Molecules of Life Carbon Hydrogen Nitrogen Oxygen Phosphorus
Molecules of Life

Carbon Hydrogen Nitrogen Oxygen Phosphorus Sulfur

Macromolecules aka: Biomolecules The Molecules of Life!

Macromolecules are… • Large polymers (poly=many) • Built by monomers (mono = one) • 4 main classes (examples of carbon based biomolecules) – Carbohydrates – Lipids – Proteins – Nucleic Acids

Why do we eat? • We eat to take in more of these chemicals – Food for building materials • to make more of us (cells) • for growth • for repair – Food to make energy • calories • to make ATP

How do we make these molecules? We build them! 2006 -2007
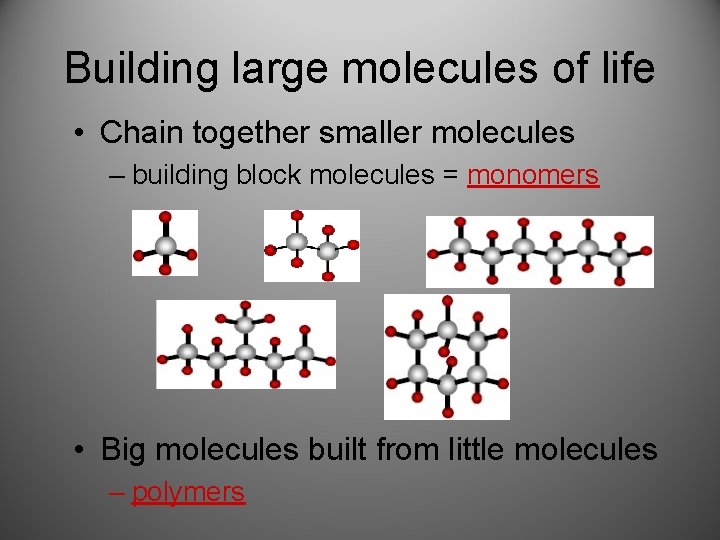
Building large molecules of life • Chain together smaller molecules – building block molecules = monomers • Big molecules built from little molecules – polymers
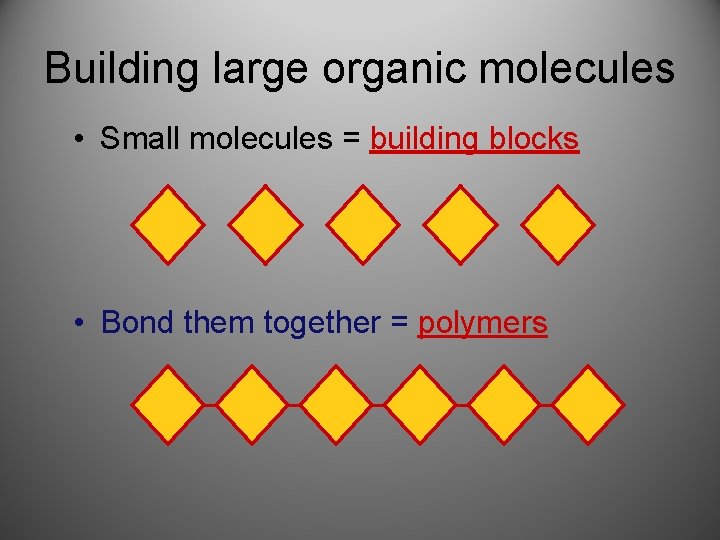
Building large organic molecules • Small molecules = building blocks • Bond them together = polymers

