Monoclonal Antibodies in the Treatment of Migraine November

Monoclonal Antibodies in the Treatment of Migraine November 2, 2018 Denise Taylor, DO Athens Neurological Associates Athens, Georgia

Objectives �Recognize undiagnosed/undertreated migraine patients �Determine which patients meet criteria for treatment with preventative therapy �Identify appropriate cases for use of monoclonal antibody therapy

Disclosure �Speaker’s Bureau for UCB - 2018

Migraine Update �Many epidemiological studies have documented high prevalence and socioeconomic and personal impacts. �In the Global Burden of Disease Study 2010 (GBD 2010), it was ranked as the third most prevalent disorder in the world. �In GBD 2015, it was ranked the third-highest cause of disability worldwide in both males and females <50 years of age.

Migraine Pathophysiology �In migraine, the pain mechanisms are peripheral, driven by meningeal neurogenic inflammation and vasodilation. �This activates peripheral trigeminal nociceptive afferents to transmit pain signals to the central nervous system. �This leads to peripheral and central neuronal sensitization.

The International Classification of Headache Disorders, 3 rd edition https: //www. ichd-3. org/ The Primary Headaches 1. Migraine 2. Tension-type headache 3. Trigeminal autonomic cephalgias 4. Other primary headache disorders The Secondary Headaches 5. Headache attributed to trauma or injury to the head and/or neck 6. Headache attributed to cranial and/or cervical vascular disorder 7. Headache attributed to non-vascular intracranial disorder 8. Headache attributed to a substance or its withdrawal 9. Headache attributed to infection 10. Headache attributed to disorder of homoeostasis 11. Headache or facial pain attributed to disorder of the cranium, neck, eyes, ears, nose, sinuses, teeth, mouth or other facial or cervical structure 12. Headache attributed to psychiatric disorder Painful Cranial Neuropathies, Other Facial Pain and Other Headaches 13. Painful lesions of the cranial nerves and other facial pain 14. Other headache disorders

Migraine: � � � � Migraine with and without aura Typical aura with and without headache Migraine with brainstem aura Hemiplegic migraine Familial hemiplegic migraine Type 1, 2, and 3 and other loci Sporadic hemiplegic migraine Retinal migraine Chronic migraine Status migrainosus Persistent aura without infarction Migrainous infarction Migraine aura-triggered seizure Probable migraine with and without aura Episodic syndromes that may be associated with migraine ◦ ◦ ◦ Recurrent gastrointestinal disturbance Cyclical vomiting syndrome Abdominal migraine Benign paroxysmal vertigo Benign paroxysmal torticollis

The International Classification of Headache Disorders, 3 rd edition (ICHD-3) Criteria for migraine without aura � A) At least five attacks fulfilling criteria B through D � B) Headache attacks lasting 4 to 72 hours (untreated or unsuccessfully treated) � C) Headache has at least two of the following characteristics: ◦ • Unilateral location, Pulsating quality, Moderate or severe pain intensity, • ◦ Aggravation by or causing avoidance of routine physical activity � D) During headache at least one of the following: ◦ Nausea, Vomiting, or both. Photophobia and Phonophobia Criteria for migraine with aura: � A) One or more of the following fully reversible aura symptoms: ◦ Visual, Sensory, Speech Motor, or Brainstem � B) At least two of the following four characteristics: ◦ ◦ At least one aura symptom spreads gradually over ≥ 5 minutes, Each individual aura symptom lasts 5 to 60 minutes At least one aura symptom is unilateral The aura is accompanied, or followed within 60 minutes, by headache

Who Needs Preventative Treatment? � Factors to consider: frequency of headaches, interference with routine activities, migraine subtype, issues with acute medications, elevated risk for headache progression or other adverse neurological outcomes, and patient preference. � Episodic migraine (<15 headache days/month) � Chronic migraine (15 or more headache days/month). � Highly impacted patients: those with high-frequency episodic migraine, chronic migraine, or medication-overuse headache. � Highly impacted patients typically have 4 or more migraine days/month associated with significant interference in routine activities despite the use of acute treatment.

