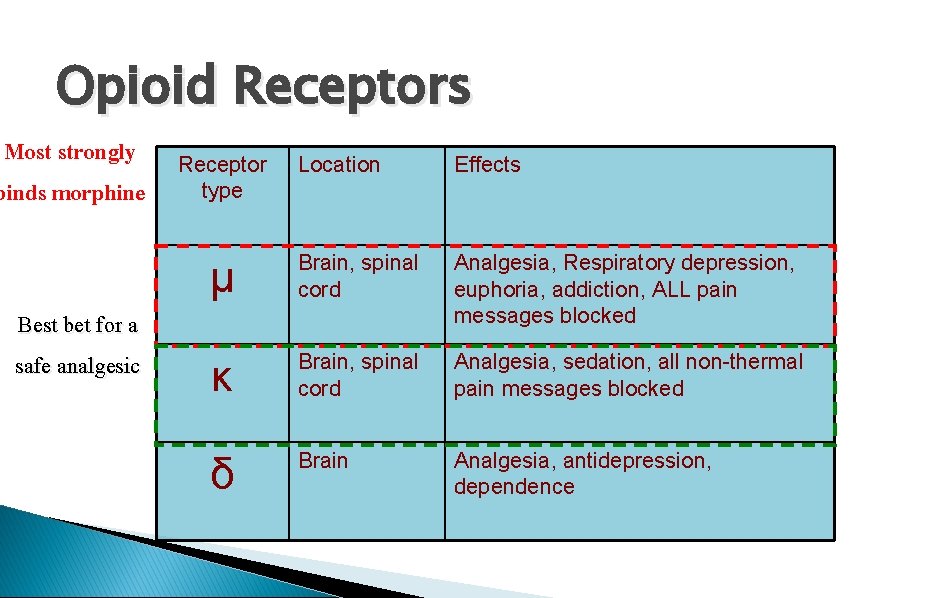
Opioid Receptors Most strongly binds morphine Best bet for a safe analgesic Receptor type Location Effects μ Brain, spinal cord Analgesia, Respiratory depression, euphoria, addiction, ALL pain messages blocked κ Brain, spinal cord Analgesia, sedation, all non-thermal pain messages blocked δ Brain Analgesia, antidepression, dependence

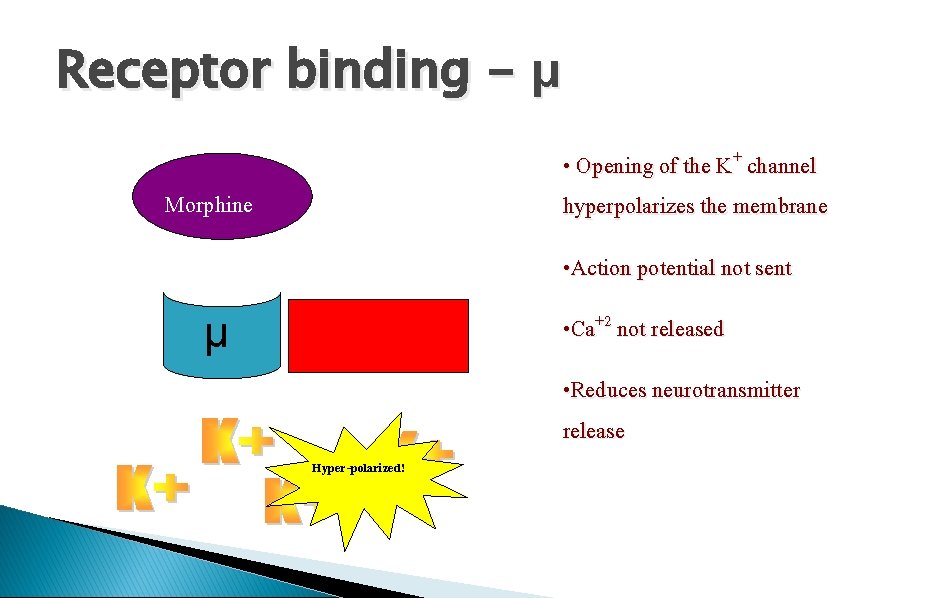
Receptor binding - μ • Opening of the K+ channel hyperpolarizes the membrane Morphine • Action potential not sent • Ca+2 not released μ • Reduces neurotransmitter release Hyper-polarized!

