More Functional Groups Carbonyl Substituents aldehydes ketones carboxylic

More Functional Groups: Carbonyl Substituents (aldehydes, ketones, carboxylic acids) & Esters Learning Goals: I will understand what the various functional groups are, and how to name and draw aldehydes, ketones, carboxylic acids & esters Copyright © Houghton Mifflin Company. All rights reserved. Intro 14 | 1

Derivatives of Hydrocarbons What we already know: 1) Alkyl halides (haloalkanes) 2) Alcohols 3) Ethers What we have to cover: 1) Aldehydes These 3 are carbonyl 2) Ketones substituents 3) Carboxylic acids 4) Esters 5) Amines 6) Amides Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 2

Carbonyl Substituents • Carbonyl substituents are groups that have a C=O bond associated with them • Three different types: i) Aldehydes ii) Ketones iii) Carboxylic acids Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 3

Naming Aldehydes • The =O is at the end of the molecule • Ending is ‘al’ • start numbering at the carbonyl in the aldehyde group. You leave out the -1 -al part, since this is always carbon # 1 • • Hierarchy of Importance (lowest #) Carboxylic acids Aldehydes Ketones Alcohols Alkenes Alkynes Halogens and branches– alphabetical Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 4

Naming Aldehydes: Example Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 5
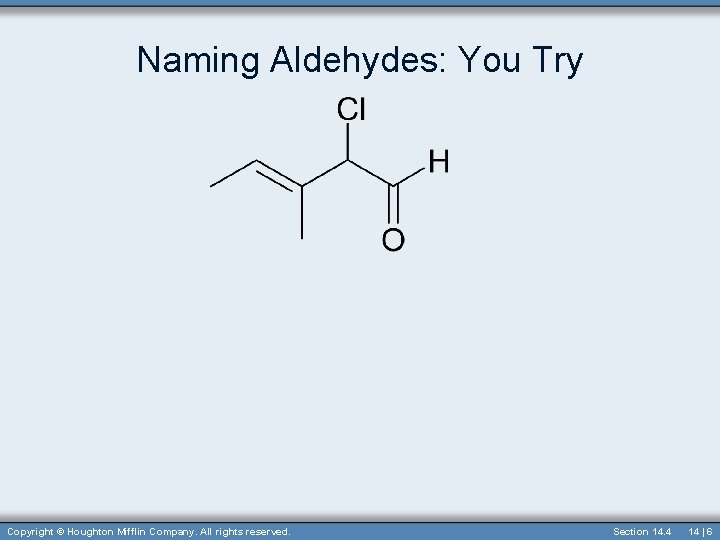
Naming Aldehydes: You Try Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 6
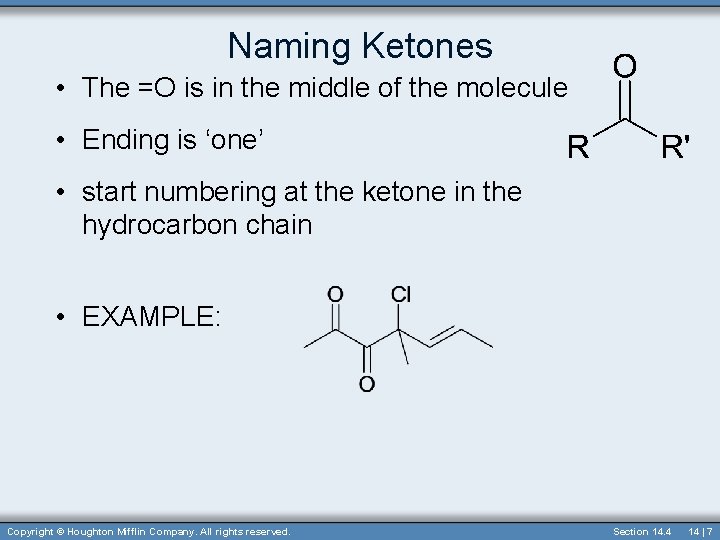
Naming Ketones • The =O is in the middle of the molecule • Ending is ‘one’ • start numbering at the ketone in the hydrocarbon chain • EXAMPLE: Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 7
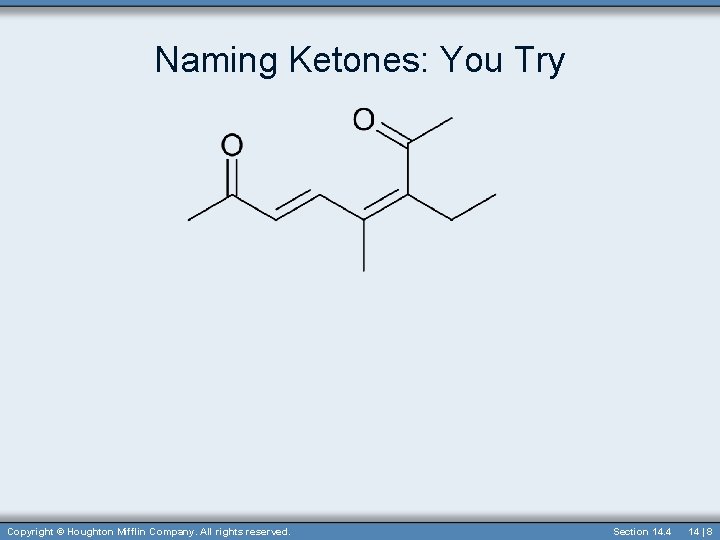
Naming Ketones: You Try Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 8