Barriers to Preventive �The American Migraine Prevalence and Treatment Prevention (AMPP) study showed that approximately 2/3 of individuals with migraine who qualify for preventive treatments do not receive them �Some people with migraine may never seek medical care or lapse from care and many diagnosed patients may not get adequate treatment, acute or preventive, at the time of diagnosis or follow-up. �Follow-up is important as the initial treatment may be suboptimal or a patient’s condition may change over time, therefore treatments may need to be adjusted to account for changes in efficacy,

Goals of Preventive Treatment �Reduce attack frequency, severity, and duration. �Improve responsiveness to treatment of acute attacks. �Improve function and reduce disability. �Prevent progression or transformation of episodic migraine to chronic migraine.

Preventive Medications Found Effective in Controlled Trials: Both on- and off-label include: ◦ ◦ ◦ antiepileptics (e. g. topiramate, divalproex) antidepressants (e. g. amitriptyline, venlafaxine) beta-blockers (e. g. propranolol, timolol, metoprolol) Botulinum toxin (e. g. onabotulinumtoxin. A) CGRP antagonist (e. g. erenumab-aooe- Aimovig) Approximately 50 -75% of patients given any of these drugs will have a 50% reduction in frequency of headaches but doses required may lead to intolerable side effects. Clinical trials suggest efficacy is often first noted at 4 weeks and can continue to increase for 3 months.

Choice of Preventive Medication �Comorbid conditions ◦ (depression/HTN/epilepsy/insomnia/obesit y) �Medication side effect profile �Medication cost Example: women of childbearing age, valproate/topiramate are problematic due to teratogenicity and are associated with increased risk of birth defects.

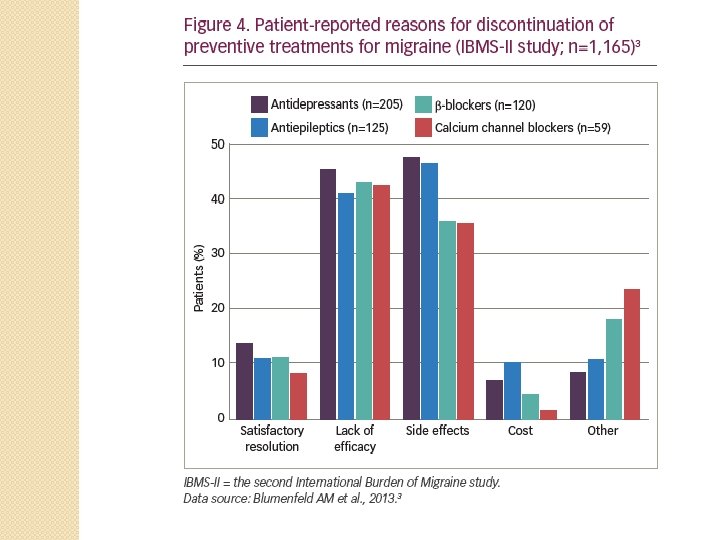
Who Discontinues Treatment? �A retrospective claims database analysis of 8, 688 patients experiencing >15 headache days/month found only 20% remained adherent (defined as prescription claims covering at least 80% of days) to preventive medication after 1 year. �Reasons for this poor adherence were assessed in the second International Burden of Migraine study 2010, in 1, 165 adults with migraine. �Discontinuation of preventive therapy was reported in 24% of respondents with episodic migraine (n=672) and 41% of patients with chronic migraine (n=493). ◦ The two most common reasons for discontinuation were a lack of efficacy and side effects

Newest Treatment Option: Calcitonin Gene-Related Peptide (CGRP)
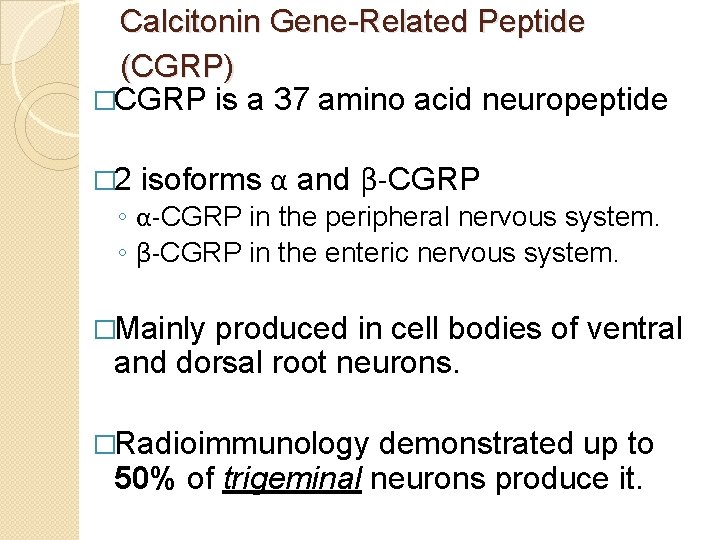
Calcitonin Gene-Related Peptide (CGRP) �CGRP is a 37 amino acid neuropeptide � 2 isoforms α and β-CGRP ◦ α-CGRP in the peripheral nervous system. ◦ β-CGRP in the enteric nervous system. �Mainly produced in cell bodies of ventral and dorsal root neurons. �Radioimmunology demonstrated up to 50% of trigeminal neurons produce it.