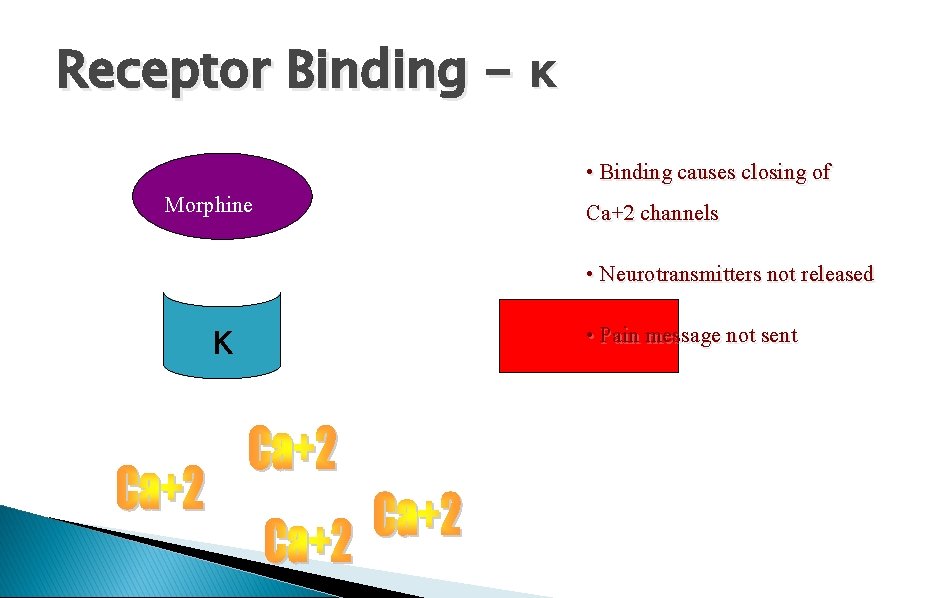
Receptor Binding - κ Morphine • Binding causes closing of Ca+2 channels • Neurotransmitters not released κ • Pain message not sent

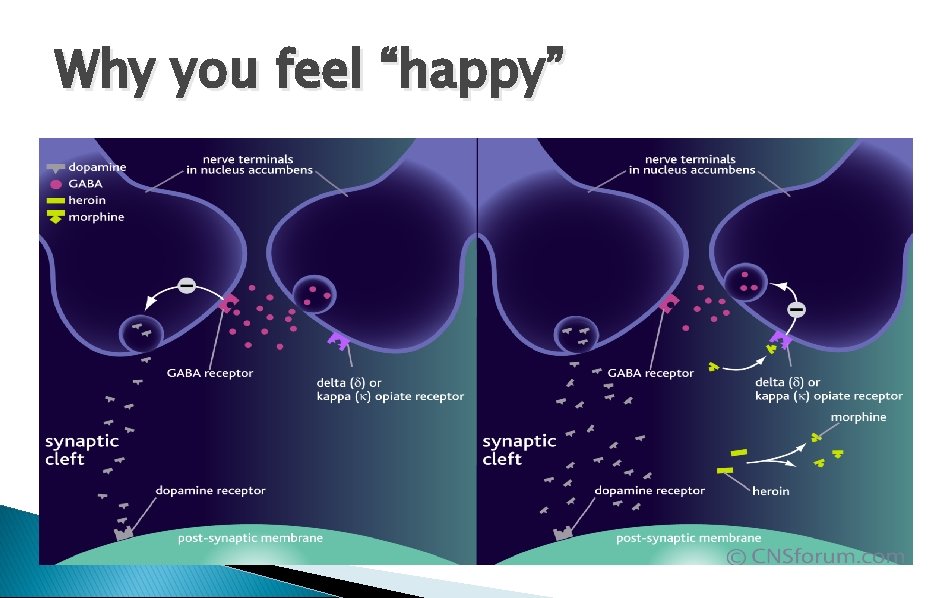
Why you feel “happy”