How to build large molecules • Synthesis – building bigger molecules from smaller molecules – building cells & bodies • repair • growth • reproduction + ATP
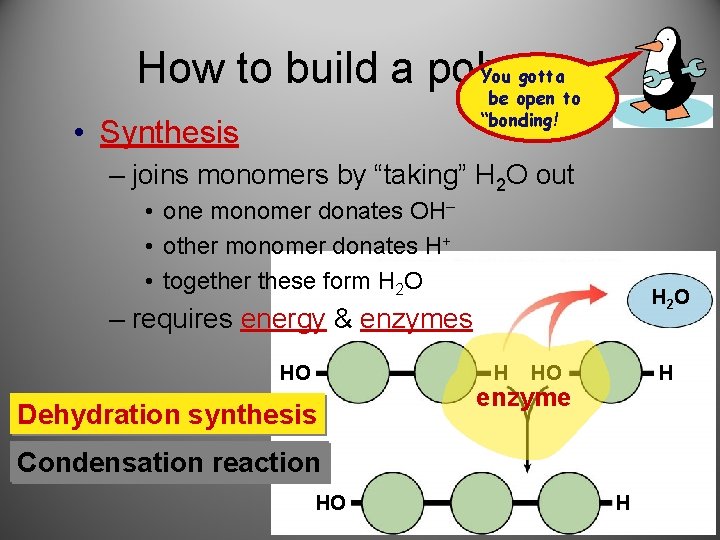
You gotta How to build a polymer be open to “bonding! • Synthesis – joins monomers by “taking” H 2 O out • one monomer donates OH– • other monomer donates H+ • together these form H 2 O H 2 O – requires energy & enzymes HO H Dehydration synthesis HO H enzyme Condensation reaction HO H
How to take large molecules apart • Digestion – taking big molecules apart – getting raw materials • for synthesis & growth – making energy (ATP) • for synthesis, growth & everyday functions + ATP
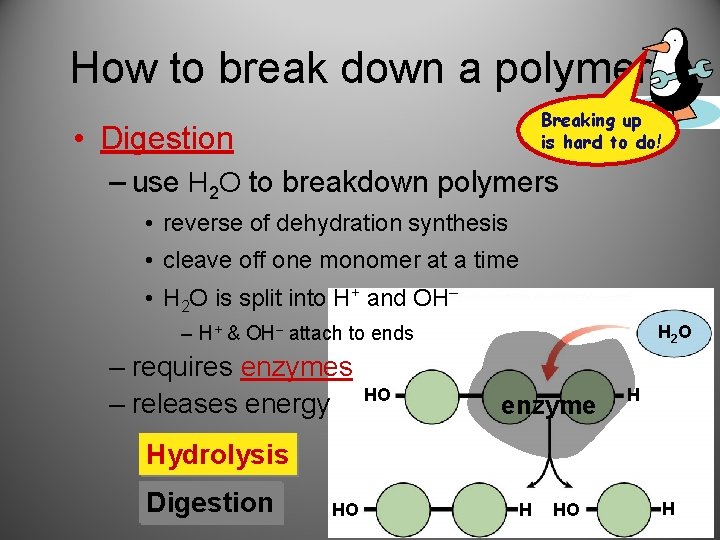
How to break down a polymer Breaking up is hard to do! • Digestion – use H 2 O to breakdown polymers • reverse of dehydration synthesis • cleave off one monomer at a time • H 2 O is split into H+ and OH– – H+ & OH– attach to ends – requires enzymes – releases energy HO H 2 O enzyme H Hydrolysis Digestion HO H
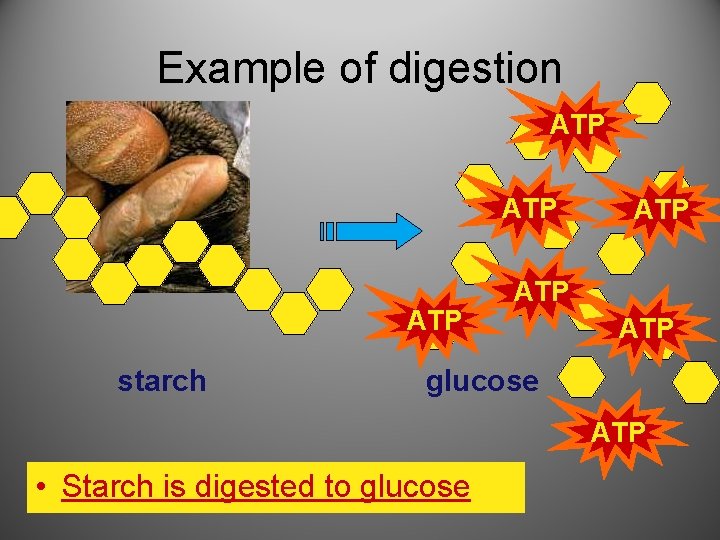
Example of digestion ATP ATP starch ATP ATP glucose ATP • Starch is digested to glucose
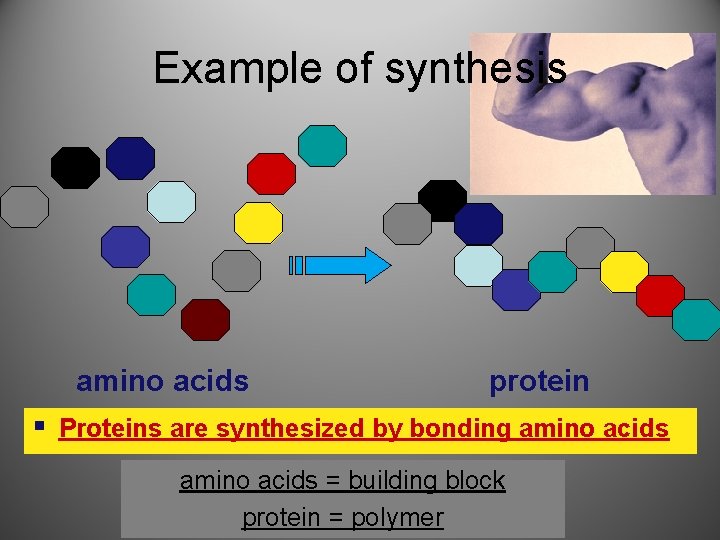
Example of synthesis amino acids protein § Proteins are synthesized by bonding amino acids = building block protein = polymer