Physiological Role of CGRP in Migraine �CGRP is one of the most potent endogenous vasodilators with a long duration of action. �CGRP released from trigeminal terminals results in vasodilation via CGRP receptors on smooth muscle cells of the meningeal and cerebral blood vessels.
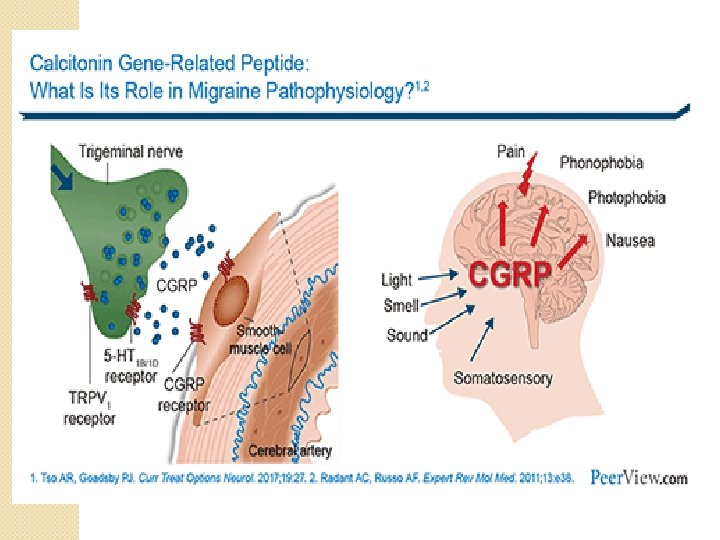

Jugular levels of CGRP increase during migraine attacks and IV CGRP administration induces migraine like headaches.

The CGRP mechanism can be antagonized by targeting either the peptide or its receptor with either a small molecule antagonist or monoclonal antibody. 1 drug against CGRP receptor: Erenumab (approved by FDA 05/2018) 3 drugs against CGRP molecule itself: Eptinezumab Fremanezumab (approved by FDA 09/2018) Galcanezumab (approved by FDA 09/2018)


-triptans and CGRP �Studies have shown that –triptans and other 5 -HT 1 receptor agonists block/suppress trigeminal activation and CGRP release. �CGRP level decrease after administration of -triptans in parallel with symptomatic relief.

Risk of Inducing Vasoconstriction by inhibiting CGRP �Major cardiovascular effects could be a concern with CGRP inhibition including medication induced HTN, counterbalancing effects of HTN drugs, inhibition of stress/ischemia induced vasodilation and impairment of cardioprotective mechanisms. �In vitro studies have shown that CGRP receptor antagonists and antibodies do not have vasoconstrictor activity.
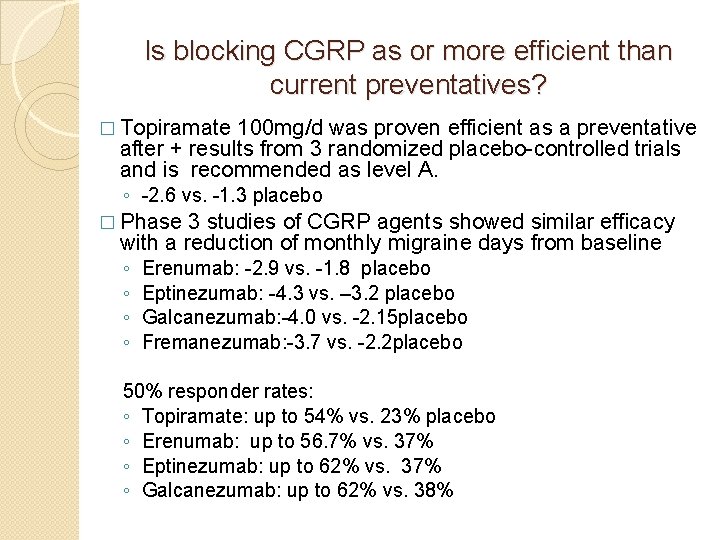
Is blocking CGRP as or more efficient than current preventatives? � Topiramate 100 mg/d was proven efficient as a preventative after + results from 3 randomized placebo-controlled trials and is recommended as level A. ◦ -2. 6 vs. -1. 3 placebo � Phase 3 studies of CGRP agents showed similar efficacy with a reduction of monthly migraine days from baseline ◦ ◦ Erenumab: -2. 9 vs. -1. 8 placebo Eptinezumab: -4. 3 vs. – 3. 2 placebo Galcanezumab: -4. 0 vs. -2. 15 placebo Fremanezumab: -3. 7 vs. -2. 2 placebo 50% responder rates: ◦ Topiramate: up to 54% vs. 23% placebo ◦ Erenumab: up to 56. 7% vs. 37% ◦ Eptinezumab: up to 62% vs. 37% ◦ Galcanezumab: up to 62% vs. 38%

