More reduced more H Can be more oxidized
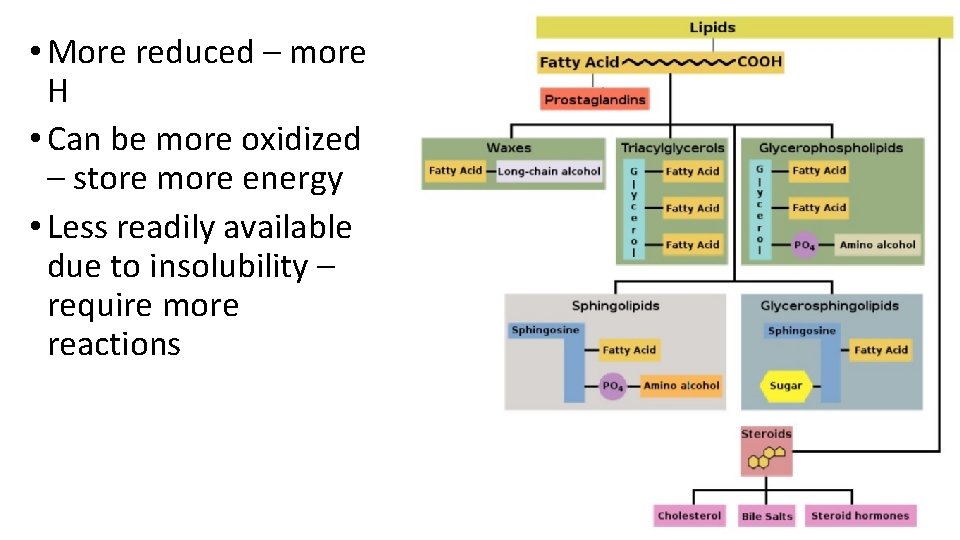
• More reduced – more H • Can be more oxidized – store more energy • Less readily available due to insolubility – require more reactions
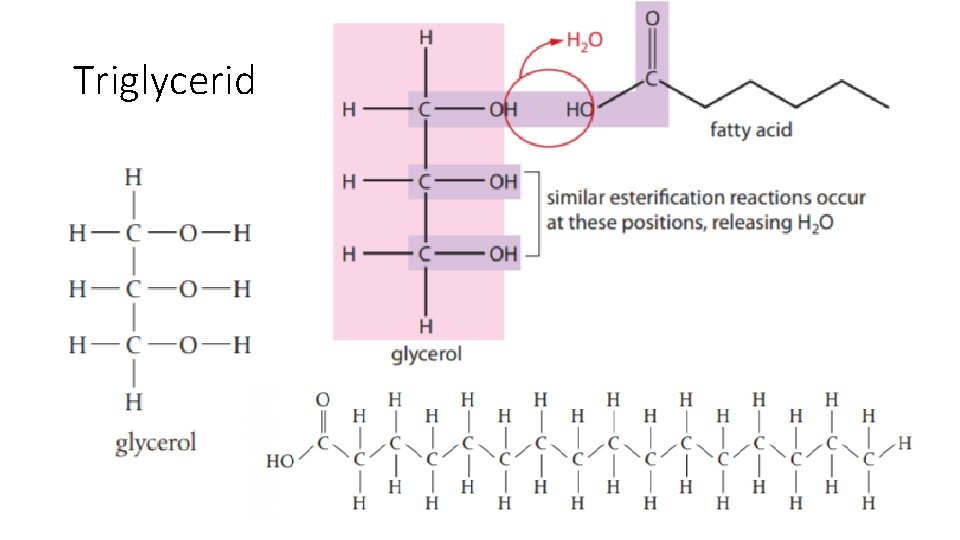
Triglycerides


Saturated • Tetrahedral • Packed closely • High London dispersion forces • High melting point

Unsaturated • One or more double C=C bonds, so one of more 120 trigonal planar • Lower melting points • Melting point increases with increase molar mass