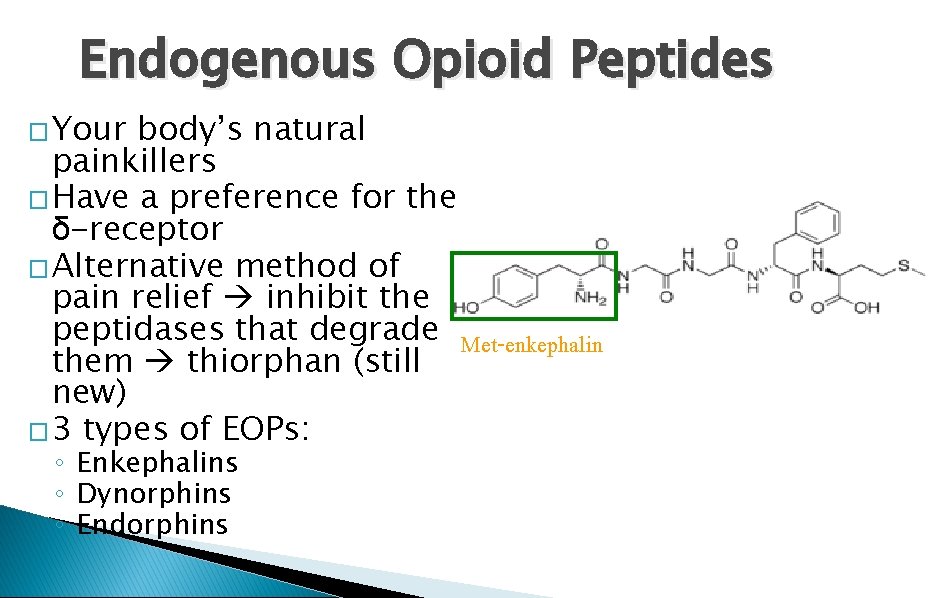
Endogenous Opioid Peptides � Your body’s natural painkillers � Have a preference for the δ-receptor � Alternative method of pain relief inhibit the peptidases that degrade Met-enkephalin them thiorphan (still new) � 3 types of EOPs: ◦ Enkephalins ◦ Dynorphins ◦ Endorphins