Safety and Tolerability Data from phase II and III trials � No significant change in vital signs or ECGs. � No change in hepatic enzymes which was seen with initial small molecule CGRP receptor antagonist telcagepant. � Most common treatment-related adverse event was injection site reaction. � Although all monocolonal ab have been humanized to reduce immunogenicity, antibodies against these treatments can still form. � Anti-drug antibodies did not appear to affect drug concentration, efficacy, or adverse events.


erenumab-aooe - Aimovig �FDA Approval Date: 05/17/2018 �Amgen, Inc. �Erenumab-aooe is a human monoclonal antibody that binds to the calcitonin generelated peptide (CGRP) receptor and antagonizes CGRP receptor function. �Indicated for the preventive treatment of migraine in adults.

�The efficacy of AIMOVIG was evaluated as a preventive treatment of episodic or chronic migraine in three randomized, double-blind, placebo-controlled studies: ◦ two studies in patients with episodic migraine (4 to 14 migraine days per month) (Study 1 and Study 2) ◦ one study in patients with chronic migraine (≥ 15 headache days per month with ≥ 8 migraine days per month) (Study 3). �The studies enrolled patients with a history of migraine, with or without aura, according to the International Classification of Headache Disorders (ICHD-III) diagnostic
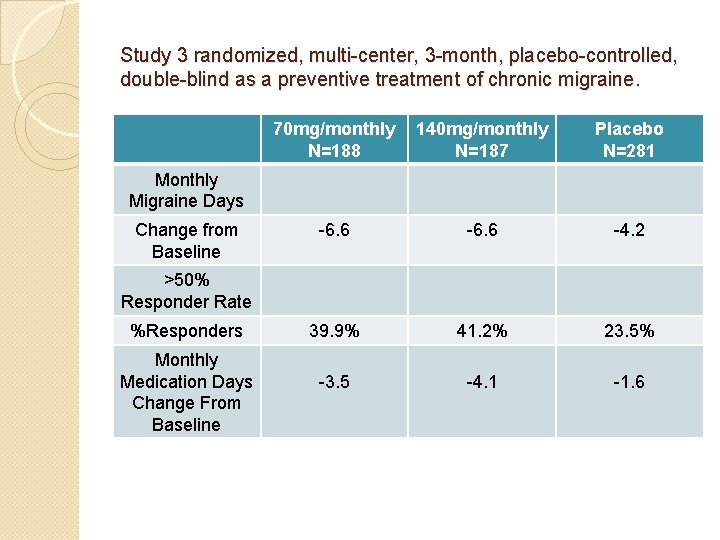
Study 3 randomized, multi-center, 3 -month, placebo-controlled, double-blind as a preventive treatment of chronic migraine. 70 mg/monthly N=188 140 mg/monthly N=187 Placebo N=281 -6. 6 -4. 2 39. 9% 41. 2% 23. 5% -3. 5 -4. 1 -1. 6 Monthly Migraine Days Change from Baseline >50% Responder Rate %Responders Monthly Medication Days Change From Baseline

Aimovig Administration �For subcutaneous use only. Injection: 70 mg/m. L solution in a single-dose prefilled Sure. Click® autoinjector. �The recommended dosage is 70 mg injected SC once monthly. �Some patients may benefit from a dosage of 140 mg once monthly, which is administered as two consecutive SC injections of 70 mg each. �Administer in the abdomen, thigh, or upper arm SC.