Iodine number • Grams of iodine reacting with 100 g of fat • Measure of unsaturation (1 mol I 2 reacts with each mol of C=C) 100 g LA 1 mol LA 2 mol I 2 254 g I 2 We would expect C 17 H 35 COOH if totally saturated. 280 g LA 1 mol I 2 4 Hydrogens fewer – 2 H removed for each C=C Therefore, 2 C=C’s 181 g I 2 Iodine number 181 1 mol Linoleic acid 2 mol of C=C 2 mol I 2
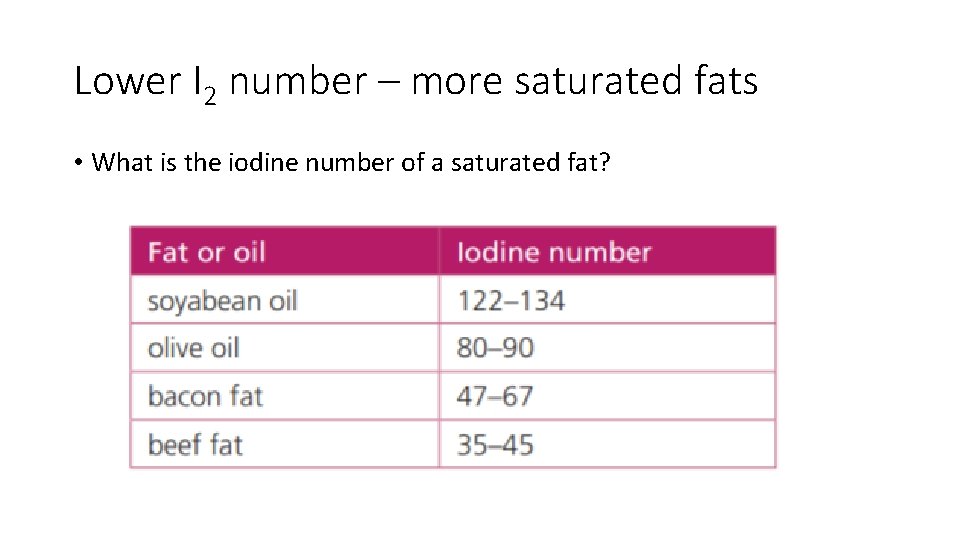
Lower I 2 number – more saturated fats • What is the iodine number of a saturated fat?
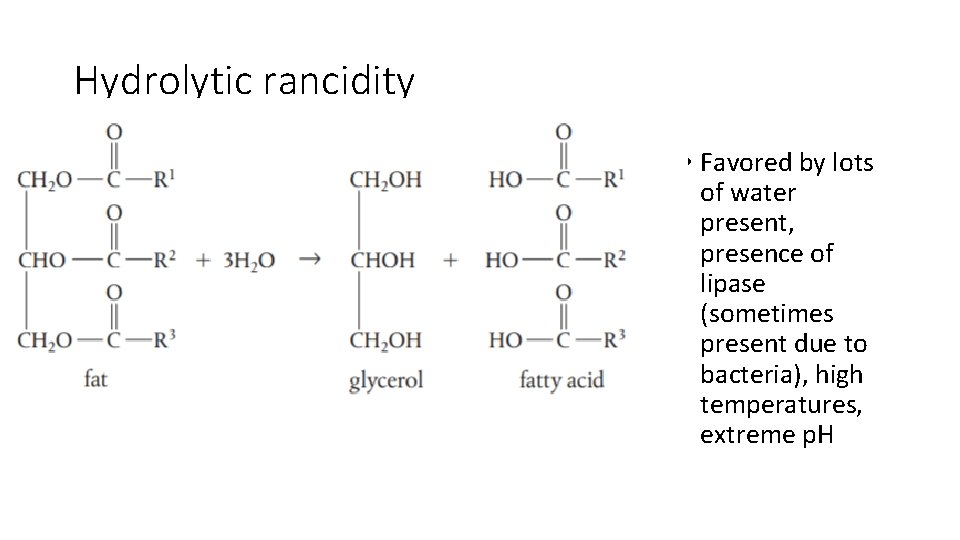
Hydrolytic rancidity • Favored by lots of water present, presence of lipase (sometimes present due to bacteria), high temperatures, extreme p. H

Oxidative rancidity • At C=C in unsaturated fats • Accelerated by light, enzymes, or metal ions
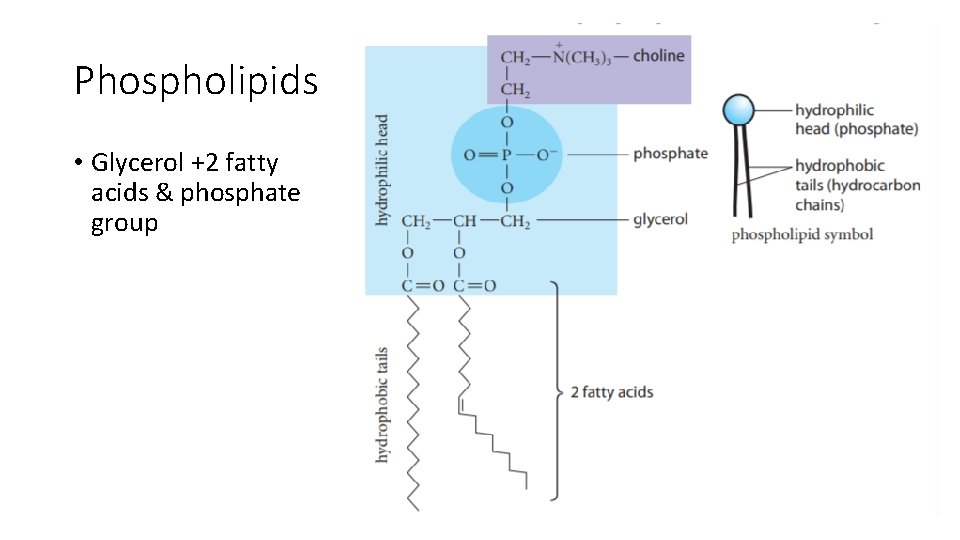
Phospholipids • Glycerol +2 fatty acids & phosphate group

