More biologically active amines More biologically active amines
…More biologically active amines
…More biologically active amines

Basicity of amines n Amines are slightly basic. This because they have a lone pair of electrons to donate to a proton. This same feature makes them nucleophiles. n Typical amines have Kb values = 10 -3 to 10 -4

…Basicity of amines n However, instead of measuring an amine’s basicity using the above equilibrium, chemists usually refer to the acidity of the conjugate acid of the amine. The weaker the conjugate acid, the stronger the base strength of the amine. n Typical amines have Ka values (of their conjugate acids) of 10 -10 to 10 -11 (p. Ka values of 10 to 11. )

p. Ka values of conjugate acids of amines CH 3 NH 2 CH 3 CH 2 NH 2 10. 7 10. 8 (CH 3)2 NH (CH 3 CH 2)2 NH 10. 7 10. 5 (CH 3)3 N (CH 3 CH 2)3 N 9. 8 11. 0 All are about the same value. The same substituent effects that stabilize carbocations that are more highly substituted by alkyl groups are offset by diminished stabilization by solvent due to crowding.
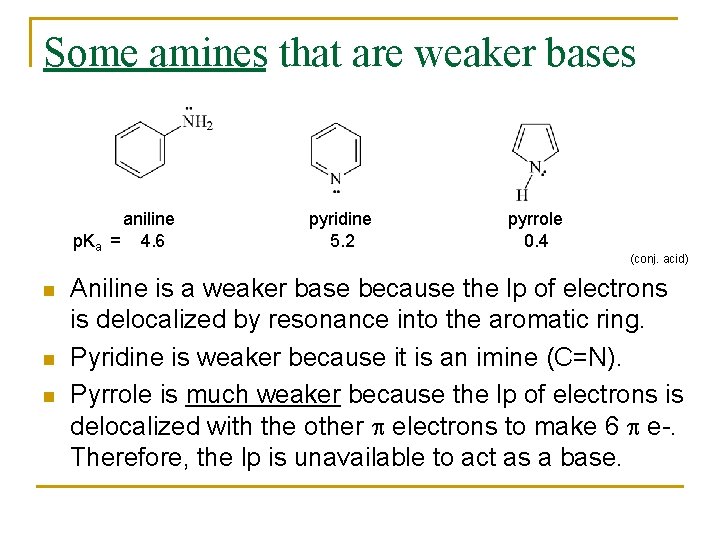
Some amines that are weaker bases aniline p. Ka = 4. 6 n n n pyridine 5. 2 pyrrole 0. 4 (conj. acid) Aniline is a weaker base because the lp of electrons is delocalized by resonance into the aromatic ring. Pyridine is weaker because it is an imine (C=N). Pyrrole is much weaker because the lp of electrons is delocalized with the other p electrons to make 6 p e-. Therefore, the lp is unavailable to act as a base.
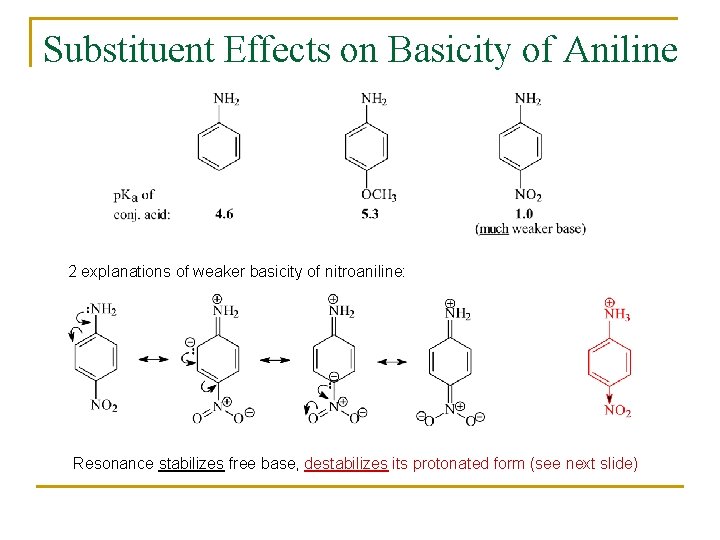
Substituent Effects on Basicity of Aniline 2 explanations of weaker basicity of nitroaniline: Resonance stabilizes free base, destabilizes its protonated form (see next slide)

Substituent Effects on Basicity of Anilines

An amine that is a stronger base n n There is one type of amine that is a stronger base: guanidine p. Ka = 13. 6 (conj. acid) Guanidine is a strong base because its conjugate acid is stabilized by resonance: A guanidine group is part of the structure of the amino acid arginine.

Amines are Protonated at Physiological p. H n Recall the Henderson-Hasselbach equation: n Consider the neurotransmitter dopamine, a typical amine (having a p. Ka of its conjugate acid = 10. 6) in a living cell (buffered at p. H = 7. 3): n That is, the concentration of the protonated amine is 2000 x that of the neutral amine! Typical amines are >99. 9% protonated at physiological p. H.
- Slides: 10