Carbohydrates Fuel and Building Material for Life!

Carbohydrates are… Monomer = 1 C: 2 H: 1 O Monosaccharides (simple sugars) • Most abundant carbon compounds found in living things • Sugars (example) = quick energy • Monosaccharides- simple sugars » Glucose C 6 H 12 O 6 • Disaccharides- sugars built of 2 monosaccharides » Sucrose+Fructose

Carbos cont… • Starch (example) = Polysaccharides: act as nutrient storage-and form structural components of living things • Glycogen- stores glucose in muscle tissue for quick energy • Cellulose- provides rigid structure » Cell wall of plant cells
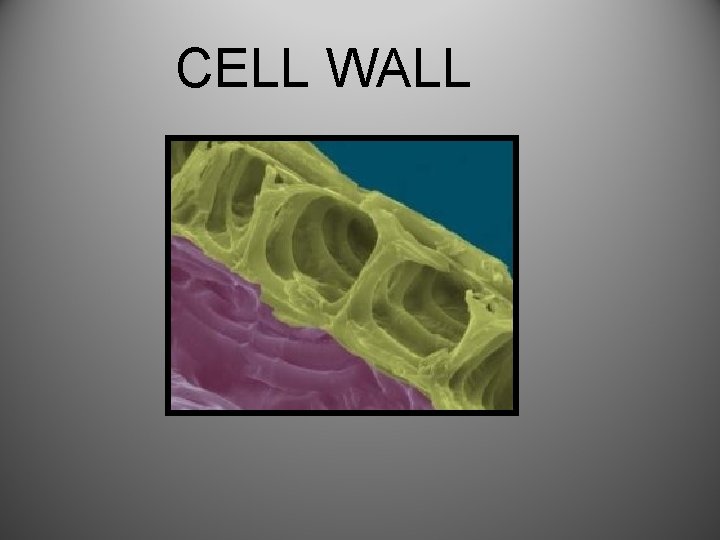
CELL WALL

Monomer: C, H, O - Glycerol and fatty acids LIPIDS • Fats: gylcerol and fatty acids – Saturated • Solidifies- bad • Ex: animal fat and butter – Unsaturated • No solidification- good • Ex: vegetable oils – Fats are used for energy storage • Long-term food reserves stored in adipose (fat)cells

Lipids cont… • Fat (adipose) provides insulation for warmth I’m – Whales, seals Fat! • Fat provides cushioning for organs
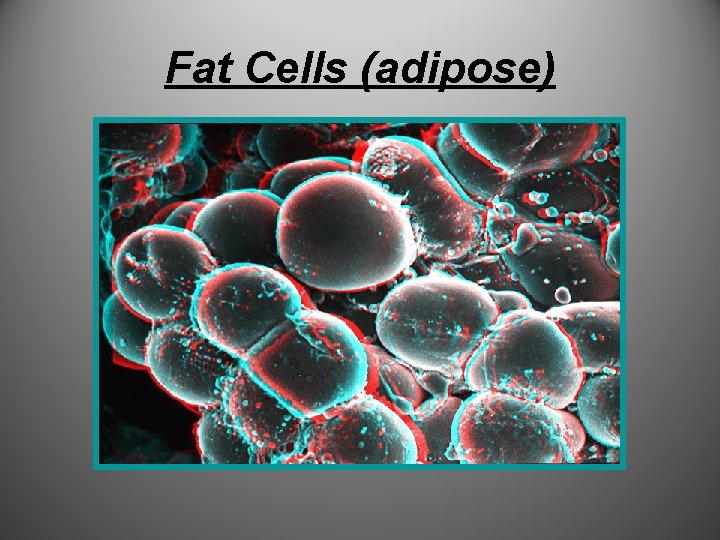
Fat Cells (adipose)