Aimovig Side Effects: �Most common adverse reactions in clinical studies (occurring in at least 3% of treated patients and more often than placebo) ◦ Injection site reactions ◦ Constipation

Aimovig Pharmacodynamics/Kinetics �Half-life elimination: 28 days �Time to peak: ~6 days

Aimovig Specific Populations � Pregnancy: no adequate data. � Elderly: no adequate data over 65 yoa. � Allergic to rubber/latex. Needle shield is within the white cap of the Sure. Click autoinjectors and the gray needle cap of the single-dose prefilled syringes contain dry natural rubber. � Hepatic/Renal Failure: Population pharmacokinetic analysis of integrated data from the AIMOVIG clinical studies did not reveal a difference in the pharmacokinetics in patients with mild or moderate renal impairment relative to those with normal renal function. � Patients with severe renal impairment (e. GFR < 30) have not been studied. � No dedicated clinical studies were conducted to evaluate the effect of hepatic impairment or renal impairment on the pharmacokinetics of erenumab

Aimovig Drug Interaction �Not metabolized by cytochrome P 450 enzymes. �In an open-label drug interaction study in healthy female volunteers, (140 mg SC single-dose) did not affect the pharmacokinetics of a combined oral contraceptive containing ethinyl estradiol and norgestimate.
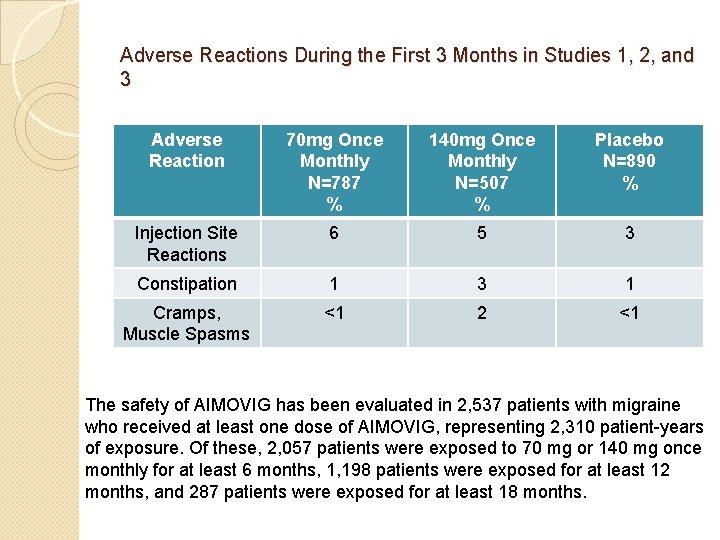
Adverse Reactions During the First 3 Months in Studies 1, 2, and 3 Adverse Reaction 70 mg Once Monthly N=787 % 140 mg Once Monthly N=507 % Placebo N=890 % Injection Site Reactions 6 5 3 Constipation 1 3 1 Cramps, Muscle Spasms <1 2 <1 The safety of AIMOVIG has been evaluated in 2, 537 patients with migraine who received at least one dose of AIMOVIG, representing 2, 310 patient-years of exposure. Of these, 2, 057 patients were exposed to 70 mg or 140 mg once monthly for at least 6 months, 1, 198 patients were exposed for at least 12 months, and 287 patients were exposed for at least 18 months.

Aimovig Storage �Store refrigerated 36°to 46°F in the original carton to protect from light until time of use. �If removed from the refrigerator, keep at room temperature up to 77°F in the original carton and must be used within 7 days. �Do not freeze.

Aimovig �Pricing: US -Disclaimer: reference price only. �Solution Auto-injector � 70 mg/m. L (per m. L): $345. 00

Galcanezumab - Emgality FDA approved 09/2018 Eli Lilly and Co Galcanezumab-gnlm is a humanized monoclonal antibody that binds to calcitonin gene-related peptide (CGRP) ligand blocks its binding to the receptor. 120 -mg self-administered, SQ injection. Recommended dose is 240 mg once as a loading dose, administered in two consecutive injections of 120 mg each, followed by monthly injections at 120 mg.