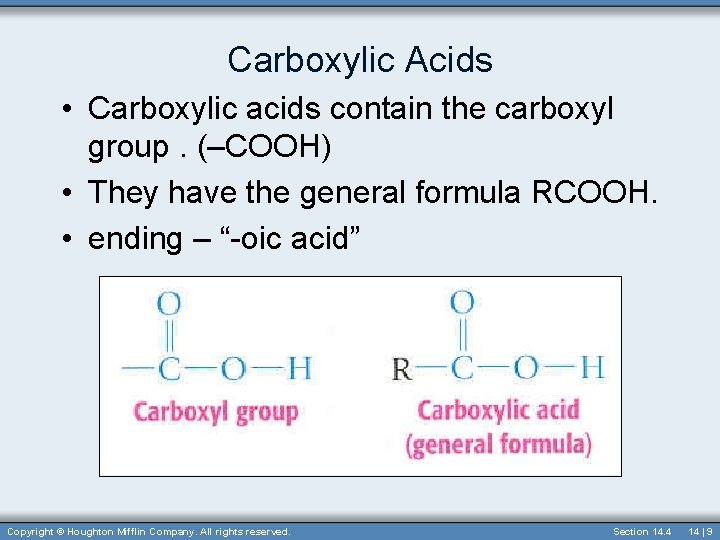
Carboxylic Acids • Carboxylic acids contain the carboxyl group. (–COOH) • They have the general formula RCOOH. • ending – “-oic acid” Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 9
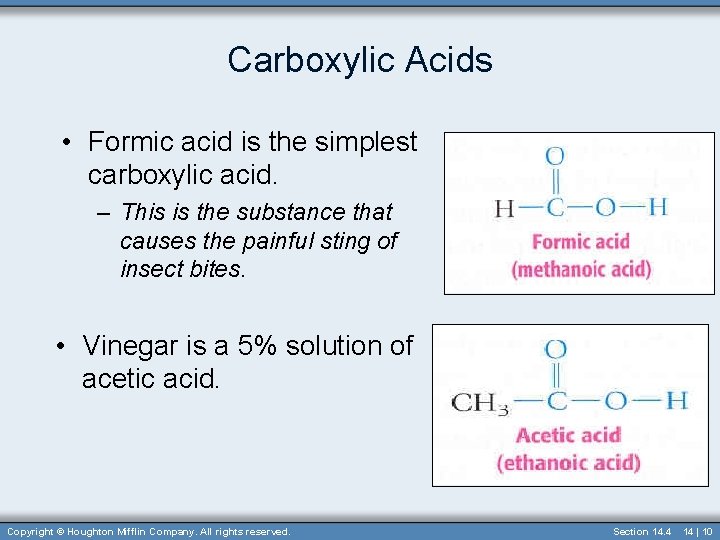
Carboxylic Acids • Formic acid is the simplest carboxylic acid. – This is the substance that causes the painful sting of insect bites. • Vinegar is a 5% solution of acetic acid. Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 10
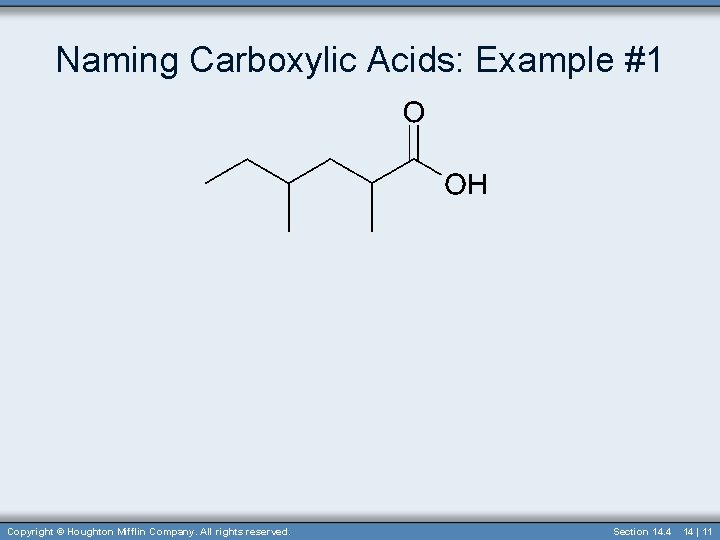
Naming Carboxylic Acids: Example #1 Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 11
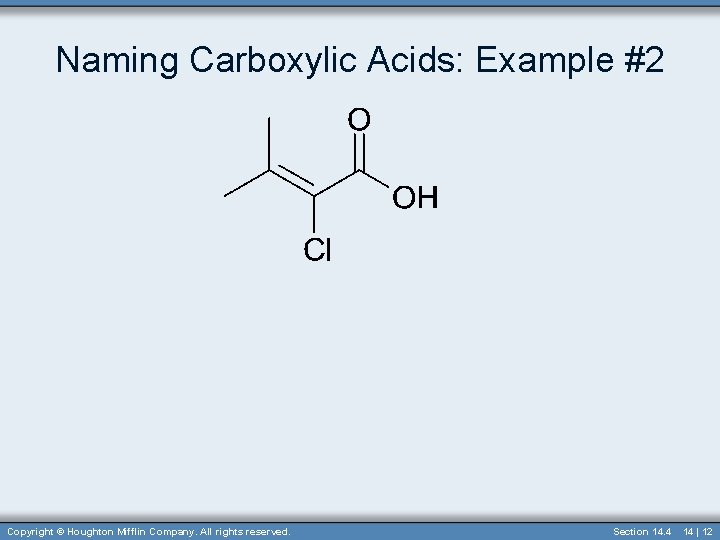
Naming Carboxylic Acids: Example #2 Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 12
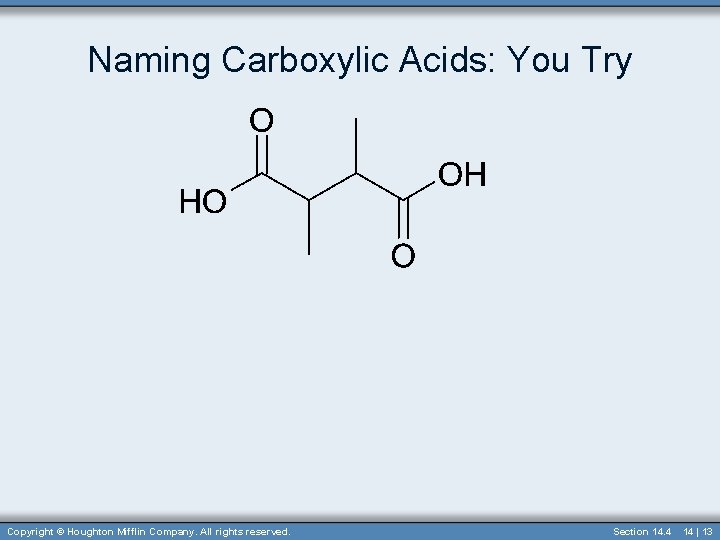
Naming Carboxylic Acids: You Try Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 13