Lipids cont… • Phospholipids – Make up cell membranes The bilayer forms a boundary between the cell and the external environment.
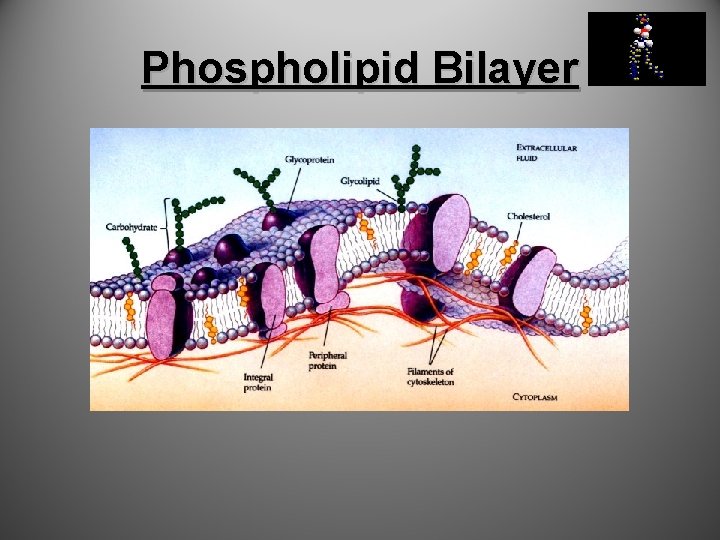
Phospholipid Bilayer

Proteins The Ultimate Polymer!
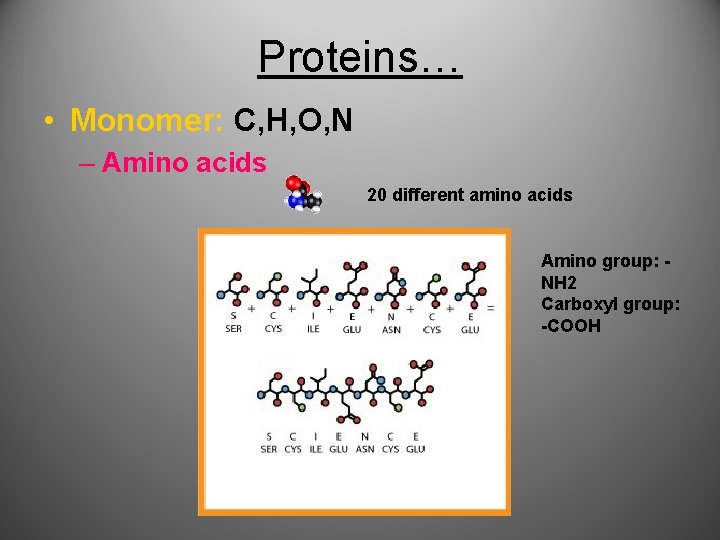
Proteins… • Monomer: C, H, O, N – Amino acids 20 different amino acids Amino group: NH 2 Carboxyl group: -COOH
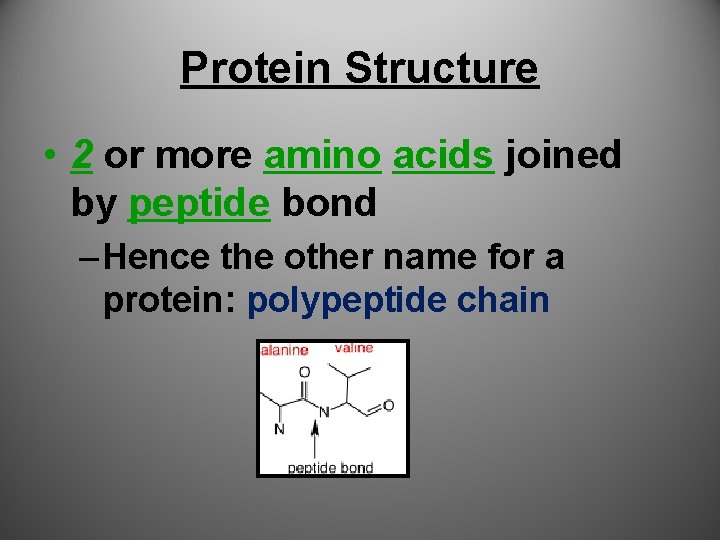
Protein Structure • 2 or more amino acids joined by peptide bond – Hence the other name for a protein: polypeptide chain