Partial Kappa Agonist

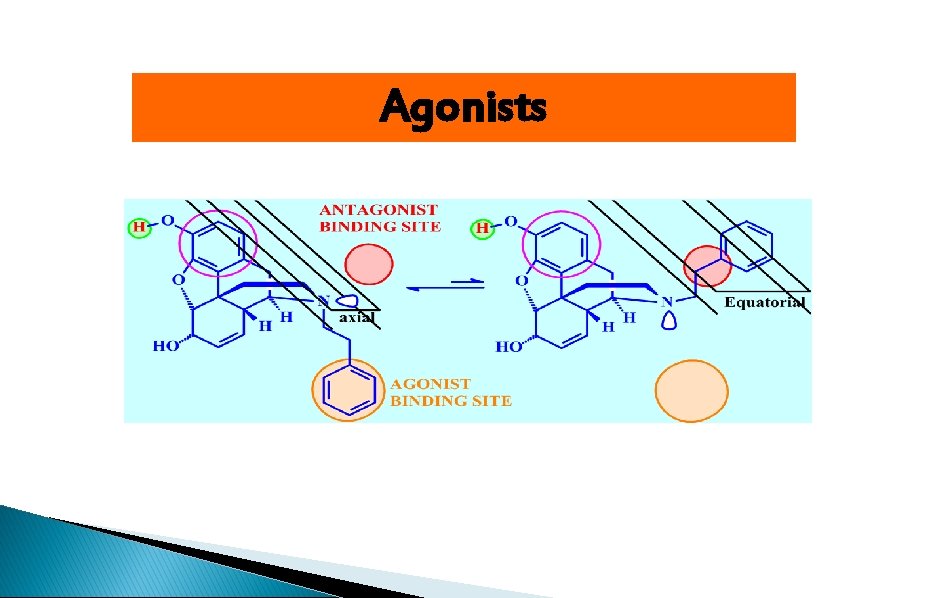
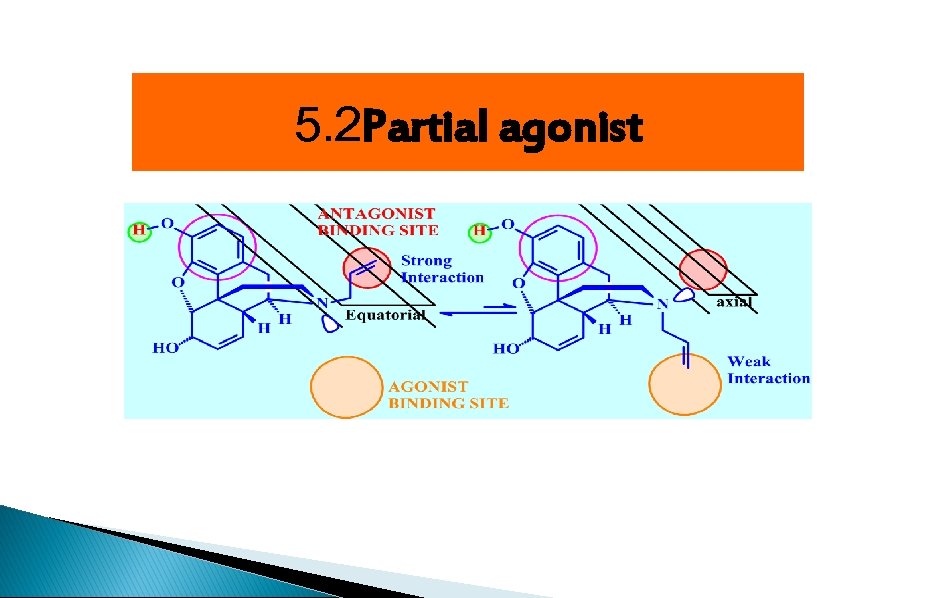
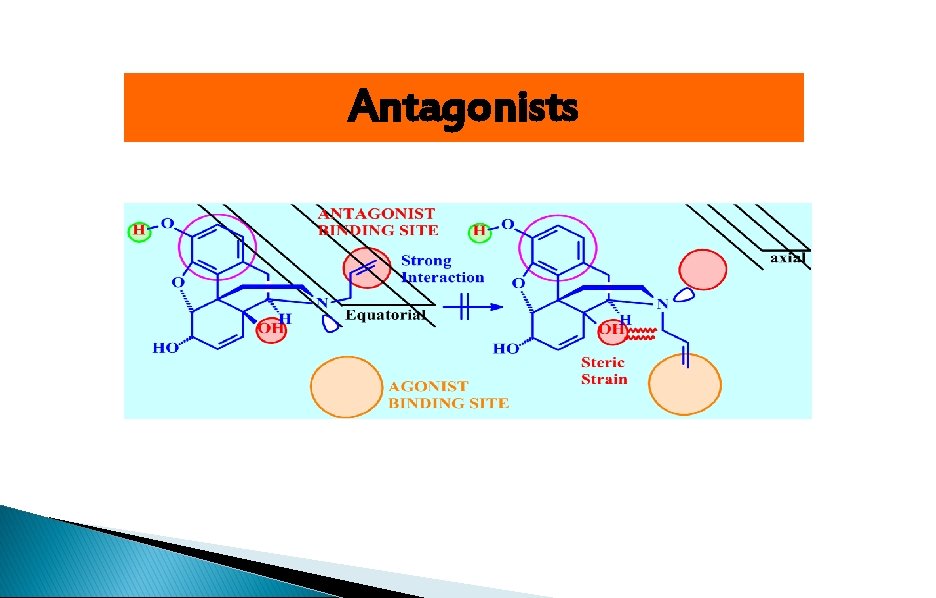
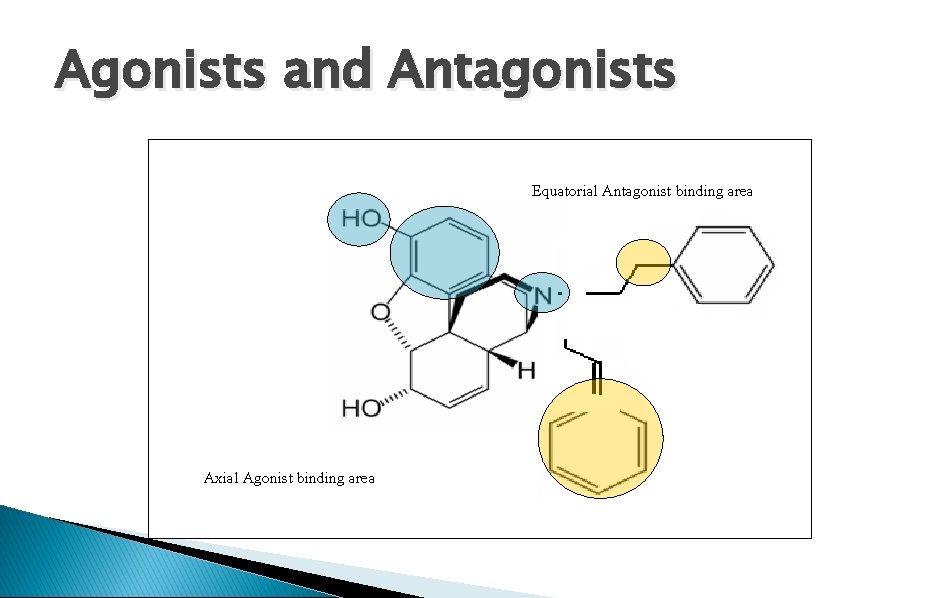
Receptor binding sites 1)Essential binding sites: center phenolic-OH, Ar, ionized N 2 (Agonist binding sites (Hydrophobic( 3 (Antagonist binding sites (Hydrophobic(

