Moles Molar Mass 10 1 The Mole Molar





















- Slides: 23

Moles & Molar Mass 10. 1 The Mole &Molar Mass 10. 2 Mole Relationships in Chemical Equations 12. 2 Mass Calculations for Reactions 1

Collection Terms 1 trio = 3 singers 1 six-pack Cola = 6 cans Cola drink 1 dozen donuts = 12 donuts 1 gross of pencils = 144 pencils 2

A Moles of Particles Contains 6. 02 x 1023 particles 1 mole C = 6. 02 x 1023 C atoms 1 mole H 2 O = 6. 02 x 1023 H 2 O molecules 1 mole Na. Cl = 6. 02 x 1023 Na+ ions and 6. 02 x 1023 Cl– ions 3

Examples of Moles of elements 1 mole Mg = 6. 02 x 1023 Mg atoms 1 mole Au = 6. 02 x 1023 Au atoms Moles of compounds 1 mole NH 3 = 6. 02 x 1023 NH 3 molecules 1 mole C 9 H 8 O 4 = 6. 02 x 1023 aspirin molecules 4

Avogadro’s Number 6. 02 x 1023 particles 1 mole or 1 mole 6. 02 x 1023 particles 5

Learning Check Suppose we invented a new collection unit called a mep. One mep contains 8 objects. A. How many paper clips in 1 mep? 1) 1 2) 4 3) 8 B. How many oranges in 2. 0 meps? 1) 4 2) 8 3) 16 C. How many meps contain 40 gummy bears? 1) 5 2) 10 3) 20 6

Solution Suppose we invented a new collection unit called a mep. One mep contains 8 objects. A. How many paper clips in 1 mep? 3) 8 B. How many oranges in 2. 0 meps? 3) 16 C. How many meps contain 40 gummy bears? 1) 5 7

Molar Mass n Number of grams in 1 mole n Equal to the numerical value of the atomic mass 1 mole of C atoms = 12. 0 g 1 mole of Mg atoms = 24. 3 g 1 mole of Cu atoms = 63. 5 g 8

Learning Check Give the molar mass to 0. 1 g A. 1 mole of Br atoms = ____ B. 1 mole of Sn atoms = ____ 9

Solution Give the molar mass to 0. 1 g A. 1 mole of Br atoms = 79. 9 g/mole B. 1 mole of Sn atoms = 118. 7 g/mole 10

Molar Mass of Compounds Mass in grams of 1 mole equal numerically to the sum of the atomic masses 1 mole of Ca. Cl 2 = 111. 1 g/mole 1 mole Ca x 40. 1 g/mole + 2 moles Cl x 35. 5 g/mole 1 mole of N 2 O 4 = 92. 0 g/mole 2 moles N x 14. 0 g/mole + 4 moles O x 16. 0 g/mole 11

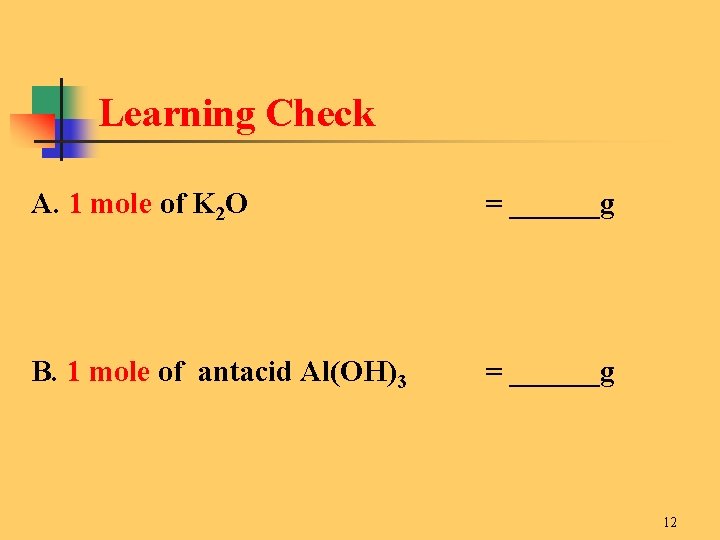
Learning Check A. 1 mole of K 2 O = ______g B. 1 mole of antacid Al(OH)3 = ______g 12