Structure=Function • Protein’s specific structure (shape) determines it’s duties (job)
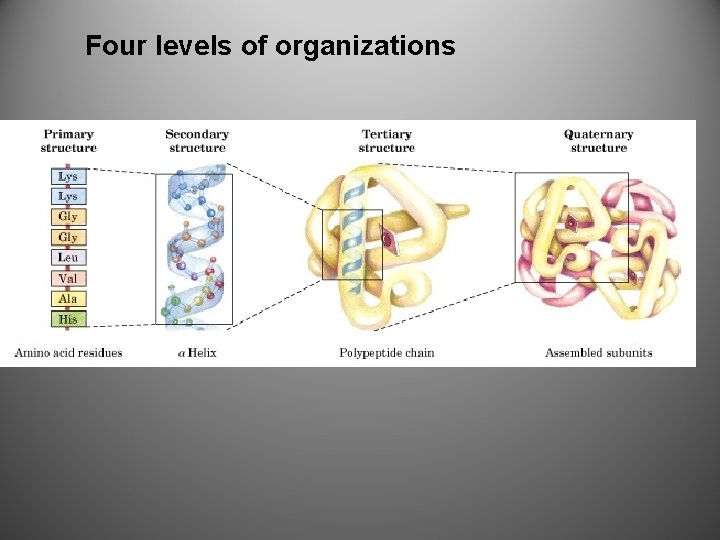
Four levels of organizations

Types of Proteins • *Structural Defensive – Support -Antibodies • Storage – Embryo food • *Transport – In and out of cell • Receptors – Drugs *Enzymes -catalysts Hormones -messages *Contractile -muscles
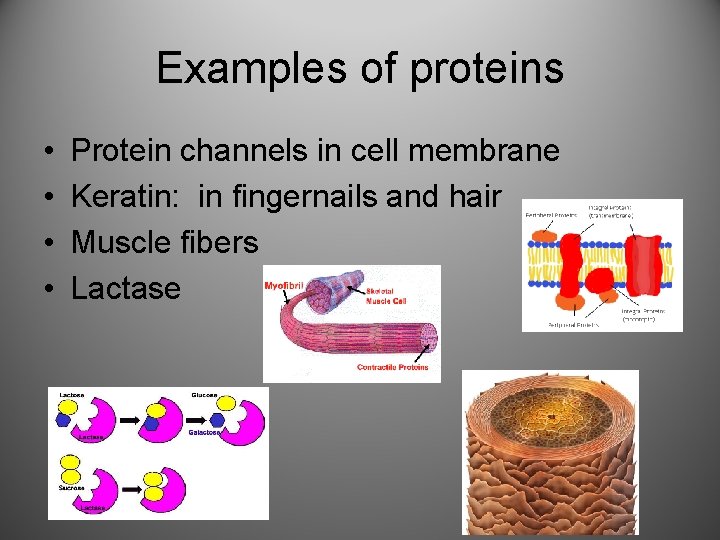
Examples of proteins • • Protein channels in cell membrane Keratin: in fingernails and hair Muscle fibers Lactase

Nucleic Acids Informational Polymers
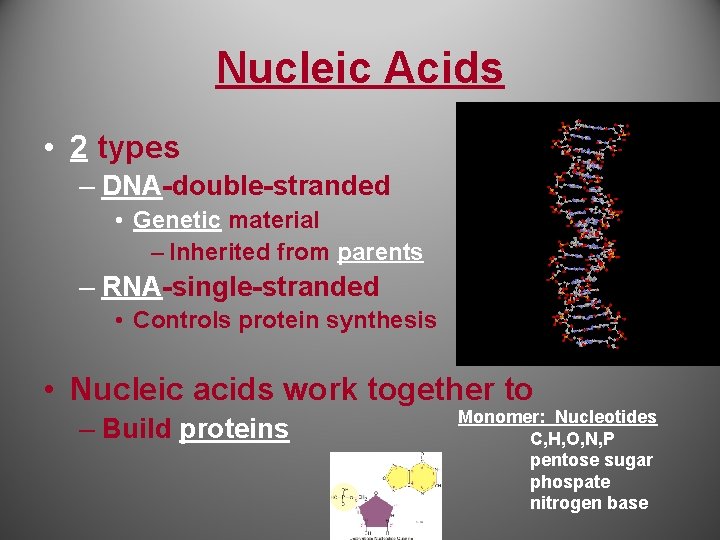
Nucleic Acids • 2 types – DNA-double-stranded • Genetic material – Inherited from parents – RNA-single-stranded • Controls protein synthesis • Nucleic acids work together to – Build proteins Monomer: Nucleotides C, H, O, N, P pentose sugar phospate nitrogen base
- Slides: 32