Esters • Ester – a compound that has the following general formula RCOOR’ • In the general formula for an ester the R and R’ can be any alkyl group. • Although R and R’ can be identical, they are usually different. • Contrary to amines, most esters have pleasant odors. • Many flowers and ripe fruits have fragrances and tastes due to one or more esters. Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 14
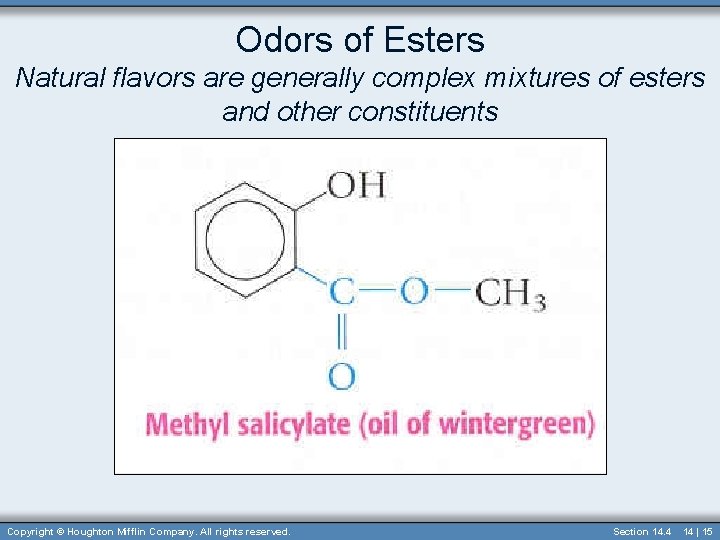
Odors of Esters Natural flavors are generally complex mixtures of esters and other constituents Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 15