Saponification

• Saponification can occur in oil paintings over time, causing visible damage and deformation. Oil paints are composed of pigment molecules suspended in an oil binding medium. • Heavy metal salts are often used as pigment molecules, such as in lead white, red lead, and zinc white. If those heavy metal salts react with free fatty acids in the oil medium, metal soaps may form in a paint layer that can then migrate outward to the painting's surface

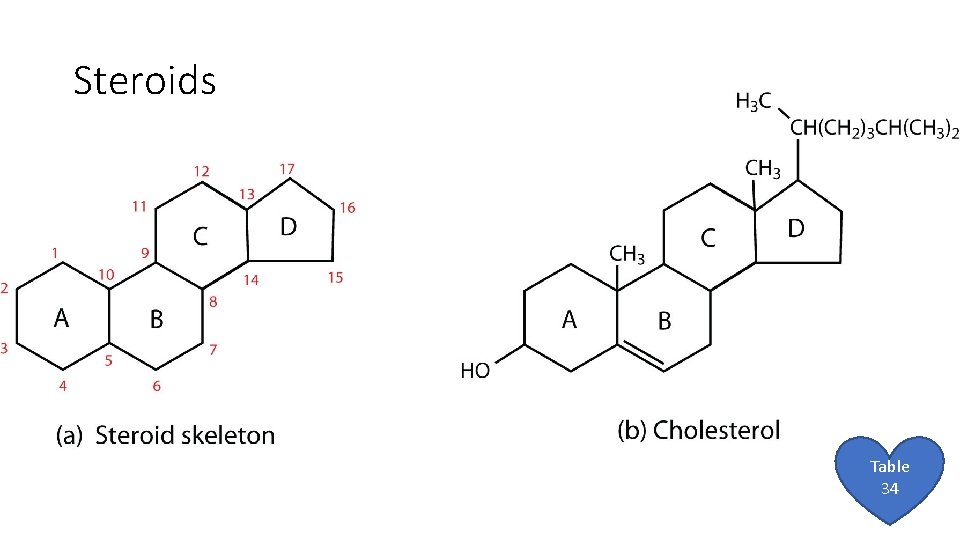
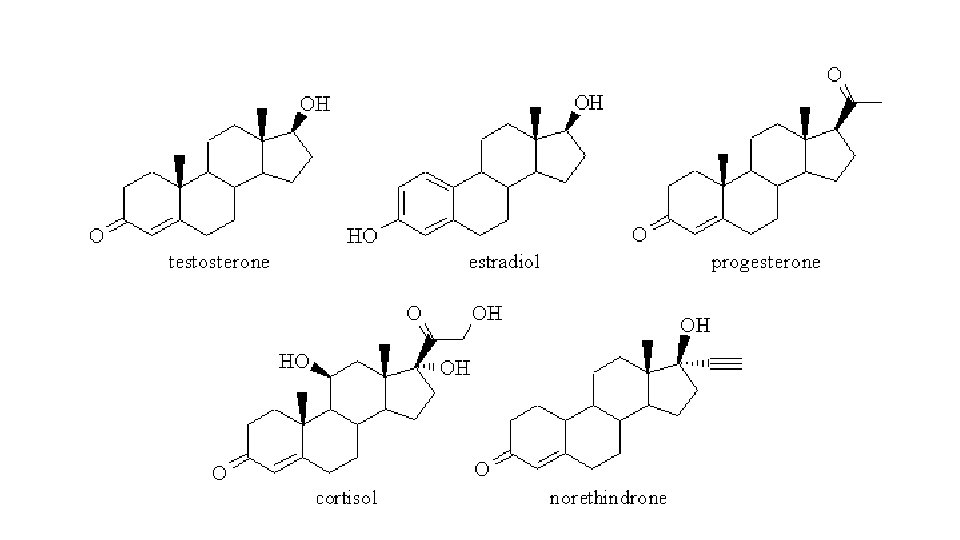
Steroids Table 34

- Slides: 17