Solution A. 1 mole of K 2 O = 94. 2 g 2 K x 39. 1 g/mole + 1 O x 16. 0 g/mole B. 1 mole of antacid Al(OH)3 = 78. 0 g 1 Al x 27. 0 g/mole + 3 O x 16. 0 g/mole + 3 x 1. 0 13

Learning Check Prozac, C 17 H 18 F 3 NO, is a widely used antidepressant that inhibits the uptake of serotonin by the brain. It has a molar mass of 1) 40. 0 g/mole 2) 262 g/mole 3) 309 g/mole 14

Solution Prozac, C 17 H 18 F 3 NO, is a widely used antidepressant that inhibits the uptake of serotonin by the brain. It has a molar mass of 3) 309 g/mole 17 C (12. 0) + 18 H (1. 0) + 3 F (19. 0) + 1 N (14. 0) + 1 O (16. 0) 15

Molar Mass Factors Methane CH 4 known as natural gas is used in gas cook tops and gas heaters. Express the molar mass of methane in the form of conversion factors. Molar mass of CH 4 = 16. 0 g CH 4 1 mole CH 4 and 16. 0 g 1 mole CH 4 16. 0 g CH 4 16

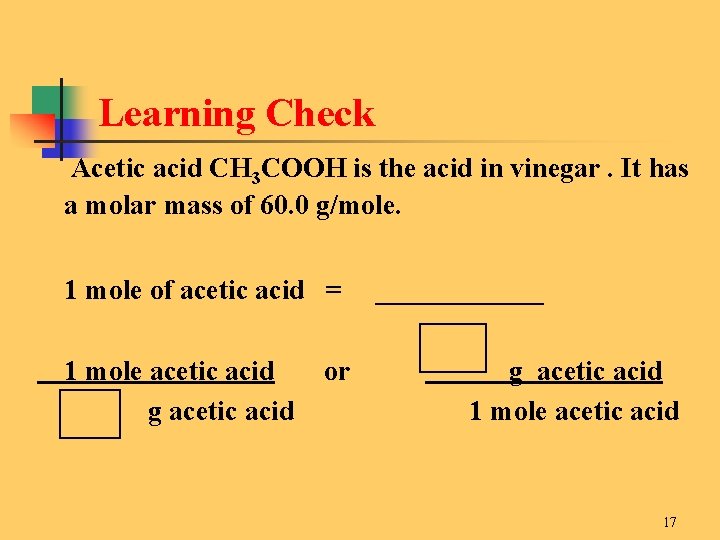
Learning Check Acetic acid CH 3 COOH is the acid in vinegar. It has a molar mass of 60. 0 g/mole. 1 mole of acetic acid = 1 mole acetic acid g acetic acid or ______ g acetic acid 1 mole acetic acid 17

Solution Acetic acid CH 3 COOH is the acid in vinegar. It has a molar mass of 60. 0 g/mole. 1 mole of acetic acid = 1 mole acetic acid 60. 0 g acetic acid or 60. 0 g acetic acid 1 mole acetic acid 18

Calculations with Molar Mass molar mass Grams Moles 19

Moles and Grams Aluminum is often used for the structure of light-weight bicycle frames. How many grams of Al are in 3. 00 moles of Al? 3. 00 moles Al ? g Al 20

1. Molar mass of Al 1 mole Al = 27. 0 g Al 2. Conversion factors for Al 27. 0 g Al 1 mol Al 3. Setup or 1 mol Al 27. 0 g Al 3. 00 moles Al Answer x 27. 0 g Al 1 mole Al = 81. 0 g Al 21

Learning Check The artificial sweetener aspartame (Nutri. Sweet) formula C 14 H 18 N 2 O 5 is used to sweeten diet foods, coffee and soft drinks. How many moles of aspartame are present in 225 g of aspartame? 22

Solution 1. Molar mass of Aspartame C 14 H 18 N 2 O 5 (14 x 12. 0) + (18 x 1. 01) + (2 x 14. 0) + (5 x 16. 0) = 294 g/mole 2. Setup 225 g aspartame x 1 mole aspartame 294 g aspartame = 0. 765 mole aspartame 23