Agonists

5. 2 Partial agonist

Antagonists

Agonists and Antagonists Equatorial Antagonist binding area Axial Agonist binding area

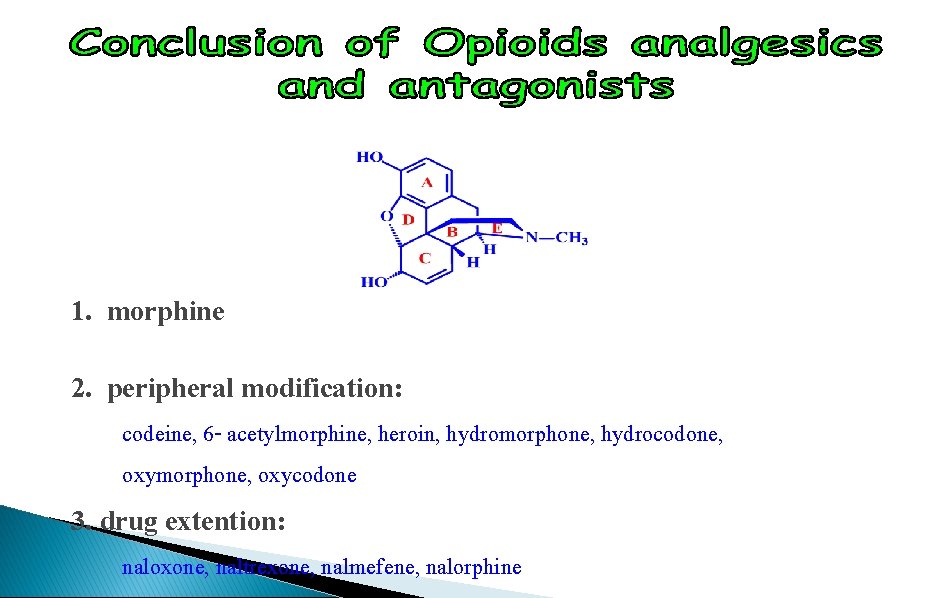
1. morphine 2. peripheral modification: codeine, 6 - acetylmorphine, heroin, hydromorphone, hydrocodone, oxymorphone, oxycodone 3. drug extention: naloxone, naltrexone, nalmefene, nalorphine