Emgality Pharmacodynamics/Kinetics �Loading dose of 240 mg achieved the serum steady-state concentration after the first dose. �The time to maximum concentration is 5 days, �Elimination half-life is 27 days.
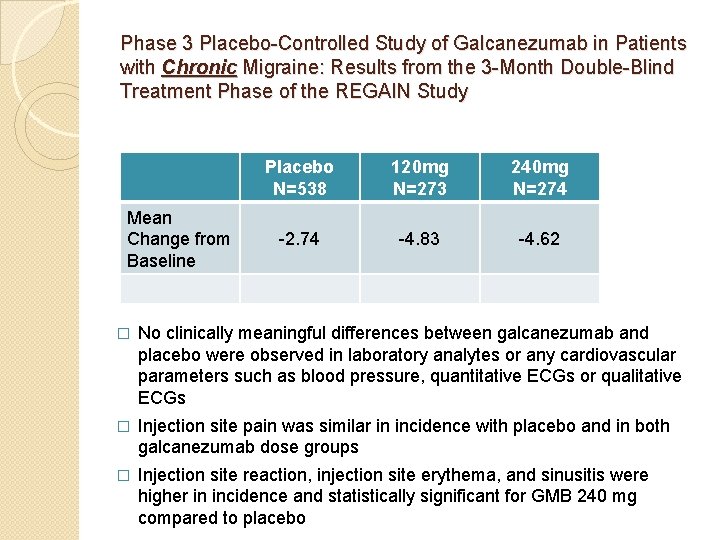
Phase 3 Placebo-Controlled Study of Galcanezumab in Patients with Chronic Migraine: Results from the 3 -Month Double-Blind Treatment Phase of the REGAIN Study Mean Change from Baseline Placebo N=538 120 mg N=273 240 mg N=274 -2. 74 -4. 83 -4. 62 � No clinically meaningful differences between galcanezumab and placebo were observed in laboratory analytes or any cardiovascular parameters such as blood pressure, quantitative ECGs or qualitative ECGs � Injection site pain was similar in incidence with placebo and in both galcanezumab dose groups � Injection site reaction, injection site erythema, and sinusitis were higher in incidence and statistically significant for GMB 240 mg compared to placebo
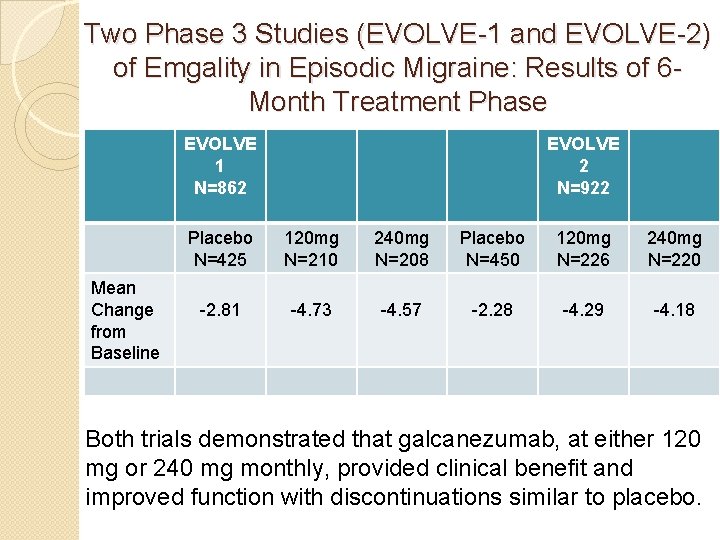
Two Phase 3 Studies (EVOLVE-1 and EVOLVE-2) of Emgality in Episodic Migraine: Results of 6 Month Treatment Phase EVOLVE 1 N=862 Mean Change from Baseline EVOLVE 2 N=922 Placebo N=425 120 mg N=210 240 mg N=208 Placebo N=450 120 mg N=226 240 mg N=220 -2. 81 -4. 73 -4. 57 -2. 28 -4. 29 -4. 18 Both trials demonstrated that galcanezumab, at either 120 mg or 240 mg monthly, provided clinical benefit and improved function with discontinuations similar to placebo.
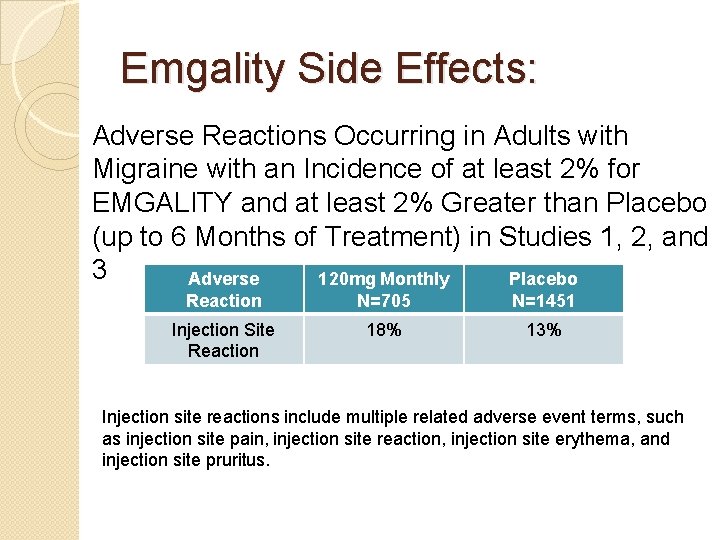
Emgality Side Effects: Adverse Reactions Occurring in Adults with Migraine with an Incidence of at least 2% for EMGALITY and at least 2% Greater than Placebo (up to 6 Months of Treatment) in Studies 1, 2, and 3 Adverse 120 mg Monthly Placebo Reaction N=705 N=1451 Injection Site Reaction 18% 13% Injection site reactions include multiple related adverse event terms, such as injection site pain, injection site reaction, injection site erythema, and injection site pruritus.