Formation of an Ester • Ester formation – the reaction of a carboxylic acid an alcohol give an ester and water – Heat is required and sulfuric acid is a catalyst. • Note, in this reaction that the –OH from the carboxylic acid unites with the H from the alcohol to form H 2 O. • The remaining two fragments bond together to form the ester. Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 16

Writing an Equation for Ester Formation An Example • Complete the equation for the sulfuric acid-catalyzed reaction between acetic acid and ethanol. Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 17
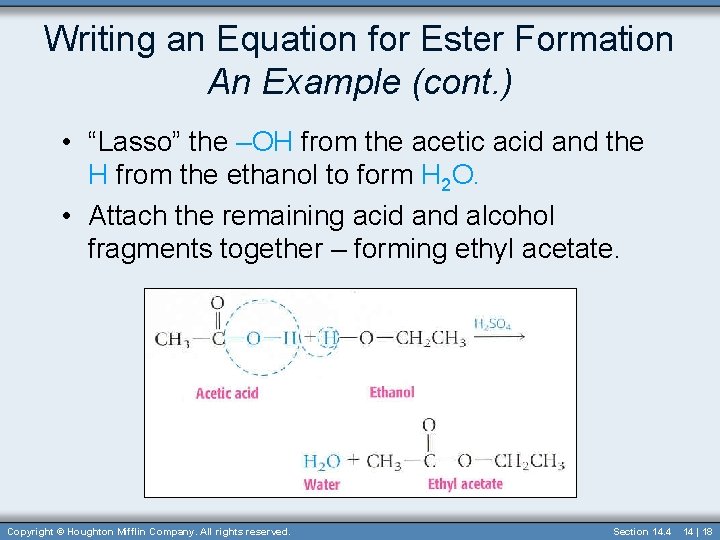
Writing an Equation for Ester Formation An Example (cont. ) • “Lasso” the –OH from the acetic acid and the H from the ethanol to form H 2 O. • Attach the remaining acid and alcohol fragments together – forming ethyl acetate. Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 18
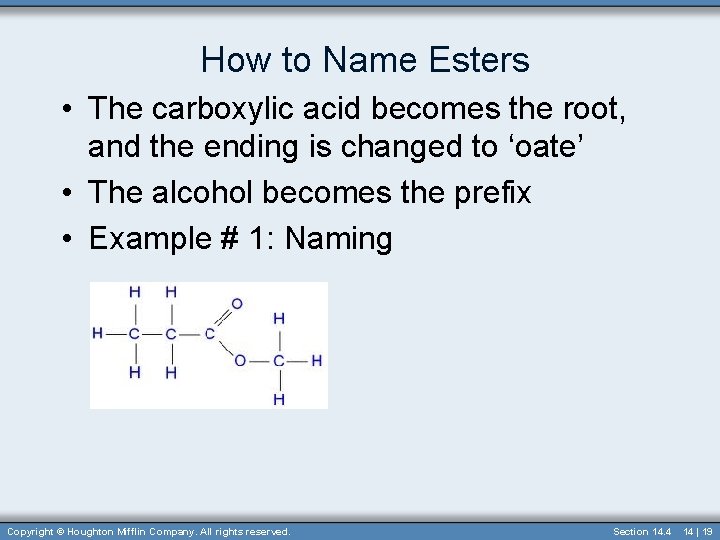
How to Name Esters • The carboxylic acid becomes the root, and the ending is changed to ‘oate’ • The alcohol becomes the prefix • Example # 1: Naming Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 19
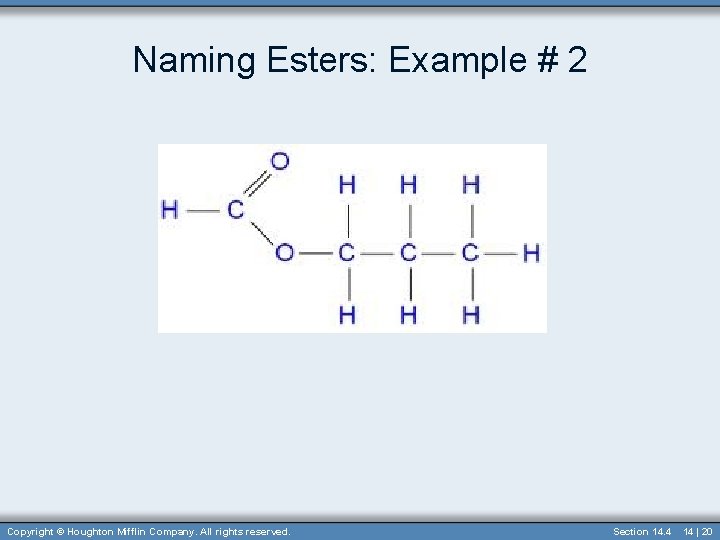
Naming Esters: Example # 2 Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 20
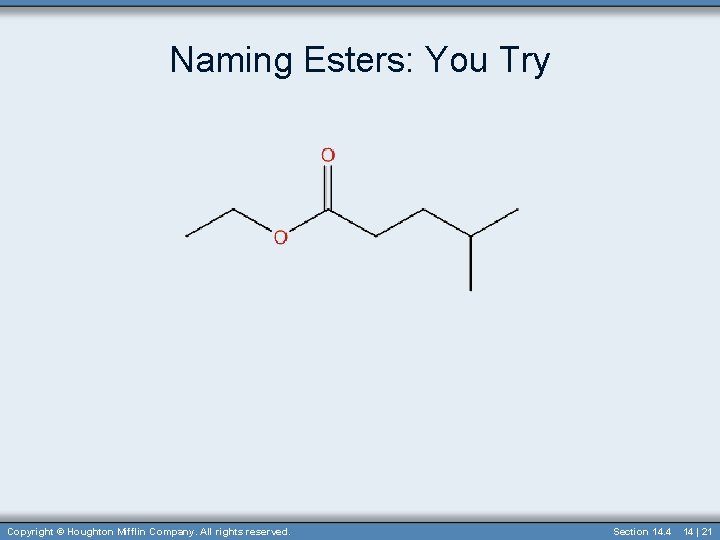
Naming Esters: You Try Copyright © Houghton Mifflin Company. All rights reserved. Section 14. 4 14 | 21

More Functional Groups: Carbonyl Substituents (aldehydes, ketones, carboxylic acids) & Esters Learning Goals: I will understand what the various functional groups are, and how to name and draw aldehydes, ketones, carboxylic acids & esters Copyright © Houghton Mifflin Company. All rights reserved. Intro 14 | 22
- Slides: 22