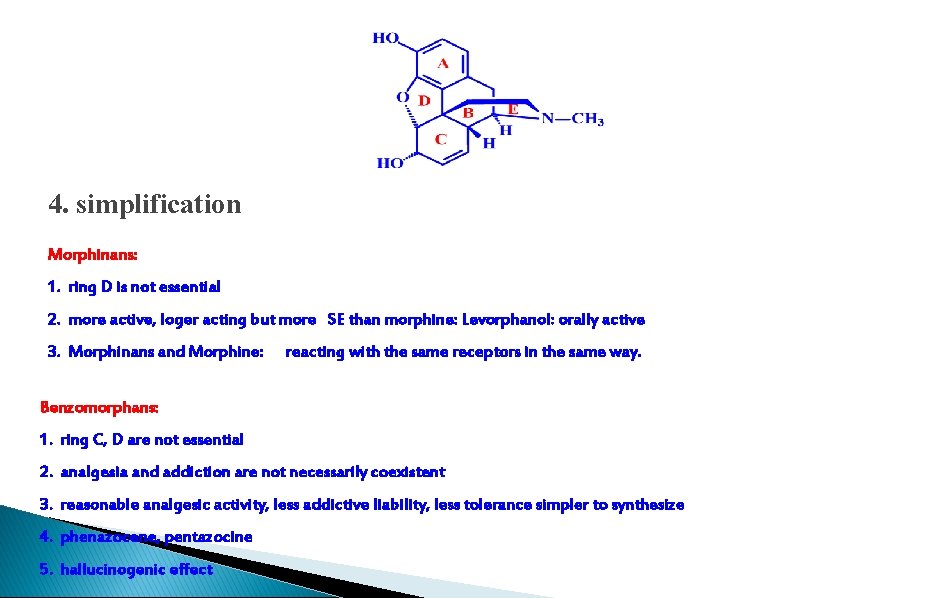
4. simplification Morphinans: 1. ring D is not essential 2. more active, loger acting but more SE than morphine: Levorphanol: orally active 3. Morphinans and Morphine: reacting with the same receptors in the same way. Benzomorphans: 1. ring C, D are not essential 2. analgesia and addiction are not necessarily coexistent 3. reasonable analgesic activity, less addictive liability, less tolerance simpler to synthesize 4. phenazocene, pentazocine 5. hallucinogenic effect

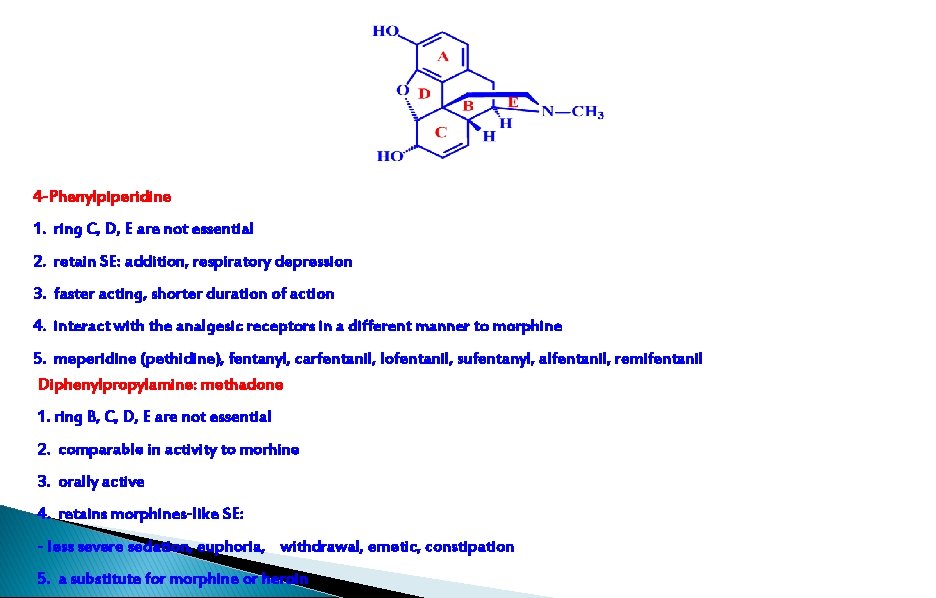
4 -Phenylpiperidine 1. ring C, D, E are not essential 2. retain SE: addition, respiratory depression 3. faster acting, shorter duration of action 4. interact with the analgesic receptors in a different manner to morphine 5. meperidine (pethidine), fentanyl, carfentanil, lofentanil, sufentanyl, alfentanil, remifentanil Diphenylpropylamine: methadone 1. ring B, C, D, E are not essential 2. comparable in activity to morhine 3. orally active 4. retains morphines-like SE: - less severe sedation, euphoria, withdrawal, emetic, constipation 5. a substitute for morphine or heroin