Emgality Specific Populations �Pregnancy: no adequate data. �Elderly: no adequate data over 65 yoa. �Not made with natural rubber latex. �Creatinine clearance did not affect the pharmacokinetics in patients with mild or moderate renal impairment. Patients with severe renal impairment (creatinine clearance <30) have not been studied. �No dedicated clinical studies were conducted to evaluate the effect of hepatic impairment or renal impairment on the pharmacokinetics.

Emgality Drug Interaction �Not metabolized by cytochrome P 450 enzymes; therefore, interactions with concomitant medications that are substrates, inducers, or inhibitors of cytochrome P 450 enzymes are unlikely.

Emgality Storage �Store refrigerated at 36°F to 46°F in the original carton to protect EMGALITY from light until use. �Do not freeze. �If removed from the refrigerator, keep at room temperature up to 77°F in the original carton and must be used within 7 days.

Fremanezumab - Ajovy Fremanezumab FDA approved 09/2018 Ajovy, Teva Pharmaceutical Fremanezumab-vfrm is a humanized monoclonal antibody that binds to calcitonin gene-related peptide (CGRP) ligand blocks its binding to the receptor. Two subcutaneous dosing options of AJOVY are available to administer the recommended dosage: • 225 mg monthly, or • 675 mg every 3 months (quarterly), which is administered as three consecutive subcutaneous injections of 225 mg each.

Ajovy Pharmacodynamics/Kinetics �After single SC administrations of 225 mg and 675 mg, median time to maximum concentrations (tmax) was 5 to 7 days. �Metabolism: Degraded by enzymatic proteolysis into small peptides and amino acids. �Half-life elimination: ~31 days. �Time to peak: 5 to 7 days. �Steady state was achieved by approximately 168 days (about 6 months) following 225 mg SC monthly and 675 mg SC quarterly dosing

Ajovy - Phase III HALO study � 12 -week treatment period � 1130 study participants were randomly assigned to receive SC injections ◦ 675 mg at initiation followed by monthly 225 mg for 2 months (monthly dose regimen) ◦ 675 mg at initiation followed by placebo for 2 months (quarterly dose regimen) ◦ three monthly doses of matching placebo.
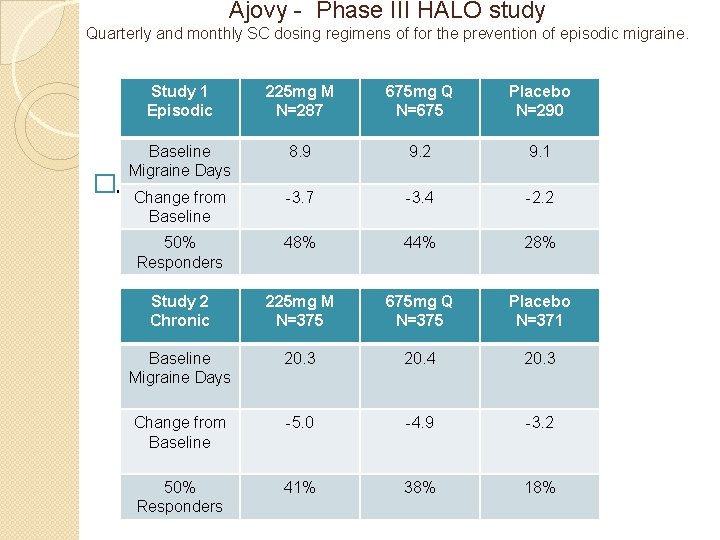
Ajovy - Phase III HALO study Quarterly and monthly SC dosing regimens of for the prevention of episodic migraine. �. Study 1 Episodic 225 mg M N=287 675 mg Q N=675 Placebo N=290 Baseline Migraine Days 8. 9 9. 2 9. 1 Change from Baseline -3. 7 -3. 4 -2. 2 50% Responders 48% 44% 28% Study 2 Chronic 225 mg M N=375 675 mg Q N=375 Placebo N=371 Baseline Migraine Days 20. 3 20. 4 20. 3 Change from Baseline -5. 0 -4. 9 -3. 2 50% Responders 41% 38% 18%