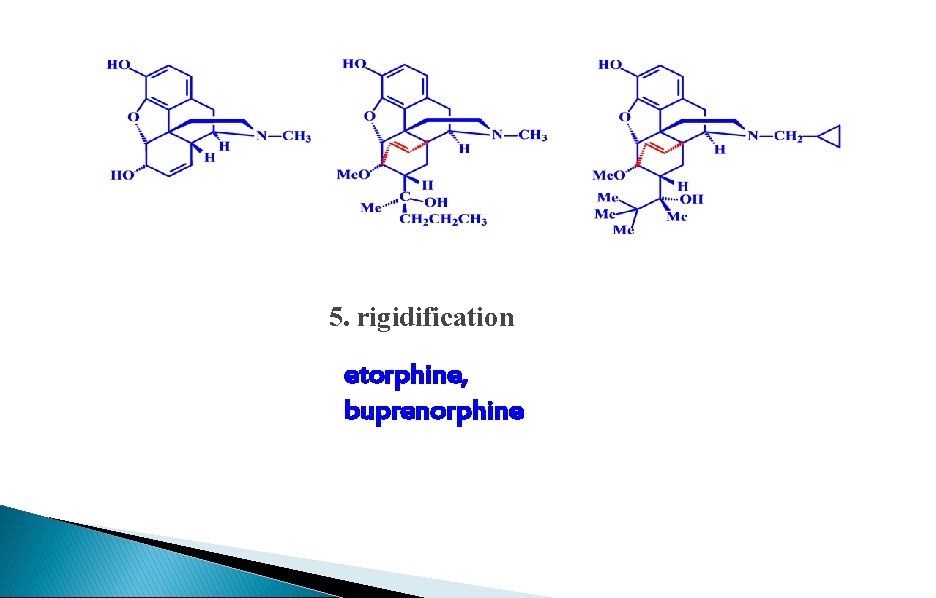
5. rigidification etorphine, buprenorphine

SIDE NOTE: � Other factors important to receptor binding: ◦ Stereochemistry �Enantiomers of many of the analogues were tested for analgesic activity. Overall, they didn’t have any. ◦ Rigidification �Used to maintain active formation and eliminate alternative conformations �Increases selectivity for receptors

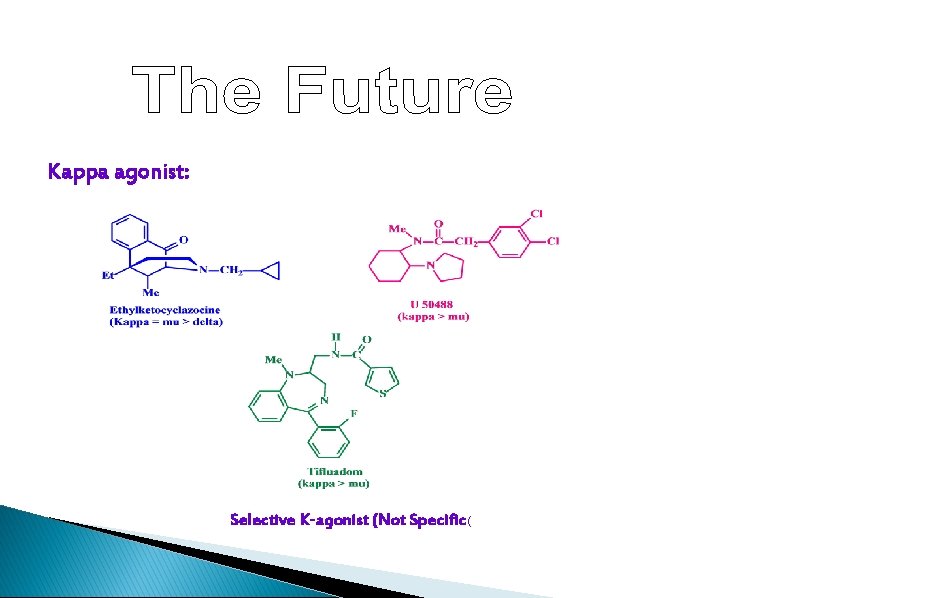
Kappa agonist: Selective K-agonist (Not Specific(

The Future � Find an agonist that solely binds to the κreceptor � Explore the μ-receptor subtypes further to see if any of them don’t cause harmful side effects � Peripheral opiate receptors – avoid BBB obstacle � Block postsynaptic receptors involved in the transmission of a pain signal � GABA � Agonists for the cannabinoid receptor

2. Selective Mu 1 agonist 3. Peripheral opiate receptor agonist

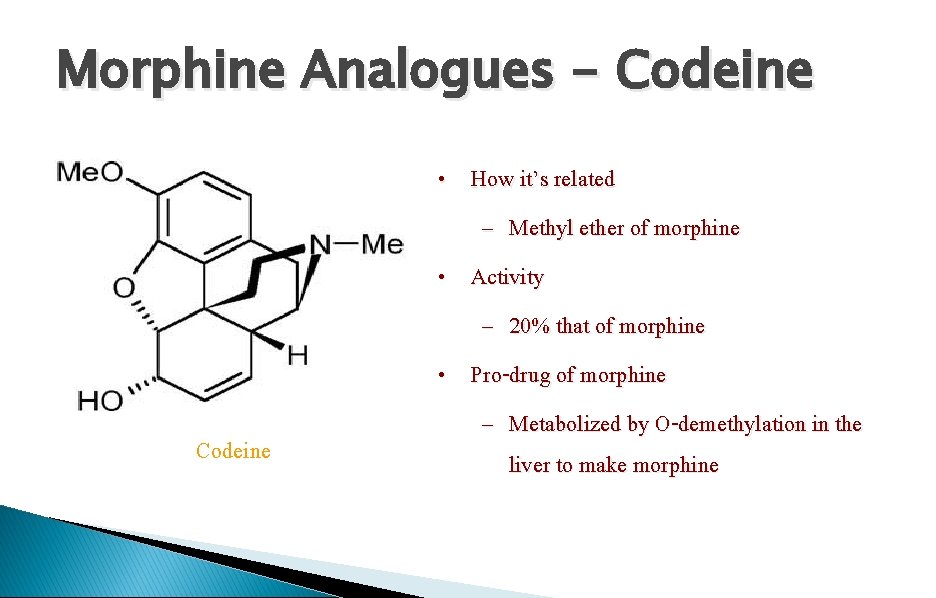
Morphine Analogues - Codeine • How it’s related – Methyl ether of morphine • Activity – 20% that of morphine • Pro-drug of morphine – Metabolized by O-demethylation in the liver to make morphine

Morphine Analogues - Codeine � Treats: ◦ Moderate pain ◦ Coughs ◦ diarrhea � Marketed as: ◦ Tylenol® with Codeine ◦ Hydrocodone ◦ Vicodin® (with Thebaine)

Morphine Analogues - Heroine � How it’s related: ◦ 3, 6 -diacetyl ester of morphine � Activity: ◦ 2 x that of morphine Heroine � Polar groups are hidden, making it easy to cross BBB. � Treats: ◦ Pain in terminally ill patients � Side effects ◦ Euphoria, addiction, tolerance � Marketed as: ◦ Heroin, “dope”

Morphine Analogues - Heroine 6 -acetylmorphine � How it’s related: ◦ 6 -acetyl of morphine � Activity ◦ 4 x that of morphine! � Polarity 6 -acetylmorphine decreased, but phenol is ready to bind receptor � Side effects: Very potent!! ◦ Euphoria, addiction, etc. � Marketed as: ◦ NOTHING! It’s banned from production in many countries

http: //www. opioids. com Patrick, G. L. 1995. An introduction to medicinal chemistry. Oxford: Oxford University Press. Wolff, M. E. 1995. Burger’s medicinal chemistry and drug discovery. vol. 1. 5 th ed. New York: John Wiley & Sons.