Ajovy Specific Populations �Pregnancy: no adequate data. �Elderly: no adequate data over 65 yoa. �Hepatic/Renal Impairment: There are no dosage adjustments provided in the manufacturer’s labeling. �Hepatic/Renal impairment is not expected to affect the pharmacokinetics of fremanezumabvfrm. �Patients with severe renal impairment (e. GFR <

Ajovy Storage/Administration � For subcutaneous use only. � Keep out of direct sunlight and allow prefilled syringe to come to room temperature for 30 minutes before administration. � Do not warm by using a heat source (eg, hot water, microwave). � Do not shake. � Administer in the abdomen, thigh, or upper arm, avoiding areas that are tender, bruised, red, or indurated. � The 675 mg dose should be administered as 3 consecutive 225 mg injections. For multiple injections,

Ajovy �Pricing: US -Disclaimer: reference price only. �Solution Prefilled Syringe (Ajovy SC) � 225 mg/1. 5 m. L (per m. L): $460. 00

Eptinezumab �Alder Bio. Pharmaceuticals �Eptinezumab (formerly known as ALD 403) was designed to selectively bind to the CGRP ligand with high affinity to block its effect at all CGRP receptors. �Currently it is in Phase 3 trials (PROMISE 1 episodic and II chronic) being evaluated as a quarterly infusion allowing the total dose administered to be immediately active to inhibit CGRP with 100% bioavailability

Eptinezumab �PROMISE-2, enrolled 1072 pts 18 to 65 years old with chronic migraine. �Infusion of 100 mg (n = 356; 86% women) �Infusion of 300 mg (n = 350; 90% women) �Matching placebo (n = 366; 89% women) �Baseline, mean MMDs were 16. 1, and 16. 2 for the three groups, respectively. �Over weeks 1 to 12, both study drug groups had significantly greater reductions in baseline MMDs (change, – 7. 7 and – 8. 2) than the placebo group ( – 5. 6; both comparisons, P <. 0001).

Eptinezumab -PROMISE 1&2 The most frequently reported AEs for 100 mg, 300 mg and Placebo: �nasopharyngitis (4%, 6%, and 4%) �upper respiratory tract infection (3%, 4%, and 4%) �nausea (2%, 3%, and 2%) �urinary tract infection (2%, 3%, and 2%) �dizziness (1%, 3%, and 1%).

CGRP antagonism Long Term Effects? With new molecular entities “You don't know what you don't know“ All companies have stated they are conducting long-term safety studies.

References: � Brauser, Deborah. Eptinezumab Safe, Effective for Migraine Prevention. Medscape. 6. 21. 2018. � Edvinsson, Lars. The Journey to Establish CGRP as a Migraine Target: A Retrospective View. Headache. 2015; 00: 00 -00. � Goaddsby, Peter. Anti-CGRP Monoclonal Ab: the Next Era of Migraine Prevention? Curr Treat Options Neurology. 2017; 19: 27. � Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3 rd edition. Cephalalgia 2018, Vol. 38(1) 1– 211. � Maassen Van Den Brink, A. Blocking CGRP in migraine patients-a review of pros and cons. The Journal of Headache and Pain. 2017; 18: 96. � Rapoport, Alan. Calcitonin Gene Related Peptide and Migraine Current Understanding and State of Development. Headache. 2013; 53: 1230 -1244. � Tepper, Stewart. Expert Perspectives-Migraine Prevention for Highly Impacted Patients. US Neurology. 2018; 14 (Suppl. 2)3 -10. � Up To Date. 10. 2018. Erenumab-aooe: Drug information, Fremanezumabvfrm: Drug information, Galcanezumab-gnlm: Drug information
- Slides: 58